ATP Assays Market Overview:
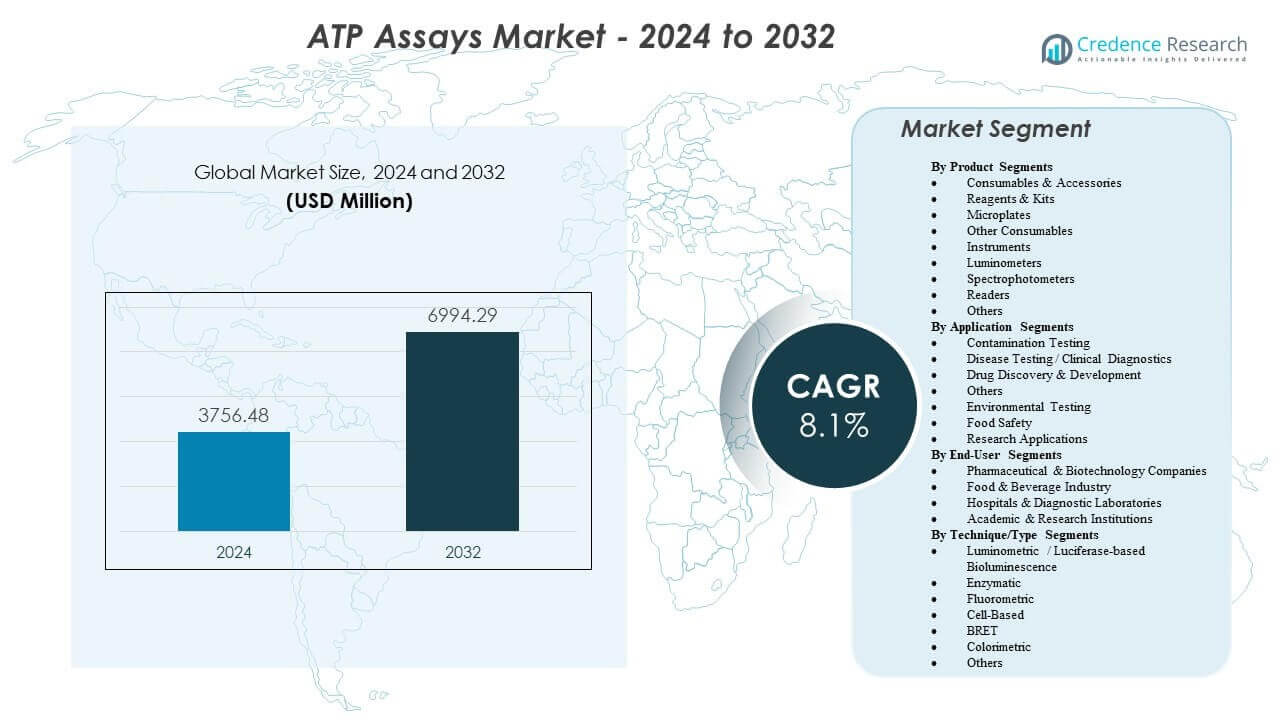
The ATP Assays Market is projected to grow from USD 3756.48 million in 2024 to an estimated USD 6994.29 million by 2032, with a CAGR of 8.1% from 2024 to 2032.
| REPORT ATTRIBUTE |
DETAILS |
| Historical Period |
2020-2023 |
| Base Year |
2024 |
| Forecast Period |
2025-2032 |
| ATP Assays Market Size 2024 |
USD 3756.48 Million |
| ATP Assays Market, CAGR |
8.1% |
| ATP Assays Market Size 2032 |
USD 6994.29 Million |
ATP Assays Market Insights:
- Strong demand for high-sensitivity viability tools and wider use in drug discovery, oncology, and toxicology drive sustained market growth.
- Limited assay stability in complex samples and high costs of advanced detection instruments create adoption barriers in smaller laboratories.
- North America leads the market due to strong biopharma research activity, while Europe follows with solid institutional and clinical demand.
- Asia Pacific shows the fastest expansion, driven by rising biomedical research capacity, laboratory modernization, and growing industrial hygiene needs.
ATP Assays Market Drivers
Rising Demand for High-Sensitivity Cell Viability Measurement Tools
Strong demand for high-sensitivity viability assessment pushes steady growth in the ATP Assays Market. Drug developers use ATP assays to measure early cellular responses with high reliability. The method offers fast detection, which supports rapid screening workflows. Researchers rely on the platform for metabolic evaluation during preclinical studies. Pharma companies adopt these assays to improve decision timelines. Laboratories benefit from lower error rates due to strong signal stability. It supports precision within low-volume samples. CRO expansion increases purchases across global research hubs.
Access crucial information at unmatched prices!
Request your sample report today & start making informed decisions powered by Credence Research Inc.!
Download Sample
Growing Use of ATP Assays in Oncology and Immunology Research Programs
Oncology and immunology teams use ATP assays to track disease biology in controlled environments. Cancer studies need accurate monitoring of cell proliferation and stress markers. ATP kits support these experiments with consistent output. Immunology models require sensitive detection during activation studies, which drives more adoption. The tool helps teams validate therapeutic behavior earlier. Labs use ATP assays during biomarker exploration. They depend on this framework to optimize assay reproducibility. The ATP Assays Market gains revenue from rising research cycles.
Rapid Expansion of High-Throughput Screening Platforms Across Laboratories
High-throughput systems create strong demand for ATP-compatible formats. Automated microplate readers improve speed for large sample sets. It helps teams standardize screening across discovery units. Pharma firms introduce expanded screening pipelines that rely on ATP-based endpoints. Robotic integration increases assay use across global facilities. CRO laboratories support clients with automated ATP workflows. Screening capacity rises due to strong compatibility with luminescence detection. The ATP Assays Market benefits from scalable adoption across research organizations.
- For instance, the Promega CellTiter-Glo 2.0 Assay is engineered for high-throughput compatibility, demonstrating a Z’-factor consistently above 0.7 when used in automated 1,536-well plate systems, which indicates superior assay robustness for large-scale compound libraries.
Increasing Funding Support for Life Science and Translational Research
Stronger research investment stimulates demand for ATP-based measurement tools. Governments direct funds toward biomedical innovation, which helps laboratories upgrade workflows. Universities build new research units that depend on ATP kits. It assists translational teams during therapeutic evaluation. Research institutions use the method to validate early cell behavior. Funding programs encourage adoption due to assay reliability. Private investors support biotech projects that rely on ATP analysis. The ATP Assays Market expands with broader research accessibility.
- For instance, the National Institutes of Health (NIH) provided funding for the development of high-density metabolic profiling, leading to the use of Agilent Seahorse XF technology which measures ATP production rates in real-time with a sensitivity capable of detecting metabolic shifts in as few as 5,000 cells.
ATP Assays Market Trends
Shift Toward Multiplexed Assay Formats for Broader Biological Insight
Multiplexed systems gain traction because researchers want deeper biological context. Teams collect ATP data alongside cytotoxicity or apoptosis markers. It reduces time spent on multiple workflows. Multiplex kits help labs conserve sample volumes. Pharma units integrate multiplex platforms into screening pipelines. Laboratories gain more insight per experiment with improved efficiency. The ATP Assays Market supports this shift through flexible product designs. Companies upgrade formats to match evolving research demands.
- For instance, Abcam’s Multiplex Cell Viability Assay Kit allows for the simultaneous measurement of ATP levels and LDH release, providing a dual-readout of metabolic activity and membrane integrity with a sensitivity threshold capable of detecting changes in as few as 500 cells.
Growing Integration of ATP Assays in 3D Cell Models and Organoid Studies
Three-dimensional systems gain global acceptance due to stronger biological relevance. Organoids require precise energy-level measurement for viability scoring. Laboratories use ATP assays to track therapeutic impact in advanced models. It supports model validation across cancer, toxicology, and regenerative studies. Research groups prefer ATP workflows due to minimal interference. Demand rises as more projects adopt 3D structures. The ATP Assays Market grows with wider organoid deployment. Vendors enhance compatibility with complex sample architectures.
Increased Adoption of Automation and AI-Enabled Laboratory Workflows
Automation changes assay execution patterns across discovery labs. AI tools assist with interpretation of luminescence data. It enables improved accuracy for high-throughput samples. Automated handlers deliver consistent liquid movement for ATP kits. Research facilities reduce variability through system integration. Pharma groups streamline workflows through intelligent scheduling modules. The ATP Assays Market aligns with these advancements. Vendors design assay formats for robotic compatibility and AI platforms.
- For instance, Agilent Technologies integrated its BioTek BioStack microplate stacker with the Synergy™ Neo2 Multi-Mode Reader, enabling the automated processing of up to 50 microplates per run with a plate exchange time of less than 6 seconds.
Rising Preference for Ready-to-Use and Pre-Validated ATP Assay Kits
Demand for ready-to-use kits rises due to workflow simplification needs. Pre-validated protocols help new users reduce setup time. Laboratories use these kits to maintain reproducible outcomes. It improves training efficiency for new personnel. Ready-use designs lower troubleshooting frequency across facilities. Adoption grows as research groups aim for higher throughput. The ATP Assays Market moves toward standardized kit structures. Suppliers invest in stability upgrades and packaging refinements.
ATP Assays Market Challenges Analysis
Technical Limitations Affecting Data Interpretation and Experimental Consistency
Some ATP assay formats face interference from serum content or reagent instability. This limits consistent readings during complex studies. It introduces challenges for laboratories handling diverse sample types. Researchers must adjust protocols to maintain accurate interpretation. Variation in luminescence output affects repeated testing sets. Multiplex environments also raise risk for cross-signal effects. The ATP Assays Market must manage user concerns about analytical dependability. Vendors strengthen formulations to reduce variability across workflows.
High Instrumentation Costs and Workflow Standardization Barriers
Advanced microplate readers, automation tools, and luminescence systems require sizable investment. High cost restricts adoption for small labs and academic groups. It influences the pace of technology upgrades across regions. Standardization challenges arise from diverse workflows. Laboratories need trained personnel to manage optimized execution. It creates onboarding hurdles for new research sites. The ATP Assays Market faces constraints linked with budget limits. Vendors explore flexible pricing to improve adoption.
ATP Assays Market Opportunities
Expansion of ATP Assays Across Personalized Medicine and Cell-Based Therapeutic Development
Personalized medicine increases demand for accurate cell viability indicators. ATP assays help teams evaluate patient-derived cells with strong precision. It supports drug-response profiling during early development. Research centers use ATP kits to track therapeutic effects in engineered cells. Cell therapy growth creates more need for sensitive viability measurement. The ATP Assays Market gains opportunity from expanded clinical research roles. Vendors introduce specialized formats for near-patient studies. Growth in precision health strengthens adoption potential.
Increasing Use of ATP Assays in Environmental, Food, and Industrial Hygiene Testing
New application areas open demand beyond life sciences. Environmental teams measure microbial loads using ATP detection. Food processors use ATP tools to verify sanitation quality. It enhances monitoring programs across production lines. Industrial hygiene groups adopt ATP workflows for surface assessment. Rising focus on safety compliance pushes more purchase activity. The ATP Assays Market benefits from diverse industry penetration. Vendors develop field-ready kits to support on-site testing requirements.
ATP Assays Market Segmentation Analysis:
By Product Segments
Consumables and accessories hold strong demand due to frequent use in routine workflows across the ATP Assays Market. Reagents and kits lead the segment because they support high-throughput studies with reliable outputs. Microplates maintain steady uptake due to compatibility with automated systems. Other consumables help laboratories manage sample handling needs. Instruments gain traction with rising adoption of advanced detection platforms. Luminometers remain essential for luminescence assays. Spectrophotometers and readers support broader analytical needs. It drives continuous upgrades with expanding research activity.
- For instance, Promega Corporation’s CellTiter-Glo 2.0 assay provides a simplified single-reagent protocol capable of detecting as few as 10 cells per well with a signal half-life of over 5 hours.
By Application Segments
Contamination testing records strong adoption because industries need rapid detection tools to maintain quality. Disease testing and clinical diagnostics use ATP assays to support precise cellular activity assessment within the ATP Assays Market. Drug discovery and development rely on ATP-based viability studies to validate early therapeutic effects. Environmental testing teams use ATP indicators for microbial load checks. Food safety programs adopt ATP systems for hygiene verification. Research applications dominate usage due to wide scientific utility. It supports consistent adoption across diversified workflows. Others cover niche testing needs.
- For instance, Revvity’s ATPLite 1step assay allows researchers to perform high-throughput screening in 1,536-well plate formats, maintaining a linear dynamic range of up to 5 orders of magnitude.
By End-User Segments
Pharmaceutical and biotechnology companies remain primary users due to extensive screening requirements in the ATP Assays Market. Food and beverage companies adopt ATP tools for sanitation verification. Hospitals and diagnostic laboratories use ATP assays for sample quality checks and metabolic evaluation. Academic and research institutions depend on ATP kits for foundational studies across biology fields. It supports high usage due to flexibility across multiple projects.
By Technique/Type Segments
Luminometric bioluminescence techniques lead due to high sensitivity and robust signal output in the ATP Assays Market. Enzymatic assays maintain stable demand for basic ATP quantification. Fluorometric and colorimetric methods provide alternatives where luminescence tools are limited. Cell-based assays support live-cell analysis needs. BRET techniques offer specialized energy transfer applications. Others address emerging detection platforms. It drives innovation across assay formats.
Segmentation:
By Product Segments
- Consumables & Accessories
- Reagents & Kits
- Microplates
- Other Consumables
- Instruments
- Luminometers
- Spectrophotometers
- Readers
- Others
By Application Segments
- Contamination Testing
- Disease Testing / Clinical Diagnostics
- Drug Discovery & Development
- Others
- Environmental Testing
- Food Safety
- Research Applications
By End-User Segments
- Pharmaceutical & Biotechnology Companies
- Food & Beverage Industry
- Hospitals & Diagnostic Laboratories
- Academic & Research Institutions
By Technique/Type Segments
- Luminometric / Luciferase-based Bioluminescence
- Enzymatic
- Fluorometric
- Cell-Based
- BRET
- Colorimetric
- Others
By Region
- North America
- Europe
- Germany
- France
- U.K.
- Italy
- Spain
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- South-east Asia
- Rest of Asia Pacific
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Middle East & Africa
- GCC Countries
- South Africa
- Rest of the Middle East and Africa
Regional Analysis:
North America holds the largest share of the ATP Assays Market, contributing close to 38% due to strong biopharmaceutical research activity and advanced laboratory infrastructure. The region benefits from high adoption of high-throughput screening tools and continuous investments in life science innovation. Research institutions and CROs maintain strong demand for ATP-based viability assays. It gains further support from strict contamination control standards across healthcare and food sectors. Leading companies expand product portfolios to strengthen their presence. Stable regulatory frameworks help laboratories adopt advanced analytical systems.
Europe accounts for nearly 28% of global share with wide assay adoption across biotechnology, diagnostics, and academic sectors. The region supports market growth through sustained R&D funding and strong participation in cell biology and precision medicine projects. Pharmaceutical hubs in Germany, the UK, and France drive extensive assay usage. Environmental and food safety programs rely on ATP systems to support quality monitoring. It benefits from the region’s focus on establishing reliable contamination detection workflows. Expansion of translational research accelerates uptake.
Asia Pacific captures close to 24% share and produces the fastest growth due to rising biomedical research capacity across China, Japan, India, and South Korea. Governments invest in laboratory modernization, which drives assay procurement. Pharmaceutical manufacturing expansion supports higher screening activity. Food and environmental sectors adopt ATP tools for hygiene verification and microbial detection. It gains momentum from increasing CRO operations in the region. Emerging markets in Southeast Asia broaden the user base.
Shape Your Report to Specific Countries or Regions & Enjoy 30% Off!
Key Player Analysis:
- Thermo Fisher Scientific Inc.
- Promega Corporation
- Merck KGaA
- PerkinElmer Inc.
- Danaher Corporation
- Agilent Technologies Inc.
- Lonza Group Ltd.
- Abcam plc
- Becton, Dickinson and Company (BD)
- Neogen Corporation
- bioMérieux SA
- 3M Company
- Quest Diagnostics Incorporated
- Biotium Inc.
- Hygiena LLC
Competitive Analysis:
The ATP Assays Market features strong competition among global assay manufacturers, instrument suppliers, and specialized biotechnology firms. Companies such as Thermo Fisher Scientific, Promega, Merck, and PerkinElmer maintain leadership through broad reagent portfolios and high-sensitivity detection systems. Competitors strengthen market presence with upgraded luminescence technologies and enhanced ready-to-use kits. It sees an increase in collaborations between assay developers and instrument manufacturers to support seamless workflow integration. Mid-tier players such as Biotium, Hygiena, and Neogen focus on contamination testing and rapid hygiene monitoring tools. Strategic moves include product launches, acquisitions, and distribution expansions that help firms reach new research and industrial customers. Growing emphasis on automation and multiplexing supports innovation across competing product lines.
Recent Developments:
- In March 2025, Biotium launched the Steady-ATP HTS Viability Assay Kit. This highly sensitive luminescent assay measures cell viability through ATP quantification in a single-step format, ideal for high-throughput screening with a stable signal lasting over 5 hours.
- In April 2024, Reaction Biology introduced the HotSpot ATP-Max KinomeScreen assay platform at the AACR Annual Meeting. This radiometric assay enables kinase profiling at physiologically relevant 1 mM ATP concentrations across a broad target portfolio, advancing drug discovery.
Report Coverage:
The research report offers an in-depth analysis based on Product Segments, Application Segments, End-User Segments, and Technique/Type Segments. It details leading market players, providing an overview of their business, product offerings, investments, revenue streams, and key applications. Additionally, the report includes insights into the competitive environment, SWOT analysis, current market trends, as well as the primary drivers and constraints. Furthermore, it discusses various factors that have driven market expansion in recent years. The report also explores market dynamics, regulatory scenarios, and technological advancements that are shaping the industry. It assesses the impact of external factors and global economic changes on market growth. Lastly, it provides strategic recommendations for new entrants and established companies to navigate the complexities of the market.
Future Outlook:
- Expansion of high-throughput screening workflows will increase the use of ATP-based viability tools across drug discovery programs.
- Rising adoption of automated platforms will strengthen demand for assays compatible with robotic handling systems.
- Growth in oncology and immunology research will elevate the need for sensitive ATP detection formats.
- Wider acceptance of organoid and 3D cell models will drive interest in ATP assays tailored for complex biology.
- Increased focus on hygiene verification in food and environmental sectors will expand industrial usage.
- Advancements in luminescence technologies will deliver stronger signal stability and faster readout efficiency.
- Broader integration of AI-supported analytics will support accurate interpretation of ATP assay outputs.
- Development of ready-to-use kits will simplify workflows for new laboratories with limited technical expertise.
- Growth in CRO operations will amplify global uptake due to outsourced research demand.
- Rising investments in biomedical research across emerging regions will diversify ATP assay consumption patterns.




