CHAPTER NO. 1 : GENESIS OF THE MARKET
1.1 Market Prelude – Introduction & Scope
1.2 The Big Picture – Objectives & Vision
1.3 Strategic Edge – Unique Value Proposition
1.4 Stakeholder Compass – Key Beneficiaries
CHAPTER NO. 2 : EXECUTIVE LENS
2.1 Pulse of the Industry – Market Snapshot
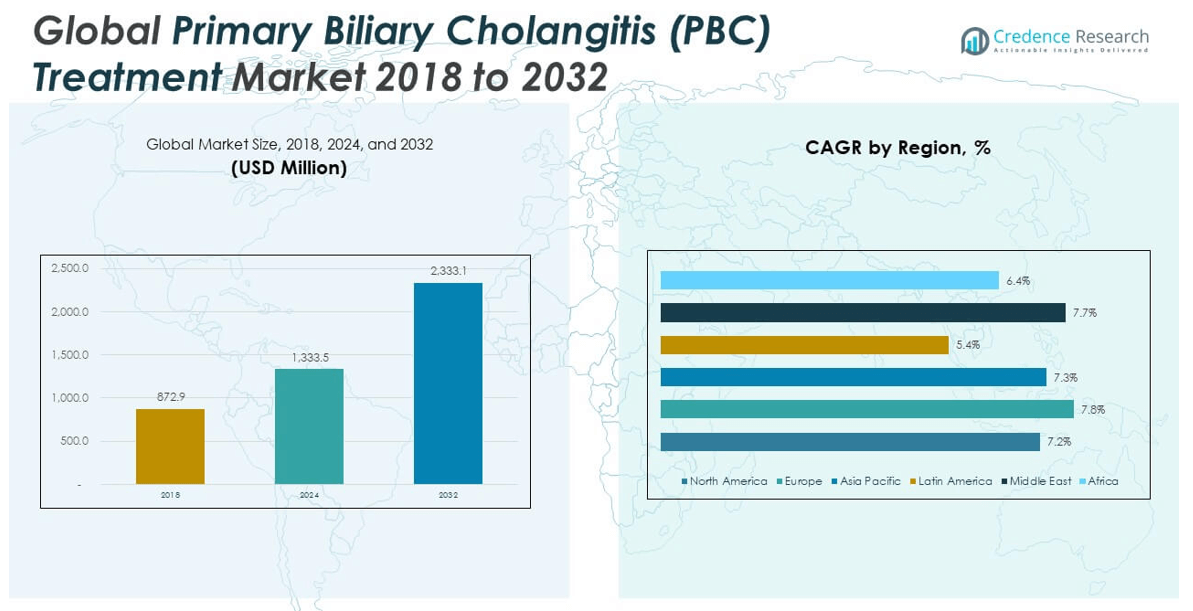
2.2 Growth Arc – Revenue Projections (USD Million)
2.3. Premium Insights – Based on Primary Interviews
CHAPTER NO. 3 : PRIMARY BILIARY CHOLANGITIS (PBC) TREATMENT MARKET FORCES & INDUSTRY PULSE
3.1 Foundations of Change – Market Overview
3.2 Catalysts of Expansion – Key Market Drivers
3.2.1 Momentum Boosters – Growth Triggers
3.2.2 Innovation Fuel – Disruptive Technologies
3.3 Headwinds & Crosswinds – Market Restraints
3.3.1 Regulatory Tides – Compliance Challenges
3.3.2 Economic Frictions – Inflationary Pressures
3.4 Untapped Horizons – Growth Potential & Opportunities
3.5 Strategic Navigation – Industry Frameworks
3.5.1 Market Equilibrium – Porter’s Five Forces
3.5.2 Ecosystem Dynamics – Value Chain Analysis
3.5.3 Macro Forces – PESTEL Breakdow
CHAPTER NO. 4 : KEY INVESTMENT EPICENTER
4.1 Regional Goldmines – High-Growth Geographies
4.2 Product Frontiers – Lucrative Product Categories
4.3 Treatment Sweet Spots – Emerging Demand Segments
CHAPTER NO. 5: REVENUE TRAJECTORY & WEALTH MAPPING
5.1 Momentum Metrics – Forecast & Growth Curves
5.2 Regional Revenue Footprint – Market Share Insights
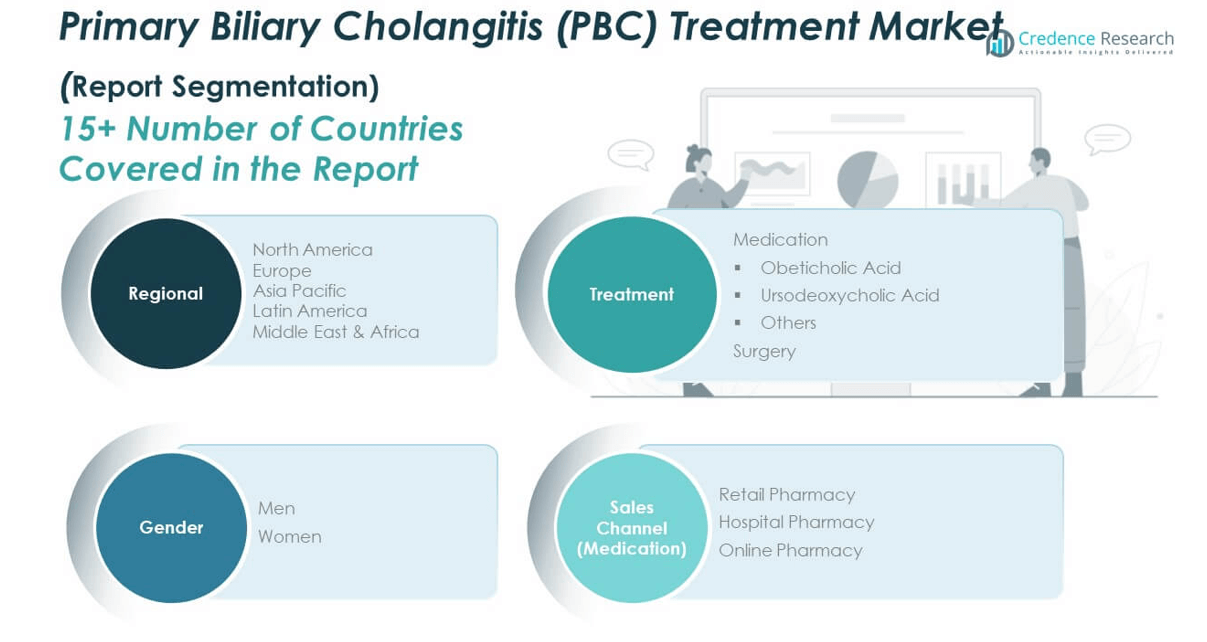
5.3 Segmental Wealth Flow – Treatment, Gender, and Sales Channel (Treatment) Revenue
CHAPTER NO. 6 : TRADE & COMMERCE ANALYSIS
6.1. Import Analysis By Region
6.1.1. Global Primary Biliary Cholangitis (PBC) Treatment Market Import Revenue By Region
6.2. Export Analysis By Region
6.2.1. Global Primary Biliary Cholangitis (PBC) Treatment Market Export Revenue By Region
CHAPTER NO. 7 : COMPETITION ANALYSIS
7.1. Company Market Share Analysis
7.1.1. Global Primary Biliary Cholangitis (PBC) Treatment Market: Company Market Share
7.2. Global Primary Biliary Cholangitis (PBC) Treatment Market Company Revenue Market Share
7.3. Strategic Developments
7.3.1. Acquisitions & Mergers
7.3.2. New Product Launch
7.3.3. Regional Expansion
7.4. Competitive Dashboard
7.5. Company Assessment Metrics, 2024
CHAPTER NO. 8 : PRIMARY BILIARY CHOLANGITIS (PBC) TREATMENT MARKET – BY TREATMENT SEGMENT ANALYSIS
8.1. Primary Biliary Cholangitis (PBC) Treatment Market Overview By Treatment Segment
8.1.1. Primary Biliary Cholangitis (PBC) Treatment Market Revenue Share By Treatment
8.2. Medication
8.2.1. Obeticholic Acid
8.2.2. Ursodeoxycholic Acid
8.2.3. Others
8.3. Surgery
CHAPTER NO. 9 : PRIMARY BILIARY CHOLANGITIS (PBC) TREATMENT MARKET – BY GENDER SEGMENT ANALYSIS
9.1. Primary Biliary Cholangitis (PBC) Treatment Market Overview By Gender Segment
9.1.1. Primary Biliary Cholangitis (PBC) Treatment Market Revenue Share By Gender
9.2. Men
9.3. Women
CHAPTER NO. 10 : PRIMARY BILIARY CHOLANGITIS (PBC) TREATMENT MARKET – BY SALES CHANNEL (TREATMENT) SEGMENT ANALYSIS
10.1. Primary Biliary Cholangitis (PBC) Treatment Market Overview By Sales Channel (Treatment) Segment
10.1.1. Primary Biliary Cholangitis (PBC) Treatment Market Revenue Share By Sales Channel (Treatment)
10.2. Retail Pharmacy
10.3. Hospital Pharmacy
10.4. Online Pharmacy
CHAPTER NO. 11 : PRIMARY BILIARY CHOLANGITIS (PBC) TREATMENT MARKET – REGIONAL ANALYSIS
11.1. Primary Biliary Cholangitis (PBC) Treatment Market Overview By Region Segment
11.1.1. Global Primary Biliary Cholangitis (PBC) Treatment Market Revenue Share By Region
10.1.2. Regions
11.1.3. Global Primary Biliary Cholangitis (PBC) Treatment Market Revenue By Region
11.1.4. Treatment
11.1.5. Global Primary Biliary Cholangitis (PBC) Treatment Market Revenue By Medicatio
11.1.6. Gender
11.1.7. Global Primary Biliary Cholangitis (PBC) Treatment Market Revenue By Gender
11.1.8. Sales Channel (Treatment)
11.1.9. Global Primary Biliary Cholangitis (PBC) Treatment Market Revenue By Sales Channel (Treatment)
CHAPTER NO. 12 : NORTH AMERICA PRIMARY BILIARY CHOLANGITIS (PBC) TREATMENT MARKET – COUNTRY ANALYSIS
12.1. North America Primary Biliary Cholangitis (PBC) Treatment Market Overview By Country Segment
12.1.1. North America Primary Biliary Cholangitis (PBC) Treatment Market Revenue Share By Region
12.2. North America
12.2.1. North America Primary Biliary Cholangitis (PBC) Treatment Market Revenue By Country
12.2.2. Treatment
12.2.3. North America Primary Biliary Cholangitis (PBC) Treatment Market Revenue By Treatment
12.2.4. Gender
12.2.5. North America Primary Biliary Cholangitis (PBC) Treatment Market Revenue By Gender
12.2.6. Sales Channel (Treatment)
12.2.7. North America Primary Biliary Cholangitis (PBC) Treatment Market Revenue By Sales Channel (Treatment)
12.3. U.S.
12.4. Canada
12.5. Mexico
CHAPTER NO. 13 : EUROPE PRIMARY BILIARY CHOLANGITIS (PBC) TREATMENT MARKET – COUNTRY ANALYSIS
13.1. Europe Primary Biliary Cholangitis (PBC) Treatment Market Overview By Country Segment
13.1.1. Europe Primary Biliary Cholangitis (PBC) Treatment Market Revenue Share By Region
13.2. Europe
13.2.1. Europe Primary Biliary Cholangitis (PBC) Treatment Market Revenue By Country
13.2.2. Treatment
13.2.3. Europe Primary Biliary Cholangitis (PBC) Treatment Market Revenue By Treatment
13.2.4. Gender
13.2.5. Europe Primary Biliary Cholangitis (PBC) Treatment Market Revenue By Gender
13.2.6. Sales Channel (Treatment)
13.2.7. Europe Primary Biliary Cholangitis (PBC) Treatment Market Revenue By Sales Channel (Treatment)
13.3. UK
13.4. France
13.5. Germany
13.6. Italy
13.7. Spain
13.8. Russia
13.9. Rest of Europe
CHAPTER NO. 14 : ASIA PACIFIC PRIMARY BILIARY CHOLANGITIS (PBC) TREATMENT MARKET – COUNTRY ANALYSIS
14.1. Asia Pacific Primary Biliary Cholangitis (PBC) Treatment Market Overview By Country Segment
14.1.1. Asia Pacific Primary Biliary Cholangitis (PBC) Treatment Market Revenue Share By Region
14.2. Asia Pacific
14.2.1. Asia Pacific Primary Biliary Cholangitis (PBC) Treatment Market Revenue By Country
14.2.2. Treatment
14.2.3. Asia Pacific Primary Biliary Cholangitis (PBC) Treatment Market Revenue By Treatment
14.2.4. Gender
14.2.5. Asia Pacific Primary Biliary Cholangitis (PBC) Treatment Market Revenue By Gender
14.2.6. Sales Channel (Treatment)
14.2.7. Asia Pacific Primary Biliary Cholangitis (PBC) Treatment Market Revenue By Sales Channel (Treatment)
14.3. China
14.4. Japan
14.5. South Korea
14.6. India
14.7. Australia
14.8. Southeast Asia
14.9. Rest of Asia Pacific
CHAPTER NO. 15 : LATIN AMERICA PRIMARY BILIARY CHOLANGITIS (PBC) TREATMENT MARKET – COUNTRY ANALYSIS
15.1. Latin America Primary Biliary Cholangitis (PBC) Treatment Market Overview By Country Segment
15.1.1. Latin America Primary Biliary Cholangitis (PBC) Treatment Market Revenue Share By Region
15.2. Latin America
15.2.1. Latin America Primary Biliary Cholangitis (PBC) Treatment Market Revenue By Country
15.2.2. Treatment
15.2.3. Latin America Primary Biliary Cholangitis (PBC) Treatment Market Revenue By Treatment
15.2.4. Gender
15.2.5. Latin America Primary Biliary Cholangitis (PBC) Treatment Market Revenue By Gender
15.2.6. Sales Channel (Treatment)
15.2.7. Latin America Primary Biliary Cholangitis (PBC) Treatment Market Revenue By Sales Channel (Treatment)
15.3. Brazil
15.4. Argentina
15.5. Rest of Latin America
CHAPTER NO. 16 : MIDDLE EAST PRIMARY BILIARY CHOLANGITIS (PBC) TREATMENT MARKET – COUNTRY ANALYSIS
16.1. Middle East Primary Biliary Cholangitis (PBC) Treatment Market Overview By Country Segment
16.1.1. Middle East Primary Biliary Cholangitis (PBC) Treatment Market Revenue Share By Region
16.2. Middle East
16.2.1. Middle East Primary Biliary Cholangitis (PBC) Treatment Market Revenue By Country
16.2.2. Treatment
16.2.3. Middle East Primary Biliary Cholangitis (PBC) Treatment Market Revenue By Treatment
16.2.4. Gender
16.2.5. Middle East Primary Biliary Cholangitis (PBC) Treatment Market Revenue By Gender
16.2.6. Sales Channel (Treatment)
16.2.7. Middle East Primary Biliary Cholangitis (PBC) Treatment Market Revenue By Sales Channel (Treatment)
16.3. GCC Countries
16.4. Israel
16.5. Turkey
16.6. Rest of Middle East
CHAPTER NO. 17 : AFRICA PRIMARY BILIARY CHOLANGITIS (PBC) TREATMENT MARKET – COUNTRY ANALYSIS
17.1. Africa Primary Biliary Cholangitis (PBC) Treatment Market Overview By Country Segment
17.1.1. Africa Primary Biliary Cholangitis (PBC) Treatment Market Revenue Share By Region
17.2. Africa
17.2.1. Africa Primary Biliary Cholangitis (PBC) Treatment Market Revenue By Country
17.2.2. Treatment
17.2.3. Africa Primary Biliary Cholangitis (PBC) Treatment Market Revenue By Treatment
17.2.4. Gender
17.2.5. Africa Primary Biliary Cholangitis (PBC) Treatment Market Revenue By Gender
17.2.6. Sales Channel (Treatment)
17.2.7. Africa Primary Biliary Cholangitis (PBC) Treatment Market Revenue By Sales Channel (Treatment)
17.3. South Africa
17.4. Egypt
17.5. Rest of Africa
CHAPTER NO. 18 : COMPANY PROFILES
18.1. Intercept Pharmaceuticals, Inc.
18.1.1. Company Overview
18.1.2. Product Portfolio
18.1.3. Financial Overview
18.1.4.Recent Developments
18.1.5. Growth Strategy
18.1.6. SWOT Analysis
18.2. Teva Pharmaceutical
18.3. Sun Pharmaceutical Limited
18.4. Mylan Inc.
18.5. Allergan Inc.
18.6. Glenmark Pharmaceuticals
18.7. Eli Lilly and Company
18.8. Dr. Reddy’s Laboratories
18.9. Endo International plc
18.10. Novartis AG
18.11. Other Key Players