Acute Respiratory Distress Syndrome Treatment Market Overview:
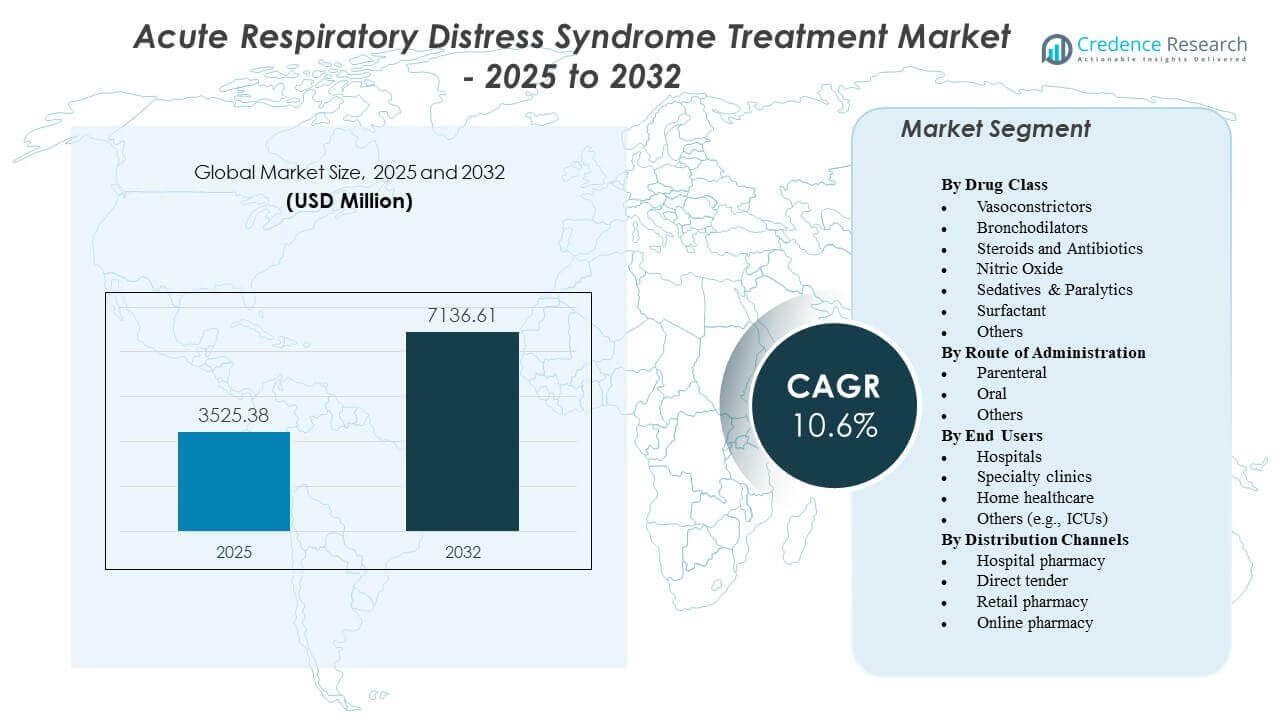
The Acute Respiratory Distress Syndrome Treatment Market is projected to grow from USD 3525.38 million in 2025 to an estimated USD 7136.61 million by 2032, with a compound annual growth rate (CAGR) of 10.6% from 2025 to 2032.
| RT ATTRIBUTE |
DETAILS |
| Historical Period |
2020-2023 |
| Base Year |
2024 |
| Forecast Period |
2025-2032 |
| Acute Respiratory Distress Syndrome Treatment Market Size 2025 |
USD 3525.38 million |
| Acute Respiratory Distress Syndrome Treatment Market, CAGR |
10.6% |
| Acute Respiratory Distress Syndrome Treatment Market Size 2032 |
USD 7136.61 million |
Acute Respiratory Distress Syndrome Treatment Market Insights:
- Rising cases of severe respiratory complications increase the need for mechanical ventilation, immunomodulators, and targeted supportive care, strengthening hospital adoption of structured ARDS treatment pathways.
- Limited ICU capacity in several regions, high treatment costs, and uneven access to specialized respiratory technologies continue to restrict wider adoption across developing healthcare systems.
- North America leads due to advanced emergency care frameworks and strong innovation in ARDS therapies, while Europe maintains steady growth supported by regulated clinical protocols and expanding hospital capabilities.
- Asia Pacific emerges as the fastest-growing region driven by rising infection burdens, healthcare modernization, and increased investment in critical care infrastructure across developing economies.
Acute Respiratory Distress Syndrome Treatment Market Drivers
Rising Global Burden of Critical Respiratory Conditions Driving Higher Treatment Demand
Growing respiratory complications in intensive care units push hospitals to adopt advanced therapies across the Acute Respiratory Distress Syndrome Treatment Market. Clinicians report steady growth in severe lung injury cases linked to infections, trauma, and sepsis. Higher patient admissions increase the need for structured ventilatory support. Medical teams focus on rapid stabilization to prevent long-term impairment. Strong clinical guidelines support early diagnosis to improve recovery rates. Healthcare providers expand ICU capabilities to manage rising caseloads. Government programs highlight the need for better acute care response systems. The Acute Respiratory Distress Syndrome Treatment Market strengthens due to broader awareness among hospital networks.
- For instance, Baxter’s PrisMax system has demonstrated high clinical precision, delivering a verified effluent accuracy of up to 0.98 through its fluid-removal catch-up algorithm. Its advanced CRRT performance supports ARDS patients who develop acute kidney injury, a comorbidity reported in 35%–44% of cases, where precise fluid balance directly influences respiratory outcomes and survival.
Access crucial information at unmatched prices!
Request your sample report today & start making informed decisions powered by Credence Research Inc.!
Download Sample
Growing Use of Mechanical Ventilation and Non-Invasive Respiratory Support Technologies
Improved respiratory devices support more efficient care delivery for patients with severe breathing challenges. Hospitals adopt high-flow oxygen systems to reduce complication risks. It helps teams stabilize critical patients faster across varied emergency settings. Clinicians rely on proven tools that support lung protection strategies. New ventilation modes help reduce pressure-related injuries. Wider device availability encourages rapid deployment during peak ICU load. Research efforts strengthen the understanding of personalized ventilation needs. Demand for safe and adaptable respiratory systems continues to rise.
Expanded Clinical Adoption of Anti-Inflammatory and Adjunctive Therapies
Drug developers introduce targeted therapies that address inflammation, which remains a major driver of ARDS severity. It supports tailored treatment plans for high-risk groups in intensive care. Clinicians use corticosteroids and adjunctive drugs to improve lung response in acute phases. Hospitals review updated protocols that reflect evolving clinical evidence. Strong research drives better therapeutic strategies for multi-organ complications. Broader treatment options help providers manage varied patient profiles. Increased trials expand the pipeline for next-generation medicines. Adoption rates improve as physicians gain confidence in updated drug regimens.
- For instance, Faron Pharmaceuticals has reported encouraging clinical findings for Traumakine (recombinant human IFN-beta-1a), showing potential to improve outcomes in patients with severe ARDS. Its trials demonstrated biologically meaningful effects on lung function and systemic inflammation, supporting continued development of the therapy as an investigational option.
Rising Emphasis on Early Intervention and Improved Critical Care Infrastructure
Growing demand for advanced emergency systems pushes governments to upgrade hospital readiness. It fuels stronger investment in diagnostic tools that detect respiratory decline sooner. ICU expansions support faster access to life-saving procedures across urban and rural areas. Tele-critical care models help specialists guide frontline teams remotely. Early response systems reduce mortality risks in severe respiratory events. Training programs enhance clinician capability in acute care management. Hospitals implement data-driven monitoring systems to track rapid patient changes. The Acute Respiratory Distress Syndrome Treatment Market benefits from stronger emergency preparedness frameworks.
Acute Respiratory Distress Syndrome Treatment Market Trends
Growing Use of Precision Medicine to Personalize Respiratory Care Pathways
Healthcare systems shift toward biomarker-based treatment plans that support tailored interventions. Clinicians review genetic and molecular data to refine therapeutic selection. It improves patient outcomes by reducing response variability across populations. Research centers explore adaptive strategies that target specific inflammatory pathways. Hospitals adopt monitoring platforms that collect real-time physiologic data. AI-enabled systems help teams predict respiratory decline with higher accuracy. Precision tools reduce the burden of trial-and-error treatment adjustments. This trend enhances trust in advanced respiratory care models.
- For instance, Thermo Fisher Scientific provides the B·R·A·H·M·S PCT assay, a highly specific biomarker used to distinguish bacterial co-infections in critically ill respiratory patients. Procalcitonin-guided protocols supported by this assay have demonstrated meaningful reductions in unnecessary antibiotic use and stronger antimicrobial stewardship in intensive care settings.
Integration of AI and Predictive Analytics in ICU Management
Intensive care units deploy smart systems that flag early deterioration signs in high-risk patients. Machine-learning tools support ventilation adjustments based on patient patterns. It helps clinicians manage complex respiratory cases with greater confidence. Automated alerts support faster decision cycles during emergencies. Hospitals invest in digital dashboards that track respiratory metrics around the clock. Predictive modeling improves resource planning under heavy ICU load. New AI platforms support safer weaning practices for ventilated patients. Technology adoption accelerates across major hospital networks.
Rising Shift Toward Minimally Invasive Ventilation Solutions
Clinicians favor non-invasive devices to reduce complications linked to traditional ventilation. Hospitals prioritize gentle support methods that protect fragile lung tissue. It improves patient mobility and reduces sedation needs. High-flow nasal oxygen systems gain wide interest for early intervention. Portable respiratory devices support flexible deployment during surge events. New mask designs improve comfort and reduce leakage issues. Demand grows for technologies that deliver stable and continuous airflow. This trend supports safer treatment pathways for moderate respiratory distress cases.
Greater Focus on Post-Acute Rehabilitation and Long-Term Recovery Programs
Many patients experience lasting impairment after acute respiratory distress episodes. Rehabilitation centers expand services targeting lung function improvement. It drives interest in structured post-ICU care models that track long-term recovery. Hospitals introduce early mobility programs to reduce weakness among survivors. Digital platforms help teams monitor breathing performance after discharge. New therapies target fibrosis risk in severe cases. Awareness campaigns highlight the importance of recovery planning. The Acute Respiratory Distress Syndrome Treatment Market gains momentum from broader rehabilitation strategies.
- For instance, Getinge’s Servo-u ventilator integrates NAVA (Neurally Adjusted Ventilatory Assist) technology, which uses the patient’s own diaphragmatic electrical activity to guide ventilation. This approach has been clinically shown to improve patient-ventilator synchrony and support smoother weaning, helping clinicians advance patients toward earlier rehabilitation.
Acute Respiratory Distress Syndrome Treatment Market Challenges Analysis
High Clinical Complexity and Limited Treatment Responsiveness Across Patient Groups
Severe respiratory distress cases vary widely, which complicates treatment planning for hospitals. Clinicians struggle to predict outcomes due to sudden physiologic changes. It increases uncertainty during ventilation adjustments in unstable patients. Drug responsiveness varies across age groups and comorbidity profiles. Limited biomarkers reduce the ability to guide targeted therapy. Hospital teams face pressure to manage sudden surges in critical cases. Long ICU stays increase resource strain during peak seasons. The Acute Respiratory Distress Syndrome Treatment Market must address these gaps to improve care reliability.
Infrastructure Limitations and High Treatment Costs Restrict Wider Adoption
Many regions lack advanced ICU systems that support modern respiratory care. It limits access to high-flow systems, advanced ventilators, and adjunctive therapies. Hospitals in low-resource settings face budget gaps that slow upgrades. Treatment expenses rise due to long ventilation duration and complex monitoring needs. Workforce shortages create challenges during emergency surges. Training gaps reduce the safe operation of advanced respiratory systems. Supply bottlenecks delay deployment of critical devices. These barriers slow broad adoption across several global markets.
Acute Respiratory Distress Syndrome Treatment Market Opportunities
Growing Scope for Advanced Ventilation Technologies and Smart Monitoring Systems
Hospitals seek next-generation respiratory tools that enhance treatment stability. It creates strong potential for innovations in lung-protective ventilation modes. AI-enabled platforms help teams refine decision-making during emergencies. Portable systems support flexible deployment across varied care settings. Remote monitoring expands access for high-risk groups in isolated areas. Device makers explore adaptive systems that align with patient-specific needs. Early diagnostic tools support faster triage in overcrowded emergency rooms. The Acute Respiratory Distress Syndrome Treatment Market benefits from rising hospital digitization efforts.
Expanding Research Pipelines and Rising Demand for Novel Drug Therapies
Drug developers focus on therapies that target inflammation and tissue injury. It encourages more clinical trials that explore combination regimens. Hospitals engage in evidence-based adoption of emerging medicines. New anti-fibrotic research supports long-term lung recovery for survivors. Partnerships between academic centers and industry push new therapeutic options forward. Wider use of adjunctive drugs supports multi-stage care models. Investment programs help expand global trial participation. These opportunities support steady innovation across acute respiratory treatment platforms.
Acute Respiratory Distress Syndrome Treatment Market Segmentation Analysis:
By Drug Class
The Acute Respiratory Distress Syndrome Treatment Market shows diverse adoption across drug classes that support both acute stabilization and ongoing respiratory management. Vasoconstrictors help maintain circulation during critical episodes, while bronchodilators support airway widening in complex cases. Steroids and antibiotics remain widely used due to strong clinical evidence in inflammatory and infection-related triggers. Nitric oxide supports oxygenation in severe cases that demand rapid improvement. Sedatives and paralytics help clinicians manage ventilation needs in unstable patients. Surfactants gain interest in treatment pathways for patients with impaired lung elasticity. Other drug options support personalized plans that reflect varied patient responses.
- For instance, Mallinckrodt Pharmaceuticals’ INOmax (nitric oxide) for inhalation acts as a selective pulmonary vasodilator that improves oxygenation in severe ARDS by reducing intrapulmonary shunting and redirecting blood flow toward well-ventilated lung regions. Clinical studies show that responders often achieve meaningful increases in the PaO₂/FiO₂ ratio, supporting its role as a rescue therapy for life-threatening hypoxemia.
By Route of Administration
The Acute Respiratory Distress Syndrome Treatment Market uses administration routes that match emergency care needs across diverse patient groups. Parenteral delivery leads due to its fast systemic action in unstable patients. It supports hospitals that treat severe attacks requiring controlled dosing. Oral formulations serve mild or supportive cases where patients maintain swallowing capability. Other routes help clinicians introduce targeted therapies that support specialized interventions. Hospitals select routes based on treatment urgency, patient tolerance, and drug performance requirements.
By End Users
The Acute Respiratory Distress Syndrome Treatment Market records strong demand from hospitals that manage high volumes of critical respiratory cases. It supports advanced ventilation procedures and continuous monitoring. Specialty clinics use tailored protocols for patients who require structured follow-up. Home healthcare teams support extended recovery for survivors who need supervised support. Other end-user groups, including ICUs, shape therapy adoption through advanced care models and rapid-response systems.
- For instance, the Mayo Clinic has implemented its AWARE (Ambient Warning and Response Evaluation) decision-support system in specialized ICUs, using real-time data integration to improve adherence to lung-protective ventilation strategies. The platform enhances clinician awareness of critical trends and supports more consistent application of ARDS care protocols.
By Distribution Channels
The Acute Respiratory Distress Syndrome Treatment Market relies on hospital pharmacies that manage emergency drug supply for critical care units. Direct tender channels help institutions secure essential therapies in bulk to support uninterrupted treatment cycles. Retail pharmacies serve patients who require ongoing prescriptions during recovery. Online pharmacy platforms expand access to supportive medications in regions with limited physical infrastructure.
Segmentation:
By Drug Class
- Vasoconstrictors
- Bronchodilators
- Steroids and Antibiotics
- Nitric Oxide
- Sedatives & Paralytics
- Surfactant
- Others
By Route of Administration
By End Users
- Hospitals
- Specialty clinics
- Home healthcare
- Others (e.g., ICUs)
By Distribution Channels
- Hospital pharmacy
- Direct tender
- Retail pharmacy
- Online pharmacy
By Region
- North America
- Europe
- Germany
- France
- U.K.
- Italy
- Spain
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- South-east Asia
- Rest of Asia Pacific
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Middle East & Africa
- GCC Countries
- South Africa
- Rest of the Middle East and Africa
Regional Analysis:
North America leads the Acute Respiratory Distress Syndrome Treatment Market with more than 38% share supported by strong ICU capacity and early adoption of advanced ventilation systems. Hospitals in the region use structured care pathways that enhance patient outcomes during severe respiratory episodes. It benefits from a high concentration of device manufacturers that introduce updated treatment technologies. Research centers expand clinical trials that support rapid approval cycles for emerging drugs. Insurance systems improve access to critical care tools. Strong awareness programs encourage faster patient reporting during respiratory distress.
Europe holds around 29% share driven by investments in specialized respiratory units and strong clinical guidelines across major countries. Hospitals adopt advanced ventilatory support systems that improve care for high-risk groups. It gains support from strict regulatory frameworks that favor evidence-based therapies. National health programs increase spending on ICU expansions that support rapid treatment access. Research collaborations between universities and companies drive interest in immunomodulatory drugs. Public health agencies promote training programs for emergency teams.
Asia Pacific records close to 22% share and shows rising adoption of critical care technologies across emerging healthcare systems. It benefits from hospital modernization programs that strengthen ICU readiness during acute respiratory events. Demand grows in China, India, and Southeast Asia due to rising incidence of severe infections. Large patient volumes push hospitals to scale ventilatory support and drug-based interventions. Government schemes expand critical care coverage for underserved regions. Growing private healthcare investments accelerate uptake of innovative respiratory solutions.
Shape Your Report to Specific Countries or Regions & Enjoy 30% Off!
Key Player Analysis:
- Faron Pharmaceuticals
- BioMarck Pharmaceuticals
- GE Healthcare
- Hamilton Company
- Medtronic plc
- Koninklijke Philips N.V.
- Athersys
- United Therapeutics
- HEALIOS K.K.
- Drägerwerk AG & Co. KGaA
- ALung Technologies Inc. (LivaNova PLC)
- Getinge
- Edesa Biotech, Inc.
- Relief Therapeutics
Competitive Analysis:
The Acute Respiratory Distress Syndrome Treatment Market features strong competition driven by companies that supply critical care equipment, drug therapies, and advanced respiratory support systems. Key players expand portfolios to meet ICU demand during severe respiratory cases. It encourages firms to upgrade ventilators, monitoring systems, and adjunctive therapies that support patient stabilization. Pharmaceutical companies invest in immunomodulators, surfactants, and targeted anti-inflammatory agents to improve clinical outcomes. Strategic partnerships support accelerated clinical development in high-priority drug categories. Device manufacturers focus on technology that enhances ventilation precision and reduces treatment-related risks. Companies also pursue global distribution alliances to strengthen presence across established and emerging markets.
Recent Developments:
- In June 2025, BioAegis Therapeutics received FDA Fast Track designation for its novel immune modulator rhu-pGSN (recombinant human plasma gelsolin), aimed at treating moderate to severe ARDS, enabling expedited regulatory review and accelerating its pathway to patients.
- In March 2025, Healios K.K. announced positive findings from clinical studies of MultiStem cell therapy (invimestrocel; HLCM-051) for ARDS treatment demonstrating reductions in ventilator usage duration and improved survival outcomes.
Report Coverage:
The research report offers an in-depth analysis based on Drug Class, Route of Administration, End Users, and Distribution Channels. It details leading market players, providing an overview of their business, product offerings, investments, revenue streams, and key applications. Additionally, the report includes insights into the competitive environment, SWOT analysis, current market trends, as well as the primary drivers and constraints. Furthermore, it discusses various factors that have driven market expansion in recent years. The report also explores market dynamics, regulatory scenarios, and technological advancements that are shaping the industry. It assesses the impact of external factors and global economic changes on market growth. Lastly, it provides strategic recommendations for new entrants and established companies to navigate the complexities of the market.
Future Outlook:
- Growing ICU modernization will support wider adoption of advanced respiratory technologies across the Acute Respiratory Distress Syndrome Treatment Market.
- Rising focus on precision therapy will push development of targeted drugs that improve patient recovery.
- Increased investment in ventilatory innovation will support safer intervention methods for diverse patient groups.
- Digital monitoring platforms will help clinicians optimize treatment decisions in real time.
- Expanding clinical research pipelines will introduce new immunomodulators that strengthen treatment outcomes.
- Greater attention to post-acute recovery will drive demand for long-term respiratory support services.
- Strengthened emergency preparedness frameworks will improve access to rapid stabilization tools.
- Higher collaboration between hospitals and technology firms will accelerate adoption of smart respiratory devices.
- Growth in public-private healthcare spending will improve availability of critical care therapies.
- Rising awareness of respiratory emergencies will support early intervention and reduce progression to severe cases.




