1. Preface
1.1. Report Description
1.1.1. Purpose of the Report
1.1.2. Target Audience
1.1.3. USP and Key Offerings
1.2. Research Scope
1.3. Market Introduction
2. Executive Summary
2.1. Market Snapshot: Global Gaucher Disease Therapeutics Market
2.1.1. Global Gaucher Disease Therapeutics Market, By Therapy Type
2.1.2. Global Gaucher Disease Therapeutics Market, By Route of Administration
2.1.3. Global Gaucher Disease Therapeutics Market, By End-User
2.1.4. Global Gaucher Disease Therapeutics Market, By Region
2.2. Insights from Primary Respondents
3. Market Dynamics & Factors Analysis
3.1. Introduction
3.1.1. Global Gaucher Disease Therapeutics Market Value, 2019-2032, (US$ Mn)
3.1.2. Y-o-Y Growth Trend Analysis
3.2. Market Dynamics
3.2.1. Gaucher Disease Therapeutics Market Drivers
3.2.2. Gaucher Disease Therapeutics Market Restraints
3.2.3. Gaucher Disease Therapeutics Market Opportunities
3.2.4. Major Gaucher Disease Therapeutics Industry Challenges
3.3. Growth and Development Patterns
3.4. Investment Feasibility Analysis
3.5. Market Opportunity Analysis
3.5.1. Therapy Type
3.5.2. Route of Administration
3.5.3. End-User
3.5.4. Geography
4. Market Competitive Landscape Analysis
4.1. Company Market Share Analysis, 2023
4.1.1. Global Gaucher Disease Therapeutics Market: Company Market Share, Value 2023
4.1.2. Global Gaucher Disease Therapeutics Market: Top 6 Company Market Share, Value 2023
4.1.3. Global Gaucher Disease Therapeutics Market: Top 3 Company Market Share, Value 2023
4.2. Global Gaucher Disease Therapeutics Market: Company Revenue Share Analysis, 2023
4.3. Company Assessment Metrics, 2023
4.3.1. Stars
4.3.2. Emerging Leaders
4.3.3. Pervasive Players
4.3.4. Participants
4.4. Startups/ SMEs Assessment Metrics, 2023
4.4.1. Progressive Companies
4.4.2. Responsive Companies
4.4.3. Dynamic Companies
4.4.4. Starting Blocks
4.5. Strategic Development
4.5.1. Acquisition and Mergers
4.5.2. New Product Launch
4.5.3. Regional Expansion
4.5.4. Partnerships
4.6. Key Player Product Matrix
4.7. Potential for New Players in the Global Gaucher Disease Therapeutics Market
5. Premium Insights
5.1. STAR (Situation, Task, Action, Results) Analysis
5.2. Porter’s Five Forces Analysis
5.2.1. Threat of New Entrants
5.2.2. Bargaining Power of Buyers/Consumers
5.2.3. Bargaining Power of Suppliers
5.2.4. Threat of Substitute Types
5.2.5. Intensity of Competitive Rivalry
5.3. PESTEL Analysis
5.3.1. Political Factors
5.3.2. Economic Factors
5.3.3. Social Factors
5.3.4. Technological Factors
5.3.5. Environmental Factors
5.3.6. Legal Factors
5.4. Key Market Trends
5.4.1. Demand Side Trends
5.4.2. Supply Side Trends
5.5. Value Chain Analysis
5.6. Technology Analysis
5.6.1. Research and development in the global market
5.6.2. Patent Analysis
5.6.3. Emerging technologies and their potential disruption to the market
5.7. Consumer Behaviour Analysis
5.7.1. Consumer Preferences and Expectations
5.7.2. Factors Influencing Consumer Buying Decisions
5.7.2.1. North America
5.7.2.2. Europe
5.7.2.3. Asia Pacific
5.7.2.4. Latin America
5.7.2.5. Middle East and Africa
5.7.3. Consumer Pain Points
5.8. Analysis and Recommendations
5.9. Adjacent Market Analysis
6. Market Positioning of Key Players, 2023
6.1. Company market share of key players, 2023
6.2. Competitive Benchmarking
6.3. Market Positioning of Key Vendors
6.4. Geographical Presence Analysis
6.5. Major Strategies Adopted by Key Players
6.5.1. Key Strategies Analysis
6.5.2. Mergers and Acquisitions
6.5.3. Partnerships
6.5.4. Product Launch
6.5.5. Geographical Expansion
6.5.6. Others
7. Impact Analysis of COVID 19 and Russia – Ukraine War on Gaucher Disease Therapeutics Market
7.1. Ukraine-Russia War Impact
7.1.1. Uncertainty and Economic Instability
7.1.2. Supply chain disruptions
7.1.3. Regional market shifts
7.1.4. Shift in government priorities
7.2. COVID-19 Impact Analysis
7.2.1. Supply Chain Disruptions
7.2.2. Demand Fluctuations
7.2.3. Shift in Product Mix
7.2.4. Reduced Industrial Activity
7.2.5. Regional Impact Analysis
7.2.5.1. North America
7.2.5.2. Europe
7.2.5.3. Asia Pacific
7.2.5.4. Latin America
7.2.5.5. Middle East and Africa
8. Global Gaucher Disease Therapeutics Market, By Therapy Type
8.1. Global Gaucher Disease Therapeutics Market Overview, by Therapy Type
8.1.1. Global Gaucher Disease Therapeutics Market Revenue Share, By Therapy Type, 2023 Vs 2032 (in %)
8.2. Enzyme Replacement Therapy (ERT)
8.2.1. Global Gaucher Disease Therapeutics Market, By Enzyme Replacement Therapy (ERT), By Region, 2019-2032 (US$ Mn)
8.2.2. Market Dynamics for Enzyme Replacement Therapy (ERT)
8.2.2.1. Drivers
8.2.2.2. Restraints
8.2.2.3. Opportunities
8.2.2.4. Trends
8.3. Substrate Reduction Therapy (SRT)
8.3.1. Global Gaucher Disease Therapeutics Market, By Substrate Reduction Therapy (SRT), By Region, 2019-2032 (US$ Mn)
8.3.2. Market Dynamics for Substrate Reduction Therapy (SRT)
8.3.2.1. Drivers
8.3.2.2. Restraints
8.3.2.3. Opportunities
8.3.2.4. Trends
8.4. Pharmacological Chaperones
8.4.1. Global Gaucher Disease Therapeutics Market, By Pharmacological Chaperones, By Region, 2019-2032 (US$ Mn)
8.4.2. Market Dynamics for Pharmacological Chaperones
8.4.2.1. Drivers
8.4.2.2. Restraints
8.4.2.3. Opportunities
8.4.2.4. Trends
8.5. Substrate Inhibitors
8.5.1. Global Gaucher Disease Therapeutics Market, By Substrate Inhibitors, By Region, 2019-2032 (US$ Mn)
8.5.2. Market Dynamics for Substrate Inhibitors
8.5.2.1. Drivers
8.5.2.2. Restraints
8.5.2.3. Opportunities
8.5.2.4. Trends
8.6. Gene Therapy
8.6.1. Global Gaucher Disease Therapeutics Market, By Gene Therapy, By Region, 2019-2032 (US$ Mn)
8.6.2. Market Dynamics for Gene Therapy
8.6.2.1. Drivers
8.6.2.2. Restraints
8.6.2.3. Opportunities
8.6.2.4. Trends
8.7. Supportive Therapies
8.7.1. Global Gaucher Disease Therapeutics Market, By Supportive Therapies, By Region, 2019-2032 (US$ Mn)
8.7.2. Market Dynamics for Supportive Therapies
8.7.2.1. Drivers
8.7.2.2. Restraints
8.7.2.3. Opportunities
8.7.2.4. Trends
9. Global Gaucher Disease Therapeutics Market, By Route of Administration
9.1. Global Gaucher Disease Therapeutics Market Overview, by Route of Administration
9.1.1. Global Gaucher Disease Therapeutics Market Revenue Share, By Route of Administration, 2023 Vs 2032 (in %)
9.2. Intravenous (IV) Administration
9.2.1. Global Gaucher Disease Therapeutics Market, By Intravenous (IV) Administration, By Region, 2019-2032 (US$ Mn)
9.2.2. Market Dynamics for Intravenous (IV) Administration
9.2.2.1. Drivers
9.2.2.2. Restraints
9.2.2.3. Opportunities
9.2.2.4. Trends
9.3. Oral Administration
9.3.1. Global Gaucher Disease Therapeutics Market, By Oral Administration, By Region, 2019-2032 (US$ Mn)
9.3.2. Market Dynamics for Oral Administration
9.3.2.1. Drivers
9.3.2.2. Restraints
9.3.2.3. Opportunities
9.3.2.4. Trends
10. Global Gaucher Disease Therapeutics Market, By End-User
10.1. Global Gaucher Disease Therapeutics Market Overview, by End-User
10.1.1. Global Gaucher Disease Therapeutics Market Revenue Share, By End-User, 2023 Vs 2032 (in %)
10.2. Hospitals and Clinics
10.2.1. Global Gaucher Disease Therapeutics Market, By Hospitals and Clinics, By Region, 2019-2032 (US$ Mn)
10.2.2. Market Dynamics for Hospitals and Clinics
10.2.2.1. Drivers
10.2.2.2. Restraints
10.2.2.3. Opportunities
10.2.2.4. Trends
10.3. Specialty Clinics
10.3.1. Global Gaucher Disease Therapeutics Market, By Specialty Clinics, By Region, 2019-2032 (US$ Mn)
10.3.2. Market Dynamics for Specialty Clinics
10.3.2.1. Drivers
10.3.2.2. Restraints
10.3.2.3. Opportunities
10.3.2.4. Trends
10.4. Home Care Settings
10.4.1. Global Gaucher Disease Therapeutics Market, By Home Care Settings, By Region, 2019-2032 (US$ Mn)
10.4.2. Market Dynamics for Home Care Settings
10.4.2.1. Drivers
10.4.2.2. Restraints
10.4.2.3. Opportunities
10.4.2.4. Trends
11. Global Gaucher Disease Therapeutics Market, By Region
11.1. Global Gaucher Disease Therapeutics Market Overview, by Region
11.1.1. Global Gaucher Disease Therapeutics Market, By Region, 2023 Vs 2032 (in%)
11.2. Therapy Type
11.2.1. Global Gaucher Disease Therapeutics Market, By Therapy Type, 2019-2032 (US$ Mn)
11.3. Route of Administration
11.3.1. Global Gaucher Disease Therapeutics Market, By Route of Administration, 2019-2032 (US$ Mn)
11.4. End-User
11.4.1. Global Gaucher Disease Therapeutics Market, By End-User, 2019-2032 (US$ Mn)
12. North America Gaucher Disease Therapeutics Market Analysis
12.1. Overview
12.1.1. Market Dynamics for North America
12.1.1.1. Drivers
12.1.1.2. Restraints
12.1.1.3. Opportunities
12.1.1.4. Trends
12.2. North America Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2032(US$ Mn)
12.2.1. Overview
12.2.2. SRC Analysis
12.3. North America Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2032(US$ Mn)
12.3.1. Overview
12.3.2. SRC Analysis
12.4. North America Gaucher Disease Therapeutics Market, by End-User, 2019-2032(US$ Mn)
12.4.1. Overview
12.4.2. SRC Analysis
12.5. North America Gaucher Disease Therapeutics Market, by Country, 2019-2032 (US$ Mn)
12.5.1. North America Gaucher Disease Therapeutics Market, by Country, 2023 Vs 2032 (in%)
12.5.2. U.S.
12.5.3. Canada
12.5.4. Mexico
13. Europe Gaucher Disease Therapeutics Market Analysis
13.1. Overview
13.1.1. Market Dynamics for Europe
13.1.1.1. Drivers
13.1.1.2. Restraints
13.1.1.3. Opportunities
13.1.1.4. Trends
13.2. Europe Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2032(US$ Mn)
13.2.1. Overview
13.2.2. SRC Analysis
13.3. Europe Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2032(US$ Mn)
13.3.1. Overview
13.3.2. SRC Analysis
13.4. Europe Gaucher Disease Therapeutics Market, by End-User, 2019-2032(US$ Mn)
13.4.1. Overview
13.4.2. SRC Analysis
13.5. Europe Gaucher Disease Therapeutics Market, by Country, 2019-2032 (US$ Mn)
13.5.1. Europe Gaucher Disease Therapeutics Market, by Country, 2023 Vs 2032 (in%)
13.5.2. UK
13.5.3. France
13.5.4. Germany
13.5.5. Italy
13.5.6. Spain
13.5.7. Benelux
13.5.8. Russia
13.5.9. Rest of Europe
14. Asia Pacific Gaucher Disease Therapeutics Market Analysis
14.1. Overview
14.1.1. Market Dynamics for Asia Pacific
14.1.1.1. Drivers
14.1.1.2. Restraints
14.1.1.3. Opportunities
14.1.1.4. Trends
14.2. Asia Pacific Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2032(US$ Mn)
14.2.1. Overview
14.2.2. SRC Analysis
14.3. Asia Pacific Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2032(US$ Mn)
14.3.1. Overview
14.3.2. SRC Analysis
14.4. Asia Pacific Gaucher Disease Therapeutics Market, by End-User, 2019-2032(US$ Mn)
14.4.1. Overview
14.4.2. SRC Analysis
14.5. Asia Pacific Gaucher Disease Therapeutics Market, by Country, 2019-2032 (US$ Mn)
14.5.1. Asia Pacific Gaucher Disease Therapeutics Market, by Country, 2023 Vs 2032 (in%)
14.5.2. China
14.5.3. Japan
14.5.4. India
14.5.5. South Korea
14.5.6. South East Asia
14.5.7. Rest of Asia Pacific
15. Latin America Gaucher Disease Therapeutics Market Analysis
15.1. Overview
15.1.1. Market Dynamics for Latin America
15.1.1.1. Drivers
15.1.1.2. Restraints
15.1.1.3. Opportunities
15.1.1.4. Trends
15.2. Latin America Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2032(US$ Mn)
15.2.1. Overview
15.2.2. SRC Analysis
15.3. Latin America Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2032(US$ Mn)
15.3.1. Overview
15.3.2. SRC Analysis
15.4. Latin America Gaucher Disease Therapeutics Market, by End-User, 2019-2032(US$ Mn)
15.4.1. Overview
15.4.2. SRC Analysis
15.5. Latin America Gaucher Disease Therapeutics Market, by Country, 2019-2032 (US$ Mn)
15.5.1. Latin America Gaucher Disease Therapeutics Market, by Country, 2023 Vs 2032 (in%)
15.5.2. Brazil
15.5.3. Argentina
15.5.4. Rest of Latin America
16. Middle East Gaucher Disease Therapeutics Market Analysis
16.1. Overview
16.1.1. Market Dynamics for Middle East
16.1.1.1. Drivers
16.1.1.2. Restraints
16.1.1.3. Opportunities
16.1.1.4. Trends
16.2. Middle East Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2032(US$ Mn)
16.2.1. Overview
16.2.2. SRC Analysis
16.3. Middle East Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2032(US$ Mn)
16.3.1. Overview
16.3.2. SRC Analysis
16.4. Middle East Gaucher Disease Therapeutics Market, by End-User, 2019-2032(US$ Mn)
16.4.1. Overview
16.4.2. SRC Analysis
16.5. Middle East Gaucher Disease Therapeutics Market, by Country, 2019-2032 (US$ Mn)
16.5.1. Middle East Gaucher Disease Therapeutics Market, by Country, 2023 Vs 2032 (in%)
16.5.2. UAE
16.5.3. Saudi Arabia
16.5.4. Rest of Middle East
17. Africa Gaucher Disease Therapeutics Market Analysis
17.1. Overview
17.1.1. Market Dynamics for Africa
17.1.1.1. Drivers
17.1.1.2. Restraints
17.1.1.3. Opportunities
17.1.1.4. Trends
17.2. Africa Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2032(US$ Mn)
17.2.1. Overview
17.2.2. SRC Analysis
17.3. Africa Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2032(US$ Mn)
17.3.1. Overview
17.3.2. SRC Analysis
17.4. Africa Gaucher Disease Therapeutics Market, by End-User, 2019-2032(US$ Mn)
17.4.1. Overview
17.4.2. SRC Analysis
17.5. Africa Gaucher Disease Therapeutics Market, by Country, 2019-2032 (US$ Mn)
17.5.1. Africa Gaucher Disease Therapeutics Market, by Country, 2023 Vs 2032 (in%)
17.5.2. South Africa
17.5.3. Egypt
17.5.4. Rest of Africa
18. Company Profiles
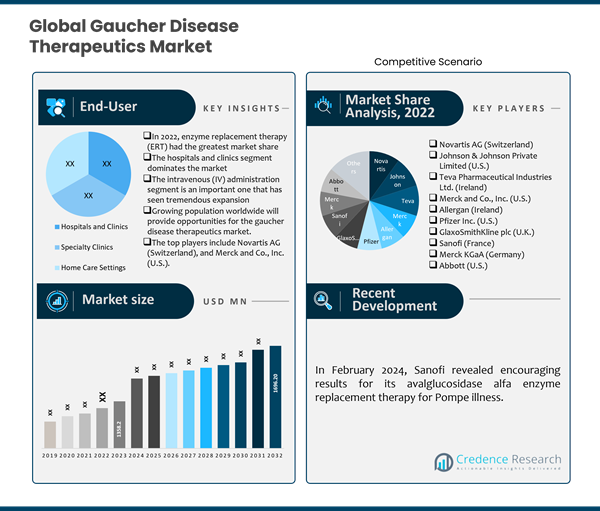
18.1. Novartis AG (Switzerland)
18.1.1. Company Overview
18.1.2. Products/Services Portfolio
18.1.3. Geographical Presence
18.1.4. SWOT Analysis
18.1.5. Financial Summary
18.1.5.1. Market Revenue and Net Profit (2019-2023)
18.1.5.2. Business Segment Revenue Analysis
18.1.5.3. Geographical Revenue Analysis
18.2. Johnson & Johnson Private Limited (U.S.)
18.3. Teva Pharmaceutical Industries Ltd. (Ireland)
18.4. Merck and Co., Inc. (U.S.)
18.5. Allergan (Ireland)
18.6. Pfizer Inc. (U.S.)
18.7. GlaxoSmithKline plc (U.K.)
18.8. Sanofi (France)
18.9. Merck KGaA (Germany)
18.10. Abbott (U.S.)
18.11. Boehringer Ingelheim International GmbH. (Germany)
18.12. Takeda Pharmaceutical Company Limited (Japan)
18.13. Amicus Therapeutics, Inc. (U.S.)
18.14. Moderna, Inc. (U.S.)
18.15. Greenovation Biotech GmbH (Germany)
18.16. Biomarin (U.S.)
18.17. JCR Pharmaceuticals Co., Ltd (Japan)
18.18. ISU ABXIS (South Korea)
18.19. Idorsia Pharmaceuticals Ltd (Switzerland)
18.20. AVROBIO, Inc. (U.S.)
18.21. Resverlogix Corp. (Canada)
18.22. Enzyvant (U.S.)
18.23. CHIESI Farmaceutici SpA (Italy)
18.24. Others
19. Research Methodology
19.1. Research Methodology
19.2. Phase I – Secondary Research
19.3. Phase II – Data Modelling
19.3.1. Company Share Analysis Model
19.3.2. Revenue Based Modelling
19.4. Phase III – Primary Research
19.5. Research Limitations
19.5.1. Assumptions
List of Figures
FIG. 1 Global Gaucher Disease Therapeutics Market: Research Methodology
FIG. 2 Market Size Estimation – Top Down & Bottom up Approach
FIG. 3 Global Gaucher Disease Therapeutics Market Segmentation
FIG. 4 Global Gaucher Disease Therapeutics Market, by Therapy Type, 2023 (US$ Mn)
FIG. 5 Global Gaucher Disease Therapeutics Market, by Route of Administration, 2023 (US$ Mn)
FIG. 6 Global Gaucher Disease Therapeutics Market, by End-User, 2023 (US$ Mn)
FIG. 7 Global Gaucher Disease Therapeutics Market, by Geography, 2023 (US$ Mn)
FIG. 8 Attractive Investment Proposition, by Therapy Type, 2023
FIG. 9 Attractive Investment Proposition, by Route of Administration, 2023
FIG. 10 Attractive Investment Proposition, by End-User, 2023
FIG. 11 Attractive Investment Proposition, by Geography, 2023
FIG. 12 Global Market Share Analysis of Key Gaucher Disease Therapeutics Market Manufacturers, 2023
FIG. 13 Global Market Positioning of Key Gaucher Disease Therapeutics Market Manufacturers, 2023
FIG. 14 Global Gaucher Disease Therapeutics Market Value Contribution, By Therapy Type, 2023 & 2032 (Value %)
FIG. 15 Global Gaucher Disease Therapeutics Market, by Enzyme Replacement Therapy (ERT), Value, 2019-2032 (US$ Mn)
FIG. 16 Global Gaucher Disease Therapeutics Market, by Substrate Reduction Therapy (SRT), Value, 2019-2032 (US$ Mn)
FIG. 17 Global Gaucher Disease Therapeutics Market, by Pharmacological Chaperones, Value, 2019-2032 (US$ Mn)
FIG. 18 Global Gaucher Disease Therapeutics Market, by Substrate Inhibitors, Value, 2019-2032 (US$ Mn)
FIG. 19 Global Gaucher Disease Therapeutics Market, by Gene Therapy, Value, 2019-2032 (US$ Mn)
FIG. 20 Global Gaucher Disease Therapeutics Market, by Supportive Therapies, Value, 2019-2032 (US$ Mn)
FIG. 21 Global Gaucher Disease Therapeutics Market Value Contribution, By Route of Administration, 2023 & 2032 (Value %)
FIG. 22 Global Gaucher Disease Therapeutics Market, by Intravenous (IV) Administration, Value, 2019-2032 (US$ Mn)
FIG. 23 Global Gaucher Disease Therapeutics Market, by Oral Administration, Value, 2019-2032 (US$ Mn)
FIG. 24 Global Gaucher Disease Therapeutics Market Value Contribution, By End-User, 2023 & 2032 (Value %)
FIG. 25 Global Gaucher Disease Therapeutics Market, by Hospitals and Clinics, Value, 2019-2032 (US$ Mn)
FIG. 26 Global Gaucher Disease Therapeutics Market, by Specialty Clinics, Value, 2019-2032 (US$ Mn)
FIG. 27 Global Gaucher Disease Therapeutics Market, by Home Care Settings, Value, 2019-2032 (US$ Mn)
FIG. 28 North America Gaucher Disease Therapeutics Market, 2019-2032 (US$ Mn)
FIG. 29 U.S. Gaucher Disease Therapeutics Market, 2019-2032 (US$ Mn)
FIG. 30 Canada Gaucher Disease Therapeutics Market, 2019-2032 (US$ Mn)
FIG. 31 Mexico Gaucher Disease Therapeutics Market, 2019-2032 (US$ Mn)
FIG. 32 Europe Gaucher Disease Therapeutics Market, 2019-2032 (US$ Mn)
FIG. 33 Germany Gaucher Disease Therapeutics Market, 2019-2032 (US$ Mn)
FIG. 34 France Gaucher Disease Therapeutics Market, 2019-2032 (US$ Mn)
FIG. 35 U.K. Gaucher Disease Therapeutics Market, 2019-2032 (US$ Mn)
FIG. 36 Italy Gaucher Disease Therapeutics Market, 2019-2032 (US$ Mn)
FIG. 37 Spain Gaucher Disease Therapeutics Market, 2019-2032 (US$ Mn)
FIG. 38 Benelux Gaucher Disease Therapeutics Market, 2019-2032 (US$ Mn)
FIG. 39 Russia Gaucher Disease Therapeutics Market, 2019-2032 (US$ Mn)
FIG. 40 Rest of Europe Gaucher Disease Therapeutics Market, 2019-2032 (US$ Mn)
FIG. 41 Asia Pacific Gaucher Disease Therapeutics Market, 2019-2032 (US$ Mn)
FIG. 42 China Gaucher Disease Therapeutics Market, 2019-2032 (US$ Mn)
FIG. 43 Japan Gaucher Disease Therapeutics Market, 2019-2032 (US$ Mn)
FIG. 44 India Gaucher Disease Therapeutics Market, 2019-2032 (US$ Mn)
FIG. 45 South Korea Gaucher Disease Therapeutics Market, 2019-2032 (US$ Mn)
FIG. 46 South-East Asia Gaucher Disease Therapeutics Market, 2019-2032 (US$ Mn)
FIG. 47 Rest of Asia Pacific Gaucher Disease Therapeutics Market, 2019-2032 (US$ Mn)
FIG. 48 Latin America Gaucher Disease Therapeutics Market, 2019-2032 (US$ Mn)
FIG. 49 Brazil Gaucher Disease Therapeutics Market, 2019-2032 (US$ Mn)
FIG. 50 Argentina Gaucher Disease Therapeutics Market, 2019-2032 (US$ Mn)
FIG. 51 Rest of Latin America Gaucher Disease Therapeutics Market, 2019-2032 (US$ Mn)
FIG. 52 Middle East Gaucher Disease Therapeutics Market, 2019-2032 (US$ Mn)
FIG. 53 UAE Gaucher Disease Therapeutics Market, 2019-2032 (US$ Mn)
FIG. 54 Saudi Arabia Gaucher Disease Therapeutics Market, 2019-2032 (US$ Mn)
FIG. 55 Rest of Middle East Gaucher Disease Therapeutics Market, 2019-2032 (US$ Mn)
FIG. 56 Africa Gaucher Disease Therapeutics Market, 2019-2032 (US$ Mn)
FIG. 57 South Africa Gaucher Disease Therapeutics Market, 2019-2032 (US$ Mn)
FIG. 58 Egypt Gaucher Disease Therapeutics Market, 2019-2032 (US$ Mn)
FIG. 59 Rest of Africa Gaucher Disease Therapeutics Market, 2019-2032 (US$ Mn)
List of Tables
TABLE 1 Market Snapshot: Global Gaucher Disease Therapeutics Market
TABLE 2 Global Gaucher Disease Therapeutics Market: Market Drivers Impact Analysis
TABLE 3 Global Gaucher Disease Therapeutics Market: Market Restraints Impact Analysis
TABLE 4 Global Gaucher Disease Therapeutics Market, by Competitive Benchmarking, 2023
TABLE 5 Global Gaucher Disease Therapeutics Market, by Geographical Presence Analysis, 2023
TABLE 6 Global Gaucher Disease Therapeutics Market, by Key Strategies Analysis, 2023
TABLE 7 Global Gaucher Disease Therapeutics Market, by Enzyme Replacement Therapy (ERT), By Region, 2019-2023 (US$ Mn)
TABLE 8 Global Gaucher Disease Therapeutics Market, by Enzyme Replacement Therapy (ERT), By Region, 2024-2032 (US$ Mn)
TABLE 9 Global Gaucher Disease Therapeutics Market, by Substrate Reduction Therapy (SRT), By Region, 2019-2023 (US$ Mn)
TABLE 10 Global Gaucher Disease Therapeutics Market, by Substrate Reduction Therapy (SRT), By Region, 2024-2032 (US$ Mn)
TABLE 11 Global Gaucher Disease Therapeutics Market, by Pharmacological Chaperones, By Region, 2019-2023 (US$ Mn)
TABLE 12 Global Gaucher Disease Therapeutics Market, by Pharmacological Chaperones, By Region, 2024-2032 (US$ Mn)
TABLE 13 Global Gaucher Disease Therapeutics Market, by Substrate Inhibitors, By Region, 2019-2023 (US$ Mn)
TABLE 14 Global Gaucher Disease Therapeutics Market, by Substrate Inhibitors, By Region, 2024-2032 (US$ Mn)
TABLE 15 Global Gaucher Disease Therapeutics Market, by Gene Therapy, By Region, 2019-2023 (US$ Mn)
TABLE 16 Global Gaucher Disease Therapeutics Market, by Gene Therapy, By Region, 2024-2032 (US$ Mn)
TABLE 17 Global Gaucher Disease Therapeutics Market, by Supportive Therapies, By Region, 2019-2023 (US$ Mn)
TABLE 18 Global Gaucher Disease Therapeutics Market, by Supportive Therapies, By Region, 2024-2032 (US$ Mn)
TABLE 19 Global Gaucher Disease Therapeutics Market, by Intravenous (IV) Administration, By Region, 2019-2023 (US$ Mn)
TABLE 20 Global Gaucher Disease Therapeutics Market, by Intravenous (IV) Administration, By Region, 2024-2032 (US$ Mn)
TABLE 21 Global Gaucher Disease Therapeutics Market, by Oral Administration, By Region, 2019-2023 (US$ Mn)
TABLE 22 Global Gaucher Disease Therapeutics Market, by Oral Administration, By Region, 2024-2032 (US$ Mn)
TABLE 23 Global Gaucher Disease Therapeutics Market, by Hospitals and Clinics, By Region, 2019-2023 (US$ Mn)
TABLE 24 Global Gaucher Disease Therapeutics Market, by Hospitals and Clinics, By Region, 2024-2032 (US$ Mn)
TABLE 25 Global Gaucher Disease Therapeutics Market, by Specialty Clinics, By Region, 2019-2023 (US$ Mn)
TABLE 26 Global Gaucher Disease Therapeutics Market, by Specialty Clinics, By Region, 2024-2032 (US$ Mn)
TABLE 27 Global Gaucher Disease Therapeutics Market, by Home Care Settings, By Region, 2019-2023 (US$ Mn)
TABLE 28 Global Gaucher Disease Therapeutics Market, by Home Care Settings, By Region, 2024-2032 (US$ Mn)
TABLE 29 Global Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2023 (US$ Mn)
TABLE 30 Global Gaucher Disease Therapeutics Market, by Therapy Type, 2024-2032 (US$ Mn)
TABLE 31 Global Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2023 (US$ Mn)
TABLE 32 Global Gaucher Disease Therapeutics Market, by Route of Administration, 2024-2032 (US$ Mn)
TABLE 33 Global Gaucher Disease Therapeutics Market, by End-User, 2019-2023 (US$ Mn)
TABLE 34 Global Gaucher Disease Therapeutics Market, by End-User, 2024-2032 (US$ Mn)
TABLE 35 Global Gaucher Disease Therapeutics Market, by Region, 2019-2023 (US$ Mn)
TABLE 36 Global Gaucher Disease Therapeutics Market, by Region, 2024-2032 (US$ Mn)
TABLE 37 North America Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2023 (US$ Mn)
TABLE 38 North America Gaucher Disease Therapeutics Market, by Therapy Type, 2024-2032 (US$ Mn)
TABLE 39 North America Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2023 (US$ Mn)
TABLE 40 North America Gaucher Disease Therapeutics Market, by Route of Administration, 2024-2032 (US$ Mn)
TABLE 41 North America Gaucher Disease Therapeutics Market, by End-User, 2019-2023 (US$ Mn)
TABLE 42 North America Gaucher Disease Therapeutics Market, by End-User, 2024-2032 (US$ Mn)
TABLE 43 North America Gaucher Disease Therapeutics Market, by Country, 2019-2023 (US$ Mn)
TABLE 44 North America Gaucher Disease Therapeutics Market, by Country, 2024-2032 (US$ Mn)
TABLE 45 United States Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2023 (US$ Mn)
TABLE 46 United States Gaucher Disease Therapeutics Market, by Therapy Type, 2024-2032 (US$ Mn)
TABLE 47 United States Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2023 (US$ Mn)
TABLE 48 United States Gaucher Disease Therapeutics Market, by Route of Administration, 2024-2032 (US$ Mn)
TABLE 49 United States Gaucher Disease Therapeutics Market, by End-User, 2019-2023 (US$ Mn)
TABLE 50 United States Gaucher Disease Therapeutics Market, by End-User, 2024-2032 (US$ Mn)
TABLE 51 Canada Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2023 (US$ Mn)
TABLE 52 Canada Gaucher Disease Therapeutics Market, by Therapy Type, 2024-2032 (US$ Mn)
TABLE 53 Canada Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2023 (US$ Mn)
TABLE 54 Canada Gaucher Disease Therapeutics Market, by Route of Administration, 2024-2032 (US$ Mn)
TABLE 55 Canada Gaucher Disease Therapeutics Market, by End-User, 2019-2023 (US$ Mn)
TABLE 56 Canada Gaucher Disease Therapeutics Market, by End-User, 2024-2032 (US$ Mn)
TABLE 57 Mexico Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2023 (US$ Mn)
TABLE 58 Mexico Gaucher Disease Therapeutics Market, by Therapy Type, 2024-2032 (US$ Mn)
TABLE 59 Mexico Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2023 (US$ Mn)
TABLE 60 Mexico Gaucher Disease Therapeutics Market, by Route of Administration, 2024-2032 (US$ Mn)
TABLE 61 Mexico Gaucher Disease Therapeutics Market, by End-User, 2019-2023 (US$ Mn)
TABLE 62 Mexico Gaucher Disease Therapeutics Market, by End-User, 2024-2032 (US$ Mn)
TABLE 63 Europe Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2023 (US$ Mn)
TABLE 64 Europe Gaucher Disease Therapeutics Market, by Therapy Type, 2024-2032 (US$ Mn)
TABLE 65 Europe Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2023 (US$ Mn)
TABLE 66 Europe Gaucher Disease Therapeutics Market, by Route of Administration, 2024-2032 (US$ Mn)
TABLE 67 Europe Gaucher Disease Therapeutics Market, by End-User, 2019-2023 (US$ Mn)
TABLE 68 Europe Gaucher Disease Therapeutics Market, by End-User, 2024-2032 (US$ Mn)
TABLE 69 Europe Gaucher Disease Therapeutics Market, by Country, 2019-2023 (US$ Mn)
TABLE 70 Europe Gaucher Disease Therapeutics Market, by Country, 2024-2032 (US$ Mn)
TABLE 71 Germany Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2023 (US$ Mn)
TABLE 72 Germany Gaucher Disease Therapeutics Market, by Therapy Type, 2024-2032 (US$ Mn)
TABLE 73 Germany Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2023 (US$ Mn)
TABLE 74 Germany Gaucher Disease Therapeutics Market, by Route of Administration, 2024-2032 (US$ Mn)
TABLE 75 Germany Gaucher Disease Therapeutics Market, by End-User, 2019-2023 (US$ Mn)
TABLE 76 Germany Gaucher Disease Therapeutics Market, by End-User, 2024-2032 (US$ Mn)
TABLE 77 France Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2023 (US$ Mn)
TABLE 78 France Gaucher Disease Therapeutics Market, by Therapy Type, 2024-2032 (US$ Mn)
TABLE 79 France Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2023 (US$ Mn)
TABLE 80 France Gaucher Disease Therapeutics Market, by Route of Administration, 2024-2032 (US$ Mn)
TABLE 81 France Gaucher Disease Therapeutics Market, by End-User, 2019-2023 (US$ Mn)
TABLE 82 France Gaucher Disease Therapeutics Market, by End-User, 2024-2032 (US$ Mn)
TABLE 83 United Kingdom Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2023 (US$ Mn)
TABLE 84 United Kingdom Gaucher Disease Therapeutics Market, by Therapy Type, 2024-2032 (US$ Mn)
TABLE 85 United Kingdom Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2023 (US$ Mn)
TABLE 86 United Kingdom Gaucher Disease Therapeutics Market, by Route of Administration, 2024-2032 (US$ Mn)
TABLE 87 United Kingdom Gaucher Disease Therapeutics Market, by End-User, 2019-2023 (US$ Mn)
TABLE 88 United Kingdom Gaucher Disease Therapeutics Market, by End-User, 2024-2032 (US$ Mn)
TABLE 89 Italy Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2023 (US$ Mn)
TABLE 90 Italy Gaucher Disease Therapeutics Market, by Therapy Type, 2024-2032 (US$ Mn)
TABLE 91 Italy Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2023 (US$ Mn)
TABLE 92 Italy Gaucher Disease Therapeutics Market, by Route of Administration, 2024-2032 (US$ Mn)
TABLE 93 Italy Gaucher Disease Therapeutics Market, by End-User, 2019-2023 (US$ Mn)
TABLE 94 Italy Gaucher Disease Therapeutics Market, by End-User, 2024-2032 (US$ Mn)
TABLE 95 Spain Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2023 (US$ Mn)
TABLE 96 Spain Gaucher Disease Therapeutics Market, by Therapy Type, 2024-2032 (US$ Mn)
TABLE 97 Spain Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2023 (US$ Mn)
TABLE 98 Spain Gaucher Disease Therapeutics Market, by Route of Administration, 2024-2032 (US$ Mn)
TABLE 99 Spain Gaucher Disease Therapeutics Market, by End-User, 2019-2023 (US$ Mn)
TABLE 100 Spain Gaucher Disease Therapeutics Market, by End-User, 2024-2032 (US$ Mn)
TABLE 101 Benelux Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2023 (US$ Mn)
TABLE 102 Benelux Gaucher Disease Therapeutics Market, by Therapy Type, 2024-2032 (US$ Mn)
TABLE 103 Benelux Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2023 (US$ Mn)
TABLE 104 Benelux Gaucher Disease Therapeutics Market, by Route of Administration, 2024-2032 (US$ Mn)
TABLE 105 Benelux Gaucher Disease Therapeutics Market, by End-User, 2019-2023 (US$ Mn)
TABLE 106 Benelux Gaucher Disease Therapeutics Market, by End-User, 2024-2032 (US$ Mn)
TABLE 107 Russia Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2023 (US$ Mn)
TABLE 108 Russia Gaucher Disease Therapeutics Market, by Therapy Type, 2024-2032 (US$ Mn)
TABLE 109 Russia Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2023 (US$ Mn)
TABLE 110 Russia Gaucher Disease Therapeutics Market, by Route of Administration, 2024-2032 (US$ Mn)
TABLE 111 Russia Gaucher Disease Therapeutics Market, by End-User, 2019-2023 (US$ Mn)
TABLE 112 Russia Gaucher Disease Therapeutics Market, by End-User, 2024-2032 (US$ Mn)
TABLE 113 Rest of Europe Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2023 (US$ Mn)
TABLE 114 Rest of Europe Gaucher Disease Therapeutics Market, by Therapy Type, 2024-2032 (US$ Mn)
TABLE 115 Rest of Europe Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2023 (US$ Mn)
TABLE 116 Rest of Europe Gaucher Disease Therapeutics Market, by Route of Administration, 2024-2032 (US$ Mn)
TABLE 117 Rest of Europe Gaucher Disease Therapeutics Market, by End-User, 2019-2023 (US$ Mn)
TABLE 118 Rest of Europe Gaucher Disease Therapeutics Market, by End-User, 2024-2032 (US$ Mn)
TABLE 119 Asia Pacific Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2023 (US$ Mn)
TABLE 120 Asia Pacific Gaucher Disease Therapeutics Market, by Therapy Type, 2024-2032 (US$ Mn)
TABLE 121 Asia Pacific Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2023 (US$ Mn)
TABLE 122 Asia Pacific Gaucher Disease Therapeutics Market, by Route of Administration, 2024-2032 (US$ Mn)
TABLE 123 Asia Pacific Gaucher Disease Therapeutics Market, by End-User, 2019-2023 (US$ Mn)
TABLE 124 Asia Pacific Gaucher Disease Therapeutics Market, by End-User, 2024-2032 (US$ Mn)
TABLE 125 China Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2023 (US$ Mn)
TABLE 126 China Gaucher Disease Therapeutics Market, by Therapy Type, 2024-2032 (US$ Mn)
TABLE 127 China Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2023 (US$ Mn)
TABLE 128 China Gaucher Disease Therapeutics Market, by Route of Administration, 2024-2032 (US$ Mn)
TABLE 129 China Gaucher Disease Therapeutics Market, by End-User, 2019-2023 (US$ Mn)
TABLE 130 China Gaucher Disease Therapeutics Market, by End-User, 2024-2032 (US$ Mn)
TABLE 131 Japan Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2023 (US$ Mn)
TABLE 132 Japan Gaucher Disease Therapeutics Market, by Therapy Type, 2024-2032 (US$ Mn)
TABLE 133 Japan Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2023 (US$ Mn)
TABLE 134 Japan Gaucher Disease Therapeutics Market, by Route of Administration, 2024-2032 (US$ Mn)
TABLE 135 Japan Gaucher Disease Therapeutics Market, by End-User, 2019-2023 (US$ Mn)
TABLE 136 Japan Gaucher Disease Therapeutics Market, by End-User, 2024-2032 (US$ Mn)
TABLE 137 India Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2023 (US$ Mn)
TABLE 138 India Gaucher Disease Therapeutics Market, by Therapy Type, 2024-2032 (US$ Mn)
TABLE 139 India Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2023 (US$ Mn)
TABLE 140 India Gaucher Disease Therapeutics Market, by Route of Administration, 2024-2032 (US$ Mn)
TABLE 141 India Gaucher Disease Therapeutics Market, by End-User, 2019-2023 (US$ Mn)
TABLE 142 India Gaucher Disease Therapeutics Market, by End-User, 2024-2032 (US$ Mn)
TABLE 143 South Korea Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2023 (US$ Mn)
TABLE 144 South Korea Gaucher Disease Therapeutics Market, by Therapy Type, 2024-2032 (US$ Mn)
TABLE 145 South Korea Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2023 (US$ Mn)
TABLE 146 South Korea Gaucher Disease Therapeutics Market, by Route of Administration, 2024-2032 (US$ Mn)
TABLE 147 South Korea Gaucher Disease Therapeutics Market, by End-User, 2019-2023 (US$ Mn)
TABLE 148 South Korea Gaucher Disease Therapeutics Market, by End-User, 2024-2032 (US$ Mn)
TABLE 149 South-East Asia Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2023 (US$ Mn)
TABLE 150 South-East Asia Gaucher Disease Therapeutics Market, by Therapy Type, 2024-2032 (US$ Mn)
TABLE 151 South-East Asia Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2023 (US$ Mn)
TABLE 152 South-East Asia Gaucher Disease Therapeutics Market, by Route of Administration, 2024-2032 (US$ Mn)
TABLE 153 South-East Asia Gaucher Disease Therapeutics Market, by End-User, 2019-2023 (US$ Mn)
TABLE 154 South-East Asia Gaucher Disease Therapeutics Market, by End-User, 2024-2032 (US$ Mn)
TABLE 155 Rest of Asia Pacific Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2023 (US$ Mn)
TABLE 156 Rest of Asia Pacific Gaucher Disease Therapeutics Market, by Therapy Type, 2024-2032 (US$ Mn)
TABLE 157 Rest of Asia Pacific Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2023 (US$ Mn)
TABLE 158 Rest of Asia Pacific Gaucher Disease Therapeutics Market, by Route of Administration, 2024-2032 (US$ Mn)
TABLE 159 Rest of Asia Pacific Gaucher Disease Therapeutics Market, by End-User, 2019-2023 (US$ Mn)
TABLE 160 Rest of Asia Pacific Gaucher Disease Therapeutics Market, by End-User, 2024-2032 (US$ Mn)
TABLE 161 Latin America Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2023 (US$ Mn)
TABLE 162 Latin America Gaucher Disease Therapeutics Market, by Therapy Type, 2024-2032 (US$ Mn)
TABLE 163 Latin America Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2023 (US$ Mn)
TABLE 164 Latin America Gaucher Disease Therapeutics Market, by Route of Administration, 2024-2032 (US$ Mn)
TABLE 165 Latin America Gaucher Disease Therapeutics Market, by End-User, 2019-2023 (US$ Mn)
TABLE 166 Latin America Gaucher Disease Therapeutics Market, by End-User, 2024-2032 (US$ Mn)
TABLE 167 Brazil Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2023 (US$ Mn)
TABLE 168 Brazil Gaucher Disease Therapeutics Market, by Therapy Type, 2024-2032 (US$ Mn)
TABLE 169 Brazil Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2023 (US$ Mn)
TABLE 170 Brazil Gaucher Disease Therapeutics Market, by Route of Administration, 2024-2032 (US$ Mn)
TABLE 171 Brazil Gaucher Disease Therapeutics Market, by End-User, 2019-2023 (US$ Mn)
TABLE 172 Brazil Gaucher Disease Therapeutics Market, by End-User, 2024-2032 (US$ Mn)
TABLE 173 Argentina Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2023 (US$ Mn)
TABLE 174 Argentina Gaucher Disease Therapeutics Market, by Therapy Type, 2024-2032 (US$ Mn)
TABLE 175 Argentina Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2023 (US$ Mn)
TABLE 176 Argentina Gaucher Disease Therapeutics Market, by Route of Administration, 2024-2032 (US$ Mn)
TABLE 177 Argentina Gaucher Disease Therapeutics Market, by End-User, 2019-2023 (US$ Mn)
TABLE 178 Argentina Gaucher Disease Therapeutics Market, by End-User, 2024-2032 (US$ Mn)
TABLE 179 Rest of Latin America Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2023 (US$ Mn)
TABLE 180 Rest of Latin America Gaucher Disease Therapeutics Market, by Therapy Type, 2024-2032 (US$ Mn)
TABLE 181 Rest of Latin America Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2023 (US$ Mn)
TABLE 182 Rest of Latin America Gaucher Disease Therapeutics Market, by Route of Administration, 2024-2032 (US$ Mn)
TABLE 183 Rest of Latin America Gaucher Disease Therapeutics Market, by End-User, 2019-2023 (US$ Mn)
TABLE 184 Rest of Latin America Gaucher Disease Therapeutics Market, by End-User, 2024-2032 (US$ Mn)
TABLE 185 Middle East Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2023 (US$ Mn)
TABLE 186 Middle East Gaucher Disease Therapeutics Market, by Therapy Type, 2024-2032 (US$ Mn)
TABLE 187 Middle East Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2023 (US$ Mn)
TABLE 188 Middle East Gaucher Disease Therapeutics Market, by Route of Administration, 2024-2032 (US$ Mn)
TABLE 189 Middle East Gaucher Disease Therapeutics Market, by End-User, 2019-2023 (US$ Mn)
TABLE 190 Middle East Gaucher Disease Therapeutics Market, by End-User, 2024-2032 (US$ Mn)
TABLE 191 UAE Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2023 (US$ Mn)
TABLE 192 UAE Gaucher Disease Therapeutics Market, by Therapy Type, 2024-2032 (US$ Mn)
TABLE 193 UAE Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2023 (US$ Mn)
TABLE 194 UAE Gaucher Disease Therapeutics Market, by Route of Administration, 2024-2032 (US$ Mn)
TABLE 195 UAE Gaucher Disease Therapeutics Market, by End-User, 2019-2023 (US$ Mn)
TABLE 196 UAE Gaucher Disease Therapeutics Market, by End-User, 2024-2032 (US$ Mn)
TABLE 197 Saudi Arabia Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2023 (US$ Mn)
TABLE 198 Saudi Arabia Gaucher Disease Therapeutics Market, by Therapy Type, 2024-2032 (US$ Mn)
TABLE 199 Saudi Arabia Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2023 (US$ Mn)
TABLE 200 Saudi Arabia Gaucher Disease Therapeutics Market, by Route of Administration, 2024-2032 (US$ Mn)
TABLE 201 Saudi Arabia Gaucher Disease Therapeutics Market, by End-User, 2019-2023 (US$ Mn)
TABLE 202 Saudi Arabia Gaucher Disease Therapeutics Market, by End-User, 2024-2032 (US$ Mn)
TABLE 203 Rest of Middle East Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2023 (US$ Mn)
TABLE 204 Rest of Middle East Gaucher Disease Therapeutics Market, by Therapy Type, 2024-2032 (US$ Mn)
TABLE 205 Rest of Middle East Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2023 (US$ Mn)
TABLE 206 Rest of Middle East Gaucher Disease Therapeutics Market, by Route of Administration, 2024-2032 (US$ Mn)
TABLE 207 Rest of Middle East Gaucher Disease Therapeutics Market, by End-User, 2019-2023 (US$ Mn)
TABLE 208 Rest of Middle East Gaucher Disease Therapeutics Market, by End-User, 2024-2032 (US$ Mn)
TABLE 209 Africa Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2023 (US$ Mn)
TABLE 210 Africa Gaucher Disease Therapeutics Market, by Therapy Type, 2024-2032 (US$ Mn)
TABLE 211 Africa Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2023 (US$ Mn)
TABLE 212 Africa Gaucher Disease Therapeutics Market, by Route of Administration, 2024-2032 (US$ Mn)
TABLE 213 Africa Gaucher Disease Therapeutics Market, by End-User, 2019-2023 (US$ Mn)
TABLE 214 Africa Gaucher Disease Therapeutics Market, by End-User, 2024-2032 (US$ Mn)
TABLE 215 South Africa Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2023 (US$ Mn)
TABLE 216 South Africa Gaucher Disease Therapeutics Market, by Therapy Type, 2024-2032 (US$ Mn)
TABLE 217 South Africa Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2023 (US$ Mn)
TABLE 218 South Africa Gaucher Disease Therapeutics Market, by Route of Administration, 2024-2032 (US$ Mn)
TABLE 219 South Africa Gaucher Disease Therapeutics Market, by End-User, 2019-2023 (US$ Mn)
TABLE 220 South Africa Gaucher Disease Therapeutics Market, by End-User, 2024-2032 (US$ Mn)
TABLE 221 Egypt Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2023 (US$ Mn)
TABLE 222 Egypt Gaucher Disease Therapeutics Market, by Therapy Type, 2024-2032 (US$ Mn)
TABLE 223 Egypt Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2023 (US$ Mn)
TABLE 224 Egypt Gaucher Disease Therapeutics Market, by Route of Administration, 2024-2032 (US$ Mn)
TABLE 225 Egypt Gaucher Disease Therapeutics Market, by End-User, 2019-2023 (US$ Mn)
TABLE 226 Egypt Gaucher Disease Therapeutics Market, by End-User, 2024-2032 (US$ Mn)
TABLE 227 Rest of Africa Gaucher Disease Therapeutics Market, by Therapy Type, 2019-2023 (US$ Mn)
TABLE 228 Rest of Africa Gaucher Disease Therapeutics Market, by Therapy Type, 2024-2032 (US$ Mn)
TABLE 229 Rest of Africa Gaucher Disease Therapeutics Market, by Route of Administration, 2019-2023 (US$ Mn)
TABLE 230 Rest of Africa Gaucher Disease Therapeutics Market, by Route of Administration, 2024-2032 (US$ Mn)
TABLE 231 Rest of Africa Gaucher Disease Therapeutics Market, by End-User, 2019-2023 (US$ Mn)
TABLE 232 Rest of Africa Gaucher Disease Therapeutics Market, by End-User, 2024-2032 (US$ Mn)