America Cancer Immunotherapy Market Overview:
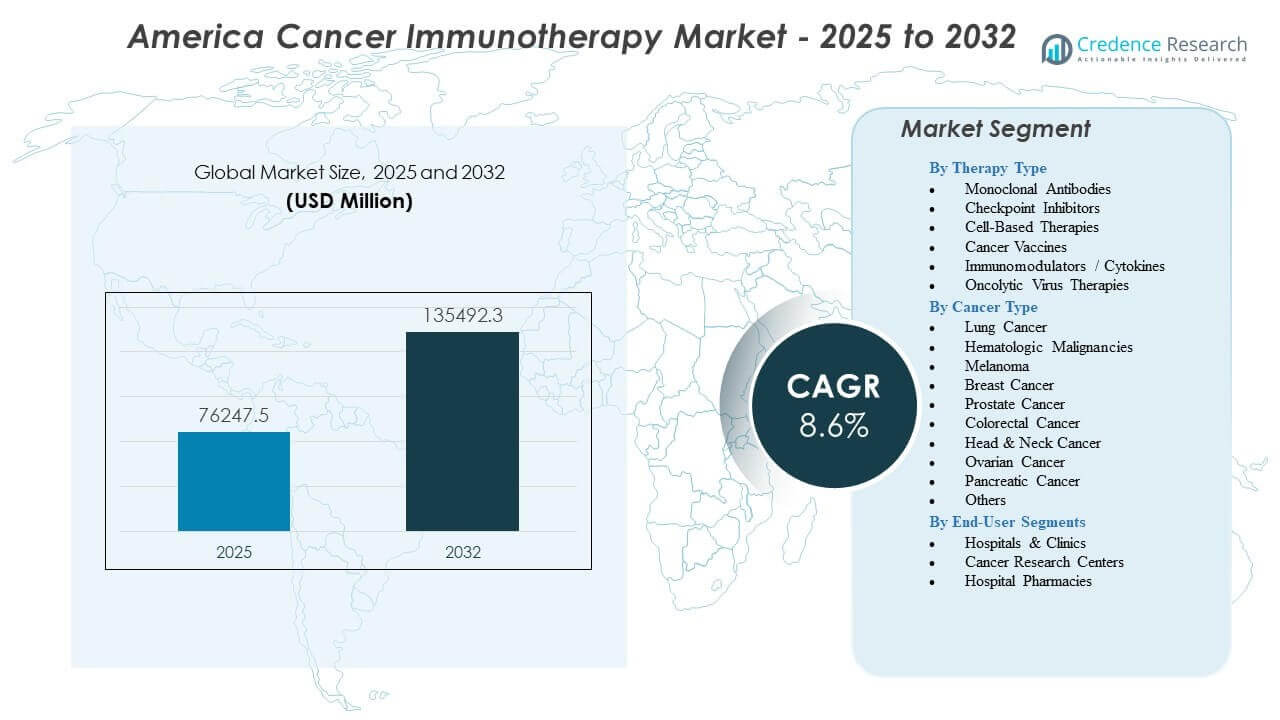
The America Cancer Immunotherapy Market is projected to grow from USD 76,247.5 million in 2025 to an estimated USD 135,492.3 million by 2032, with a compound annual growth rate (CAGR) of 8.6% from 2025 to 2032.
Growth in the market is driven by increasing cancer incidence, rapid advancement in immune-oncology pipelines, and wider clinical acceptance of checkpoint inhibitors, monoclonal antibodies, and cell-based therapies.
| REPORT ATTRIBUTE |
DETAILS |
| Historical Period |
2020-2024 |
| Base Year |
2025 |
| Forecast Period |
2026-2032 |
| America Cancer Immunotherapy Market Size 2025 |
USD 76,247.5 million |
| America Cancer Immunotherapy Market, CAGR |
8.6% |
| America Cancer Immunotherapy Market Size 2032 |
USD 135,492.3 million |
America Cancer Immunotherapy Market Insights:
- Rising cancer incidence, rapid immunotherapy innovation, and expanding adoption of checkpoint inhibitors and cell-based treatments drive sustained demand across clinical settings.
- High treatment costs, manufacturing complexity, and limited infrastructure for advanced therapies in certain regions create restraints that shape market accessibility.
- North America leads the market due to strong oncology networks, broad reimbursement, and early adoption of advanced biologics, while Canada supports growth through national cancer programs.
- Latin America emerges gradually, with Brazil and Mexico expanding specialized centers and improving diagnostics, enhancing long-term growth potential in the region.
America Cancer Immunotherapy Market Drivers
Rising Cancer Burden And Expanding Clinical Adoption Of Advanced Immunotherapies Across Major Tumor Types
Cancer incidence continues to rise across the United States and Canada. Lung, breast, colorectal, and prostate cancers drive high treatment demand. Clinicians prefer immune-based drugs for durable response rates. Checkpoint inhibitors show survival benefits in multiple tumor types. Hospitals expand oncology units to support complex biologic therapies. Favorable reimbursement policies improve patient access in key markets. Strong physician awareness supports early therapy adoption. The America Cancer Immunotherapy Market benefits from broad label expansions and clinical guideline support.
Robust Research Funding And Accelerated Regulatory Approvals Strengthening Product Pipelines
Public and private funding for oncology research remains strong. Biopharma firms invest heavily in immune-oncology pipelines. Regulatory agencies provide priority review pathways for breakthrough drugs. Fast-track designations reduce time to market for novel agents. Clinical trials expand across academic cancer centers. Collaboration between biotech firms and universities improves trial efficiency. Large pharmaceutical companies pursue licensing and acquisition strategies. This environment drives steady innovation across monoclonal antibodies and cell therapies.
- For instance, Bristol Myers Squibb’s relatlimab–nivolumab combination received FDA approval after showing doubled progression-free survival compared with nivolumab alone in RELATIVITY-047.
Access crucial information at unmatched prices!
Request your sample report today & start making informed decisions powered by Credence Research Inc.!
Download Sample
Technological Advancements In Cell Therapy Platforms And Precision Biomarker Development
CAR-T and TCR-based platforms show strong therapeutic potential. Manufacturing technologies improve cell expansion and quality control. Automated systems reduce production variability and costs. Biomarker testing guides patient selection and improves outcomes. Companion diagnostics support targeted therapy decisions. Genomic profiling enables personalized immunotherapy approaches. Treatment centers build specialized infrastructure for cell therapy delivery. Technology progress strengthens confidence among oncologists and payers.
Growing Healthcare Infrastructure And Strategic Industry Collaborations Across The Americas
Cancer centers increase capacity to administer biologics and cell therapies. Cross-border partnerships expand clinical research networks. Pharmaceutical alliances accelerate drug development programs. Contract manufacturing organizations scale biologic production. Government programs support oncology care access in underserved regions. Digital health tools improve patient monitoring and adherence. Insurance providers broaden coverage for high-value therapies. Strong ecosystem coordination sustains long-term market expansion.
- For instance, AstraZeneca and Daiichi Sankyo expanded U.S. oncology infrastructure following the success of Enhertu, which delivered a confirmed objective response rate of 52.3% in HER2-low breast cancer.
America Cancer Immunotherapy Market Trends
Shift Toward Combination Immunotherapy Regimens To Enhance Clinical Response Rates
Oncologists increasingly prescribe combination regimens for complex cancers. Checkpoint inhibitors pair with chemotherapy or targeted agents. Dual immunotherapy protocols gain approval in advanced melanoma and lung cancer. Clinical data show improved progression-free survival in select patients. Physicians tailor regimens based on tumor biology. Treatment guidelines evolve to reflect combination evidence. Biopharma companies design trials around multi-drug strategies. The America Cancer Immunotherapy Market reflects this transition toward integrated treatment models.
- For instance, Opdivo plus Yervoy from Bristol Myers Squibb doubled median progression-free survival in melanoma compared with Yervoy alone in pivotal studies.
Expansion Of Immunotherapy Use Into Earlier Lines Of Treatment And Adjuvant Settings
Drug developers pursue approvals in early-stage disease. Adjuvant immunotherapy gains traction after surgery. Neoadjuvant approaches show promise in high-risk tumors. Clinical trials test immune agents before standard chemotherapy. Physicians aim to prevent recurrence through early immune activation. Health systems adopt updated oncology protocols. Broader label indications expand eligible patient pools. Early intervention strategies redefine treatment pathways.
Adoption Of Real-World Evidence And Value-Based Care Models In Oncology
Healthcare providers rely on real-world data to assess outcomes. Payers evaluate therapy performance beyond clinical trials. Value-based contracts link reimbursement to patient response. Digital registries track safety and long-term survival. Data analytics platforms support treatment optimization. Pharmaceutical firms invest in outcomes research programs. Hospitals integrate electronic health records for better reporting. Evidence-driven decision frameworks shape therapy selection.
- For instance, real-world analyses of Merck’s pembrolizumab in metastatic lung cancer showed overall survival outcomes consistent with clinical trial results across more than 8,000 U.S. patients.
Emergence Of Next-Generation Immunotherapies Beyond Traditional Checkpoint Inhibitors
Bispecific antibodies gain attention in hematologic malignancies. Personalized cancer vaccines enter late-stage trials. Tumor-infiltrating lymphocyte therapies expand clinical exploration. Oncolytic virus platforms show immune activation potential. Novel immune targets diversify drug pipelines. Precision engineering enhances antibody specificity. Research teams explore multi-target immune modulation. Innovation focus shifts toward differentiated mechanisms of action.
America Cancer Immunotherapy Market Challenges Analysis
High Treatment Costs And Complex Manufacturing Processes Limiting Broad Patient Access
Immunotherapy drugs often carry high price points. Cell therapies require specialized production facilities. Manufacturing involves strict quality control protocols. Limited production capacity restricts supply in some regions. Insurance approval processes delay treatment initiation. Smaller hospitals face infrastructure constraints. Patients encounter financial burden despite coverage. The America Cancer Immunotherapy Market faces pressure to balance innovation with affordability.
Immune-Related Adverse Events And Variability In Patient Response Affecting Clinical Confidence
Checkpoint inhibitors may trigger immune-related toxicities. Some patients experience severe organ inflammation. Clinicians must monitor adverse reactions closely. Response rates vary across tumor types and individuals. Biomarker reliability remains imperfect in some cancers. Resistance mechanisms reduce long-term efficacy. Complex patient selection requires advanced diagnostics. Safety management demands experienced oncology teams.
America Cancer Immunotherapy Market Opportunities
Expansion Into Emerging Latin American Markets With Growing Oncology Infrastructure
Brazil and Mexico invest in modern cancer centers. Public health programs increase awareness and screening rates. Private hospitals adopt advanced biologic therapies. Regional regulatory reforms support faster drug approvals. Pharmaceutical firms explore local partnerships. Training initiatives enhance oncologist expertise. Cross-border collaborations improve clinical trial access. Expanding infrastructure creates new growth pathways.
Development Of Personalized And Precision-Based Immunotherapy Platforms For Niche Indications
Biotech firms design therapies for rare tumor subtypes. Genomic profiling enables targeted immune activation. Companion diagnostics refine patient selection criteria. Advanced manufacturing lowers production timelines. AI-driven drug discovery accelerates candidate identification. Academic partnerships foster translational research programs. Specialty clinics adopt personalized oncology services. Precision innovation positions the America Cancer Immunotherapy Market for sustained advancement.
America Cancer Immunotherapy Market Segmentation Analysis:
By Therapy Type
Monoclonal antibodies hold a strong position in the America Cancer Immunotherapy Market due to broad indications and well-established clinical outcomes. These agents target specific antigens on cancer cells and support durable responses across common tumor types. Their wide use across hospital networks strengthens adoption. Checkpoint inhibitors show rapid growth because they block immune-suppressive pathways and drive strong survival gains in lung cancer, melanoma, and renal cancers. Cell-based therapies deliver high response rates in blood cancers and require specialized centers for administration. Cancer vaccines move forward through growing clinical research focused on personalized immune activation. Immunomodulators and cytokines support immune signaling and complement combination regimens. Oncolytic virus therapies gain traction through their dual ability to lyse tumor cells and stimulate systemic immune responses.
By Cancer Type
Lung cancer drives high therapy demand due to significant disease prevalence and strong evidence supporting checkpoint inhibitor use across disease stages. Hematologic malignancies remain key for cell-based therapies and CAR-T adoption. Melanoma patients benefit from mature immunotherapy protocols with strong survival improvements. Breast cancer research expands the eligible patient pool for targeted immune strategies. Prostate and colorectal cancers integrate immunotherapy through biomarker-based pathways. Head and neck cancers show improved response rates with checkpoint inhibition. Ovarian and pancreatic cancers attract ongoing trials exploring novel immune targets. Other cancers continue to adopt immunotherapy through label extensions and research progress.
- For instance, CAR-T therapy Breyanzi from Bristol Myers Squibb delivered a 54% complete response rate in relapsed large B-cell lymphoma, highlighting strong outcomes in hematologic malignancies.
By End-User Segments
Hospitals and clinics remain the primary treatment locations in the America Cancer Immunotherapy Market due to their capability to deliver complex biologics and manage adverse events. Cancer research centers support trial enrollment and drive innovation across new therapy platforms. Hospital pharmacies manage procurement, storage, and dispensing of high-value immunotherapies and maintain key roles in treatment workflows.
- For instance, leading U.S. cancer centers administering CAR-T therapies such as Novartis’ Kymriah report treatment-related hospitalization rates above 20%, underscoring the need for specialized clinical infrastructure.
Segmentation:
By Therapy Type
- Monoclonal Antibodies
- Checkpoint Inhibitors
- Cell-Based Therapies
- Cancer Vaccines
- Immunomodulators / Cytokines
- Oncolytic Virus Therapies
By Cancer Type
- Lung Cancer
- Hematologic Malignancies
- Melanoma
- Breast Cancer
- Prostate Cancer
- Colorectal Cancer
- Head & Neck Cancer
- Ovarian Cancer
- Pancreatic Cancer
- Others
By End-User Segments
- Hospitals & Clinics
- Cancer Research Centers
- Hospital Pharmacies
Regional Analysis:
North America holds the dominant share of the America Cancer Immunotherapy Market, accounting for nearly 78% of total regional revenue. Strong healthcare infrastructure and high adoption of advanced biologics support its leadership. The United States drives most therapy demand through broad access to checkpoint inhibitors and cell-based treatments. Canada strengthens the regional base through national cancer programs and improved reimbursement structures. Research institutions across both countries expand clinical pipelines. The region maintains strong momentum due to sustained investment in oncology innovation and rising diagnosis rates.
Latin America captures roughly 17% of the market and shows steady improvement in immunotherapy uptake. Brazil leads due to expanding private healthcare networks and wider access to targeted therapies. Mexico follows through growing oncology centers and rising awareness of advanced cancer care options. Argentina and Chile increase participation in clinical trials and enhance treatment availability through public health initiatives. The Latin American segment of the America Cancer Immunotherapy Market gains traction through structured cancer policies and gradual regulatory modernization. It benefits from rising collaborations with global biopharmaceutical companies.
The remaining 5% share comes from emerging markets across Central America and the Caribbean. These regions expand capacity slowly due to economic constraints and limited specialty infrastructure. Immunotherapy access grows through selective hospital partnerships and government-led cancer care programs. Some countries improve diagnostic capabilities to support eligible patient identification. Training programs support clinician familiarity with new treatment pathways. Market penetration improves where health systems adopt progressive oncology reimbursement models. The region holds long-term potential with rising investment in tertiary care services.
Shape Your Report to Specific Countries or Regions & Enjoy 30% Off!
Key Player Analysis:
- Amgen
- Bristol-Myers Squibb
- Roche
- Merck & Co.
- AstraZeneca
- Novartis
- Pfizer
- Eli Lilly and Company
- Bayer
- Gilead Sciences
- GlaxoSmithKline
- Regeneron Pharmaceuticals
- Incyte
- BeiGene Ltd.
- Seagen
- Exelixis
- Arcus Biosciences
- Iovance Biotherapeutics
- Allogene Therapeutics
- Dragonfly Therapeutics
Competitive Analysis:
The America Cancer Immunotherapy Market features strong competition driven by innovation, regulatory approvals, and portfolio expansion. Global leaders such as Merck, Bristol Myers Squibb, Novartis, and Gilead strengthen their positions through broad immunotherapy portfolios covering monoclonal antibodies, checkpoint inhibitors, and cell-based therapies. Companies invest heavily in clinical trials to secure expanded indications across high-prevalence cancers. Strategic alliances with research centers support ongoing development of next-generation immune agents. Firms compete on efficacy profiles, safety outcomes, and real-world performance to capture larger patient segments. Biosimilars create new pricing dynamics across select monoclonal antibody categories. It maintains competitive pressure through rapid advancements in personalized immunotherapy platforms and the emergence of novel treatment modalities targeting niche tumor types.
Recent Developments:
- In January 2026, Sun Pharma launched Unloxcyt in the US, enabling full commercialization of this immunotherapy drug developed by Sun Pharma Advanced Research Company (SPARC) and manufactured at Sun Pharma’s US biologics facility to improve patient access in oncology settings.
- In June 2025, Bristol Myers Squibb (BMS) and BioNTech officially announced a global strategic partnership to co-develop and co-commercialise BNT327(also known as pumitamig), a next-generation bispecific antibody targeting PD-L1 and VEGF-A.
- In February 2025, AbbVie partnered with Xilio Therapeutics on a deal worth up to USD 2.1 billion to co-develop up to four masked T cell engager immunotherapy programs, with AbbVie securing global rights to one lead program plus options on others.
Report Coverage:
The research report offers an in-depth analysis based on Therapy Type, Cancer Type, and End-User Segments. It details leading market players, providing an overview of their business, product offerings, investments, revenue streams, and key applications. Additionally, the report includes insights into the competitive environment, SWOT analysis, current market trends, as well as the primary drivers and constraints. Furthermore, it discusses various factors that have driven market expansion in recent years. The report also explores market dynamics, regulatory scenarios, and technological advancements that are shaping the industry. It assesses the impact of external factors and global economic changes on market growth. Lastly, it provides strategic recommendations for new entrants and established companies to navigate the complexities of the market.
Future Outlook:
- The America Cancer Immunotherapy Market will move toward broader use of personalized treatment pathways supported by biomarker-driven selection.
- Expansion of approved indications across solid tumors will strengthen adoption across hospitals and specialty centers.
- Next-generation platforms such as bispecific antibodies and engineered T-cell therapies will reshape treatment strategies.
- Growth in clinical trial activity across North America and Latin America will expand patient access to emerging therapies.
- Integration of real-world evidence will guide payer decisions and shape long-term reimbursement frameworks.
- Digital health tools will support treatment monitoring and help clinicians track immune response patterns.
- The shift toward earlier-line use of immunotherapies will expand eligible patient groups and strengthen clinical outcomes.
- Latin America will see rising adoption as oncology infrastructure improves and specialist networks expand.
- Strategic partnerships between global biopharma firms and regional research institutes will accelerate pipeline progress.
- The market will benefit from rapid advancements in precision oncology, driving sustained innovation and long-term demand.




