Pipeline Analysis:
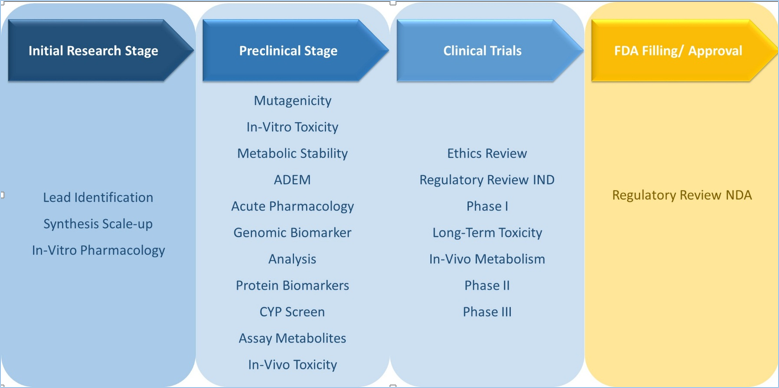
Research and development in pharmaceuticals is a complex and expensive process, drug development comprises the synchronized contributions of experts serving in various disciplines such as clinical, non-clinical, regulatory and commercial. Before entering the market pharmaceutical companies have to overcome several complications and potential pitfalls, which may lead to expensive delays or complete closedown of the process. Our expertise related to comprehensive drug development and distribution strategy helps us to offer an exhaustive study of drug pipeline. Pipeline analysis is a core competency of Credence Research, we have a wide range of reports that cover in-depth information related to drug pipeline focusing on specific diseases. In these reports, we provide sales forecast & expected launch date for the drugs present in phase III of clinical trials. In addition to that a detailed analysis of drugs presents in phase I & II of the clinical trials. Our repository comprises more than 200 reports focusing on diseases which possess strong & promising drug pipeline.
These reports cover important data points related to the drug pipeline of disease as follows:
- Competitive Environment
- Regulatory Policies & Effect on Pipeline Drugs
- Product Launch Strategies
- Pricing Strategies
- Reimbursement Policy Forecast
- Market Penetration Strategies
An elaborate analysis of pipeline drugs is carried out efficiently in our reports, data related to this research is collected through primary and secondary research. Initial secondary research comprises paid data sources and to verify this information primary interviews with key stakeholders aid in interpreting the market dynamics, data verification, and sales forecast.