Biobetters Market Overview:
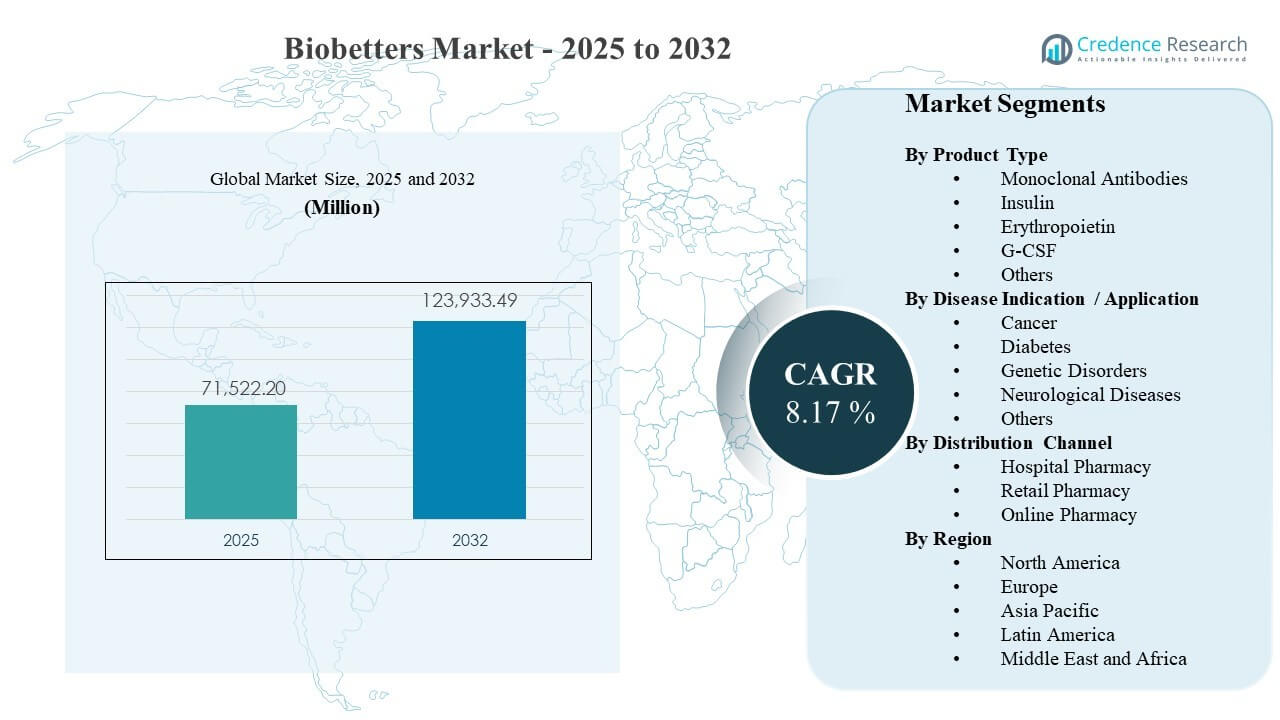
The global Biobetters Market size was estimated at USD 71522.2 million in 2025 and is expected to reach USD 123933.49 million by 2032, growing at a CAGR of 8.17% from 2025 to 2032. Growth is being reinforced by rising demand for clinically differentiated biologics that improve dosing convenience, durability of response, or safety profiles in chronic conditions. Biobetters Market adoption is also supported by expanding specialty care capacity and stronger biologics manufacturing scale that improves availability across major healthcare systems.
| REPORT ATTRIBUTE |
DETAILS |
| Historical Period |
2020-2024 |
| Base Year |
2025 |
| Forecast Period |
2026-2032 |
| Biobetters Market Size 2025 |
USD 71522.2 million |
| Biobetters Market, CAGR |
8.17% |
| Biobetters Market Size 2032 |
USD 123933.49 million |
Key Market Trends & Insights
- Biobetters Market is projected to expand at a CAGR of 8.17% (2025–2032), reflecting sustained demand for upgraded biologic formulations across chronic disease pathways.
- Insulin accounted for the largest share of 44.9% in 2025 within the By Product Type segmentation, supported by high treated prevalence and long-duration therapy.
- Diabetes accounted for the largest share of 49.3% in 2025 within the By Disease Indication / Application segmentation, driven by sustained therapy continuity and regimen optimization.
- Hospital Pharmacy accounted for the largest share of 52.6% in 2025 within the By Distribution Channel segmentation, supported by specialty handling and protocol-driven dispensing.
- North America accounted for 46.70% share in 2025, reflecting strong biologics reimbursement depth and mature specialty prescribing infrastructure.
Segment Analysis
Biobetters Market demand is closely tied to specialty prescribing and chronic therapy adherence, which makes differentiated biologics more attractive where dosing convenience and outcome stability influence long-term utilization. Biobetters Market purchasing decisions also reflect care-site readiness, including cold-chain logistics, trained staff, and monitoring protocols that support safe initiation. As a result, institutional channels typically anchor early adoption before broader diffusion into outpatient maintenance use.
Biobetters Market segmentation by therapy class and indication reflects where treated populations are large and where clinical differentiation is most valued. Diabetes-driven utilization remains structurally large because therapy duration is long and switching decisions are frequent when improved profiles reduce hypoglycemia risk or simplify administration. Oncology-focused growth remains supported by a steady cadence of advanced biologic innovation, where differentiated mechanisms and improved pharmacokinetics can create measurable clinical value.
Access crucial information at unmatched prices!
Request your sample report today & start making informed decisions powered by Credence Research Inc.!
Download Sample
By Product Type Insights
Insulin accounted for the largest share of 44.9% in 2025. Biobetters Market leadership in insulin is reinforced by large treated populations and long treatment duration in diabetes care pathways. Biobetters Market uptake is supported when upgraded insulin profiles improve dosing flexibility, reduce adverse events, and enhance patient adherence. Biobetters Market formulary decisions also favor products with clear real-world persistence benefits and reliable supply continuity.
By Disease Indication / Application Insights
Diabetes accounted for the largest share of 49.3% in 2025. Biobetters Market revenue concentration in diabetes is supported by continuous therapy needs and frequent regimen refinement across disease progression stages. Biobetters Market switching is more likely when differentiated biologics reduce complications and lower downstream healthcare utilization. Biobetters Market demand is further sustained by structured care pathways that prioritize measurable outcomes and long-term adherence.
By Distribution Channel Insights
Hospital Pharmacy accounted for the largest share of 52.6% in 2025. Biobetters Market channel dominance for hospital pharmacies is supported by specialty handling, cold-chain requirements, and protocol-led initiation in advanced care settings. Biobetters Market access is influenced by institutional purchasing, formulary governance, and multidisciplinary decision-making in specialty centers. Biobetters Market distribution also benefits from hospital-led patient education and monitoring that improves persistence after therapy start.
Biobetters Market Drivers
Rising chronic disease treatment intensity across specialty pathways
Biobetters Market expansion is supported by rising treatment intensity across chronic disease pathways where biologics represent a growing share of therapy value. Biobetters Market demand increases when clinicians prioritize improved dosing convenience, better tolerability, or more durable responses compared to earlier biologics. Biobetters Market adoption is reinforced by structured treatment guidelines that encourage optimization after suboptimal response. Biobetters Market growth is also supported by increasing specialist capacity and broader diagnostic access in major healthcare systems.
- For instance, in rheumatoid arthritis, the introduction of citrate‑free adalimumab at 40 mg in 0.4 mL (vs 0.8 mL previously) with a smaller 29‑gauge needle significantly reduced injection‑site pain scores on the FPS‑R VAS from a range of 3–10 to 0–3, with 100% of patients reporting only mild pain, supporting treatment intensification and persistence on biologic therapy.
Lifecycle optimization and differentiation in biologic product design
Biobetters Market growth is reinforced by lifecycle optimization strategies that create clinically differentiated versions of established biologics. Biobetters Market value increases when improved pharmacokinetics, delivery systems, or dosing intervals translate into better adherence and outcomes. Biobetters Market adoption improves when upgrades reduce administration burden for patients and care teams. Biobetters Market commercialization is strengthened when manufacturers align product differentiation with payer and provider value frameworks.
- For instance, AbbVie’s reformulated adalimumab introduced before Humira’s patent expiry increased concentration to 100 mg/mL, halving volume to 0.4 mL for a 40 mg dose and using a 29‑gauge needle to reduce injection‑site reactions relative to the original 0.8 mL, 27‑gauge presentation.
Scaling biologics manufacturing and supply resilience
Biobetters Market demand is supported by broader investments in biologics manufacturing capacity that reduce supply constraints and improve reliability. Biobetters Market uptake accelerates when expanded fill-finish and cold-chain networks improve availability across regions and care settings. Biobetters Market purchasing decisions favor consistent supply for chronic therapies that require uninterrupted continuity. Biobetters Market growth is further supported by process innovation and quality systems that enable scale while maintaining regulatory compliance.
Institutional adoption and specialty channel enablement
Biobetters Market growth is strengthened by institutional adoption mechanisms, including formulary approvals and protocol-based use in specialty centers. Biobetters Market traction improves when hospital systems standardize biologic pathways and expand infusion or specialty dispensing capabilities. Biobetters Market performance benefits from integrated care models that combine initiation, monitoring, and follow-up within coordinated networks. Biobetters Market expansion is also supported by specialty pharmacy services that improve adherence, access navigation, and patient education.
Biobetters Market Challenges
Biobetters Market growth can be constrained by complex reimbursement processes and payer management that increases administrative burden and slows switching decisions. Biobetters Market access may be limited when clinical differentiation is not clearly demonstrated in outcomes that matter to payers and providers. Biobetters Market pricing pressure can intensify in therapy areas with multiple competing biologics and aggressive contracting. Biobetters Market adoption may also face delays when healthcare systems prioritize cost containment over incremental clinical improvements.
- For instance, registry data from European rheumatology centers show high one‑year drug retention rates above 80% on established adalimumab biosimilars, which can make payers reluctant to fund switches to marginally differentiated biobetters without compelling outcomes or cost offsets.
Biobetters Market scaling faces operational challenges tied to manufacturing complexity, cold-chain logistics, and stringent quality requirements across global supply networks. Biobetters Market commercialization can be slowed by regulatory variation and country-specific evidence requirements for differentiated biologics. Biobetters Market penetration may be uneven in regions with limited specialty capacity or lower affordability for biologic therapies. Biobetters Market demand can also be affected by clinician inertia when switching from established regimens requires workflow changes and monitoring adjustments.
Biobetters Market Trends and Opportunities
Biobetters Market opportunity is expanding as healthcare systems increasingly value therapies that reduce administration burden and improve real-world persistence. Biobetters Market innovation is trending toward longer-acting formats, improved delivery devices, and engineered profiles that enhance durability of response. Biobetters Market adoption can accelerate when evidence generation expands beyond trials into real-world outcomes that support payer value assessment. Biobetters Market participants can also benefit from partnering models that strengthen specialty distribution and patient support services.
Biobetters Market growth potential is increasing in emerging markets as specialty care infrastructure expands and biologics access frameworks mature. Biobetters Market demand is supported by localized manufacturing and regional supply strategies that improve affordability and availability. Biobetters Market positioning is also evolving as providers seek standardized pathways and interoperable monitoring that reduces variation in specialty care delivery. Biobetters Market companies that align differentiation with measurable outcomes and reliable access can capture faster uptake.
- For instance, Samsung Bioepis reported US$280.3 million in revenue for Q1 2025 a 43% year-over-year increase driven by strong biosimilar and biobetter sales across Europe and the US, supported by 11 global regulatory approvals and a commercialization network spanning partnerships with Biogen, Organon, and Sandoz across more than 100 countries, illustrating how scale and structured access frameworks drive emerging-market penetration.
Regional Insights
North America
North America accounted for 46.70% share in 2025. Biobetters Market demand in North America is supported by mature specialty prescribing, higher biologics utilization, and strong reimbursement depth across major therapy areas. Biobetters Market adoption is reinforced by large integrated delivery networks and specialty pharmacy infrastructure that improves initiation and persistence. Biobetters Market competition is typically shaped by formulary contracting, evidence generation, and supply reliability across chronic therapy pathways.
Europe
Europe accounted for 26.40% share in 2025. Biobetters Market performance in Europe is supported by structured healthcare delivery and established biologics use in large national systems. Biobetters Market uptake is influenced by tendering processes, cost-effectiveness thresholds, and guideline-driven initiation in specialty centers. Biobetters Market momentum strengthens when differentiation is clear and when manufacturers align access strategy with country-level reimbursement frameworks.
Asia Pacific
Asia Pacific accounted for 19.10% share in 2025. Biobetters Market growth in Asia Pacific is supported by expanding specialty capacity, rising chronic disease burden, and increasing biologics manufacturing scale across key markets. Biobetters Market adoption remains uneven due to reimbursement variability and affordability constraints in certain countries. Biobetters Market participants benefit from localization, access partnerships, and distribution capabilities that extend specialty therapies beyond top-tier urban centers.
Latin America
Latin America accounted for 5.20% share in 2025. Biobetters Market uptake in Latin America is shaped by access concentration in larger markets and specialty centers with stronger procurement capability. Biobetters Market growth is supported when public and private payers expand coverage and improve continuity for chronic biologic therapy. Biobetters Market adoption can be constrained by budget pressure and logistical complexity in cold-chain distribution.
Middle East & Africa
Middle East & Africa accounted for 2.60% share in 2025. Biobetters Market demand in Middle East & Africa is supported by specialty center expansion and gradual improvements in biologics access frameworks. Biobetters Market penetration is typically higher in markets with stronger private coverage and tertiary hospital capacity. Biobetters Market growth can accelerate with investments in cold-chain logistics, specialist availability, and standardized treatment pathways.
Competitive Landscape
Biobetters Market competition is characterized by differentiation-led portfolios, lifecycle management strategies, and increasing focus on real-world evidence to support switching and reimbursement. Biobetters Market leaders prioritize scalable manufacturing, specialty distribution strength, and patient support capabilities that improve persistence in chronic therapy areas. Biobetters Market positioning also depends on formulary access, contracting depth, and the ability to demonstrate meaningful clinical and operational value for providers.
Novo Nordisk A/S is positioned as a key participant by combining large-scale biologics manufacturing capability with sustained investment in chronic disease innovation. Biobetters Market presence for Novo Nordisk A/S is supported by deep disease-area specialization and strong commercialization infrastructure across mature and growth markets. Biobetters Market strategy typically emphasizes supply reliability, differentiated product profiles, and patient-centered delivery approaches that support adherence and long-term outcomes. Biobetters Market momentum is reinforced when upgrades translate into measurable persistence benefits in real-world care.
The industry research and growth report includes detailed analyses of the competitive landscape of the market and information about key companies, including:
- Novo Nordisk A/S
- Eli Lilly and Company
- Amgen Inc.
- Merck & Co., Inc.
- F. Hoffmann-La Roche AG
- Sanofi SA
- Biogen Inc.
- Teva Pharmaceutical Industries Ltd.
- CSL Behring GmbH
- Celltrion Healthcare Co., Ltd.
Qualitative and quantitative analysis of companies has been conducted to help clients understand the wider business environment as well as the strengths and weaknesses of key industry players. Data is qualitatively analyzed to categorize companies as pure play, category-focused, industry-focused, and diversified; it is quantitatively analyzed to categorize companies as dominant, leading, strong, tentative, and weak.
Shape Your Report to Specific Countries or Regions & Enjoy 30% Off!
Recent Developments
- In February 2025, BioNTech company completed its acquisition of Biotheus to strengthen its oncology pipeline and secure full global rights to BNT327, while also adding Biotheus’ antibody platform, bispecific ADC capability, and an advanced biologics manufacturing facility to its network.
- In June 2025, BioNTech and Bristol Myers Squibb, the companies announced a global strategic partnership to co-develop and co-commercialize BNT327, an investigational bispecific antibody for multiple solid tumor types, with both companies jointly advancing the candidate in monotherapy and combination settings.
- In February 2026, Novo Nordisk entered into a landmark partnership agreement with Vivtex Corporation to develop next-generation oral biologic medicines for obesity, diabetes, and associated comorbidities. Under the terms of the deal, Vivtex will license select oral drug-delivery technologies to Novo Nordisk, with the collaboration potentially valued at up to $2.1 billion in upfront payments, research funding, and milestone payments, along with tiered royalties on future product sales.
Report Scope
| Report Attribute |
Details |
| Market size value in 2025 |
USD 71522.2 million |
| Revenue forecast in 2032 |
USD 123933.49 million |
| Growth rate (CAGR) |
8.17% (2025–2032) |
| Base year |
2025 |
| Forecast period |
2026-2032 |
| Quantitative units |
USD million |
| Segments covered |
By Product Type; By Disease Indication / Application; By Distribution Channel |
| Regional scope |
North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
| Key companies profiled |
Novo Nordisk A/S; Eli Lilly and Company; Amgen Inc.; Merck & Co., Inc.; F. Hoffmann-La Roche AG; Sanofi SA; Biogen Inc.; Teva Pharmaceutical Industries Ltd.; CSL Behring GmbH; Celltrion Healthcare Co., Ltd. |
| No.of Pages |
330 |
Segmentation
By Product Type
- Monoclonal Antibodies
- Insulin
- Erythropoietin
- G-CSF
- Others
By Disease Indication / Application
- Cancer
- Diabetes
- Genetic Disorders
- Neurological Diseases
- Others
By Distribution Channel
- Hospital Pharmacy
- Retail Pharmacy
- Online Pharmacy
By Region
- North America
- Europe
- Germany
- France
- U.K.
- Italy
- Spain
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- South-east Asia
- Rest of Asia Pacific
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Middle East & Africa
- GCC Countries
- South Africa
- Rest of the Middle East and Africa




