Adult Malignant Glioma Therapeutics Market Overview:
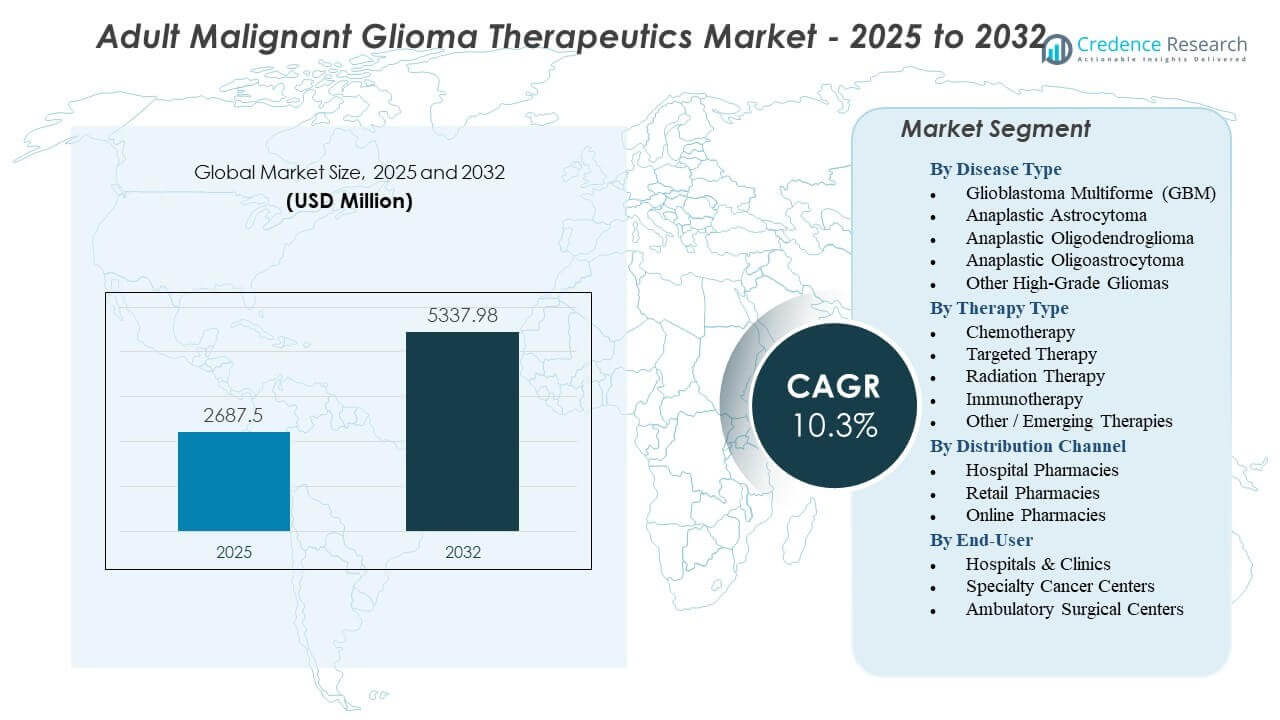
The Adult Malignant Glioma Therapeutics Market is projected to grow from USD 2687.5 million in 2025 to an estimated USD 5337.98 million by 2032, with a compound annual growth rate (CAGR) of 10.3% from 2025 to 2032.
| RT ATTRIBUTE |
DETAILS |
| Historical Period |
2020-2023 |
| Base Year |
2024 |
| Forecast Period |
2025-2032 |
| Adult Malignant Glioma Therapeutics Market Size 2025 |
USD 2687.5 million |
| Adult Malignant Glioma Therapeutics Market, CAGR |
10.3% |
| Adult Malignant Glioma Therapeutics Market Size 2032 |
USD 5337.98 million |
Adult Malignant Glioma Therapeutics Market Insights:
- Rising incidence of high-grade gliomas and broader use of targeted and immunotherapy options continue to strengthen therapeutic demand across major cancer centers.
- Market restraints include high treatment costs, limited long-term effectiveness of current therapies, and regulatory challenges linked to complex clinical trial requirements.
- North America holds the leading share due to strong oncology infrastructure and rapid uptake of next-generation therapies, followed by Europe with expanding precision-medicine programs.
- Asia Pacific shows the fastest improvement in access and diagnostic capability, while emerging regions gain traction through gradual investment in screening and treatment services.
Adult Malignant Glioma Therapeutics Market Drivers
Rising Incidence of High-Grade Gliomas and Growing Need for Effective Therapeutic Solutions
The Adult Malignant Glioma Therapeutics Market grows due to steady increases in high-grade tumor diagnoses among aging populations. Many patients reach clinics with advanced symptoms that demand faster therapeutic planning. Hospitals expand diagnostic capacity to manage rising caseloads more efficiently. Strong awareness programs encourage adults to seek neurological evaluations earlier. Early detection improves access to multimodal treatment pathways. Health systems invest in oncology infrastructure that supports wider drug adoption. Research bodies promote new protocols that enhance treatment sequencing. The market responds to these needs through expanded innovation pipelines.
Access crucial information at unmatched prices!
Request your sample report today & start making informed decisions powered by Credence Research Inc.!
Download Sample
Advancement in Targeted Therapy Platforms Strengthening Precision Treatment Pathways
Targeted therapy improves treatment accuracy by focusing on tumor-specific mutations. Drug developers design molecules that block key pathways linked to tumor expansion. These platforms create higher interest among oncology teams seeking tailored care. Better biomarker panels give clinicians deeper insights into patient profiles. Molecular mapping improves accuracy in high-risk populations. Treatment optimization supports more predictable outcomes across clinical centers. Precision-based approaches encourage investment from global drug makers. The Adult Malignant Glioma Therapeutics Market benefits from wider adoption of these advanced molecular strategies.
Stronger Research Funding and Growth of Academic–Industry Collaboration Networks
Funding bodies increase support for neuro-oncology research focused on aggressive tumor types. Grants promote discovery programs that explore novel molecular targets. Universities collaborate with pharmaceutical companies to accelerate therapeutic development. These networks speed preclinical validation timelines. Such partnerships build stronger access to infrastructure that supports drug optimization. Researchers adopt advanced screening technology to test therapeutic combinations. Wider collaboration improves clinical understanding of complex tumor behavior. The market grows as these programs translate experimental findings into viable clinical candidates.
- For example, the National Cancer Institute allocated USD 236.1 million to Brain and CNS tumor research in FY 2023, supporting a broad portfolio of glioma-focused studies. This funding helps maintain over 140 active clinical trials targeting glioma and glioblastoma across NCI-supported research networks.
Expanding Adoption of Combination Therapy Models for Enhanced Survival Outcomes
Combination therapy becomes a core treatment approach across leading oncology centers. Clinicians integrate surgery, radiation, and pharmacological regimens to improve survival periods. Sequential therapy models help manage recurrence patterns more effectively. Improved clinical mapping supports better selection of supportive drugs. Hospitals adopt digital tools that track treatment progression more precisely. Patients benefit from tailored approaches that improve quality of life. Trial frameworks test new combinations with improved safety outcomes. It encourages broader interest in advanced therapy portfolios among global stakeholders.
- For instance, the Stupp protocol combining temozolomide with radiotherapy improved two-year survival rates from 10% to 26% in landmark clinical evaluations.
Adult Malignant Glioma Therapeutics Market Trends
Growing Integration of Immuno-Oncology Agents into Standard Therapeutic Pathways
Immuno-oncology gains traction as developers introduce checkpoint inhibitors and cellular therapies. These agents aim to strengthen immune responses against aggressive tumor cells. Trial enrollment expands across key oncology hubs worldwide. Researchers explore immune-modulating combinations to reduce relapse patterns. Broader acceptance among clinicians supports ongoing clinical use. Digital imaging tools improve tracking of therapy-induced immune activity. Hospitals adopt new frameworks that help teams evaluate immune-driven responses. The Adult Malignant Glioma Therapeutics Market observes growing interest in these novel immune-focused regimens.
- For instance, the CheckMate-143 trial by Bristol Myers Squibb enrolled over 440 glioblastoma patients globally to evaluate nivolumab’s immunotherapeutic impact against standard therapy.
Increasing Use of AI-Driven Tools to Improve Treatment Planning and Diagnostic Accuracy
AI platforms support clinicians by analyzing complex tumor data more quickly. Predictive models help guide treatment pathways with stronger precision. Automated workflows reduce diagnostic delays in busy cancer centers. Machine-learning tools identify subtle tumor changes on imaging scans. These insights improve therapy selections during early planning. Hospitals deploy AI-supported systems to strengthen survival analysis. Research teams use AI to optimize trial design and patient matching. This trend drives digital transformation across neuro-oncology practices.
Shift Toward Personalized Cellular Therapies Supported by Strong Clinical Momentum
Cellular therapies gain wider attention as developers refine engineering methods. Advancements in manufacturing support greater scalability for clinical use. Trial networks test engineered immune cells in advanced tumor cases. Improved delivery systems support safer patient administration. Clinicians value these therapies for their potential to target deeper tumor layers. Specialized labs expand capacity to support rising production volumes. Regulatory bodies evaluate new frameworks for cell-based submissions. This trend reshapes long-term therapeutic innovation across global oncology markets.
- For instance, City of Hope’s IL13Rα2-targeted CAR-T program reported radiographic tumor regression in multiple glioblastoma patients, including a documented case showing marked response across several intracranial sites.
Expansion of Real-World Evidence Programs to Strengthen Post-Approval Decision Making
Real-world evidence programs help validate clinical outcomes across diverse populations. Hospitals collect treatment data that supports stronger regulatory evaluation. Payers use real-world insights to refine reimbursement policies. Researchers identify variation patterns that influence therapy selection. Digital registries help track long-term survival trends. Pharmaceutical companies rely on these datasets to guide future development. Real-world frameworks improve visibility into treatment performance beyond trial settings. It supports more informed market expansion strategies.
Adult Malignant Glioma Therapeutics Market Challenges Analysis
High Tumor Heterogeneity and Limited Effectiveness of Current Treatment Approaches
Tumor heterogeneity slows progress across the Adult Malignant Glioma Therapeutics Market. Complex cell variations weaken therapeutic response rates in many patients. Clinicians struggle to predict how tumors adapt during treatment cycles. Limited penetration of some drugs reduces impact on aggressive tumor regions. Surgical limitations prevent complete tumor removal in several cases. Recurrence patterns remain common even after strong therapy schedules. Research teams work to decode tumor evolution through genomic tools. Drug developers face challenges designing therapies that target diverse cell populations.
Regulatory Complexity and High Development Costs Hindering Market Expansion
Therapeutic development faces long approval timelines due to strict regulatory evaluations. Trial protocols demand strong safety evidence for high-risk patients. Developers must invest heavily in extended trial cycles. High research expenses restrict smaller companies from entering the field. Manufacturing advanced therapies requires specialized equipment. Payers show caution when reimbursing new treatments with uncertain long-term data. Access to high-cost therapies remains limited in several regions. It slows growth momentum for emerging therapeutic platforms.
Adult Malignant Glioma Therapeutics Market Opportunities
Emergence of Next-Generation Modalities Creating New Therapeutic Pathways
Next-generation technologies create strong expansion potential for the Adult Malignant Glioma Therapeutics Market. Gene-editing tools provide access to deeper tumor targets. Nanotechnology platforms support controlled drug delivery with higher precision. Researchers explore oncolytic viruses that attack tumor cells directly. These methods promise higher tolerance among adult patients. Trial sponsors focus on novel pathways to reduce relapse patterns. Better engineering frameworks strengthen therapeutic safety across complex cases. Global investors show interest in companies developing next-generation oncology ecosystems.
Rising Focus on Early Detection, Digital Monitoring, and Long-Term Care Models
Early detection programs open opportunities for new diagnostic platforms. AI-powered imaging systems help find small tumor formations earlier. Monitoring tools track progression in real time for high-risk populations. Hospitals expand digital care models that integrate continuous data review. Clinicians value remote insights that help adjust therapies faster. Stronger patient engagement supports long-term treatment compliance. Pharmaceutical companies design drugs that align with earlier intervention windows. These trends build a multi-layered opportunity landscape across therapeutic development.
Market Segmentation Analysis:
By Disease Type
The Adult Malignant Glioma Therapeutics Market sees strong dominance from Glioblastoma Multiforme due to its high prevalence and aggressive nature. It drives wider adoption of multimodal treatment pathways that support complex clinical needs. Anaplastic Astrocytoma and Anaplastic Oligodendroglioma show steady demand for molecularly guided therapies that improve survival prospects. Treatment strategies for Anaplastic Oligoastrocytoma rely on refined diagnostic workflows that support precise classification. Other high-grade gliomas create demand for niche therapies that target rare but challenging tumor variants. Each disease type expands treatment diversity across clinical centers. Providers focus on early detection to manage progression effectively. These variations shape long-term growth across neuro-oncology segments.
- For instance, Foundation Medicine’s comprehensive genomic profiling detects over 300 tumor-associated alterations in glioma samples, enabling precise classification across multiple high-grade subtypes.
By Therapy Type
The Adult Malignant Glioma Therapeutics Market benefits from strong demand for chemotherapy due to its established role in first-line care. It supports consistent use of temozolomide and combination regimens across multiple disease types. Targeted therapies gain traction with growing interest in IDH and EGFR pathway inhibitors. Radiation therapy maintains a critical role with expanding use of IMRT and stereotactic systems. Immunotherapy attracts attention from trial sponsors developing checkpoint inhibitors and cell-based platforms. Other emerging therapies such as TTFields and LITT strengthen advanced treatment pathways. These options enhance personalization opportunities for complex tumor management. The segment widens access to differentiated interventions.
By Distribution Channel
The Adult Malignant Glioma Therapeutics Market depends heavily on hospital pharmacies that manage most treatment prescriptions for high-grade cases. It supports continuous drug dispensing for complex therapy schedules. Retail pharmacies handle supportive medications used during long-term care. Online pharmacies gain traction through improved access for remote patients seeking refills. Digital channels help reduce travel barriers for adults managing chronic treatment cycles. Hospitals monitor therapy adherence through integrated pharmacy systems. These channels work together to maintain uninterrupted treatment supply. Growth across each channel supports better patient continuity in neuro-oncology care.
By End-User
The Adult Malignant Glioma Therapeutics Market is anchored by hospitals and clinics that deliver most frontline interventions. It supports comprehensive treatment paths including surgery, radiation, and drug therapies. Specialty cancer centers strengthen advanced care by offering clinical trials and precision-medicine programs. Ambulatory surgical centers perform select procedures that support early-stage planning. Each end-user group adopts digital tools that improve coordination across oncology teams. Providers emphasize early diagnosis to reduce progression complexity. These facilities shape treatment accessibility for diverse patient groups. Their adoption strategies determine future therapeutic integration across global markets.
- For instance, MD Anderson Cancer Center manages over 1,500 active clinical trials at any time, including multiple glioblastoma programs that expand therapeutic access for complex cases.
Segmentation:
By Disease Type
- Glioblastoma Multiforme (GBM)
- Anaplastic Astrocytoma
- Anaplastic Oligodendroglioma
- Anaplastic Oligoastrocytoma
- Other High-Grade Gliomas
By Therapy Type
- Chemotherapy
- Targeted Therapy
- Radiation Therapy
- Immunotherapy
- Other / Emerging Therapies
By Distribution Channel
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
By End-User
- Hospitals & Clinics
- Specialty Cancer Centers
- Ambulatory Surgical Centers
By Region
- North America
- Europe
- Germany
- France
- U.K.
- Italy
- Spain
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- South-east Asia
- Rest of Asia Pacific
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Middle East & Africa
- GCC Countries
- South Africa
- Rest of the Middle East and Africa
Regional Analysis:
North America holds the largest share of the Adult Malignant Glioma Therapeutics Market at nearly 45%, driven by strong diagnostic capacity and broad access to advanced oncology therapies. The region benefits from high treatment awareness and extensive adoption of precision-medicine tools. Pharmaceutical companies invest in clinical trials that support continuous innovation. It expands access to new therapeutic pathways across major cancer centers. Hospitals implement advanced radiation and immunotherapy models to improve patient outcomes. The region maintains leadership due to strong reimbursement structures and ongoing research commitments.
Europe accounts for roughly 30% of the Adult Malignant Glioma Therapeutics Market and maintains steady demand for combination therapies. Countries expand molecular testing programs that support more accurate classification of high-grade gliomas. Treatment centers adopt new targeted therapy protocols supported by regulatory approvals. Clinical networks encourage trial participation for emerging therapies across multiple nations. It strengthens long-term adoption of advanced neuro-oncology interventions. Growth stays consistent through strong academic engagement and rising investment in high-grade glioma research.
Asia Pacific captures about 18% of the Adult Malignant Glioma Therapeutics Market and shows the fastest growth due to rising cancer incidence and improving oncology infrastructure. Governments strengthen diagnostic capacity across developing healthcare systems. Regional manufacturers increase focus on affordable treatment options. Hospitals upgrade radiotherapy equipment to support advanced clinical workflows. It encourages wider adoption of multimodal treatment strategies in urban centers. Remaining regions, including Latin America and the Middle East & Africa, represent the final 7%, supported by growing investment but limited by infrastructure gaps.
Shape Your Report to Specific Countries or Regions & Enjoy 30% Off!
Key Player Analysis:
Competitive Analysis:
The Adult Malignant Glioma Therapeutics Market features strong competition among global pharmaceutical companies working to expand oncology pipelines. Firms like Merck, Roche, Pfizer, and AbbVie invest in immunotherapy and targeted drug development to strengthen clinical outcomes. It drives deeper focus on molecularly guided therapies that support higher treatment precision. Companies expand partnerships with academic centers to accelerate clinical trial timelines. New entrants explore cell therapy and oncolytic virus platforms to differentiate their portfolios. Established firms use strong commercial networks to maintain market presence across high-demand regions. Developers also refine radiation-linked innovations to support combination therapy adoption. Competitive intensity grows as companies pursue next-generation strategies for aggressive glioma management.
Recent Developments:
- In April 2025, Biocon Biologics received FDA approval for Jobevne (bevacizumab-nwgd), a biosimilar indicated for multiple cancer types including recurrent glioblastoma in the adult malignant glioma therapeutics market, enhancing affordable treatment access amid rising biosimilar competition
- In February 2025, the FDA granted priority review to dordaviprone (from Jazz Pharmaceuticals, following its USD 935 million acquisition of Chimerix) for H3K27M-mutant diffuse glioma, with a PDUFA target action date of August 18, 2025, marking a potential first approval for this rare subtype.
- In October 2024, Merck & Co., Inc. acquired Modifi Biosciences for $30 million upfront (with potential milestones up to $1.3 billion) gaining a preclinical asset (MOD-246) targeting glioblastoma, a key adult malignant glioma, to address temozolomide-resistant cases.
Report Coverage:
The research report offers an in-depth analysis based on Disease Type, Therapy Type, Distribution Channel, End-User, and Region. It details leading market players, providing an overview of their business, product offerings, investments, revenue streams, and key applications. Additionally, the report includes insights into the competitive environment, SWOT analysis, current market trends, as well as the primary drivers and constraints. Furthermore, it discusses various factors that have driven market expansion in recent years. The report also explores market dynamics, regulatory scenarios, and technological advancements that are shaping the industry. It assesses the impact of external factors and global economic changes on market growth. Lastly, it provides strategic recommendations for new entrants and established companies to navigate the complexities of the market.
Future Outlook:
- Growing adoption of targeted and immunotherapy platforms will shape long-term therapeutic advancement across global oncology networks.
- Rising investment in molecular diagnostics will support broader access to precision-based approaches for aggressive glioma cases.
- Expansion of combination therapy models will strengthen survival outcomes across major cancer centers.
- Increasing trial activity in cell therapy and oncolytic virus platforms will open new treatment pathways.
- Higher adoption of AI-driven planning tools will support faster treatment decisions and improved clinical accuracy.
- Greater availability of radiotherapy innovations will help enhance tumor control in advanced disease stages.
- Strengthening collaboration between academic institutions and pharmaceutical firms will accelerate therapy development cycles.
- Wider deployment of digital monitoring tools will support more effective long-term disease management.
- Growth in reimbursement support across developed markets will enhance access to next-generation therapies.
- Rising focus on early diagnosis will expand treatment windows and influence future adoption patterns across the Adult Malignant Glioma Therapeutics Market.




