Abdominal Aortic Aneurysm Repair Devices Market Overview:
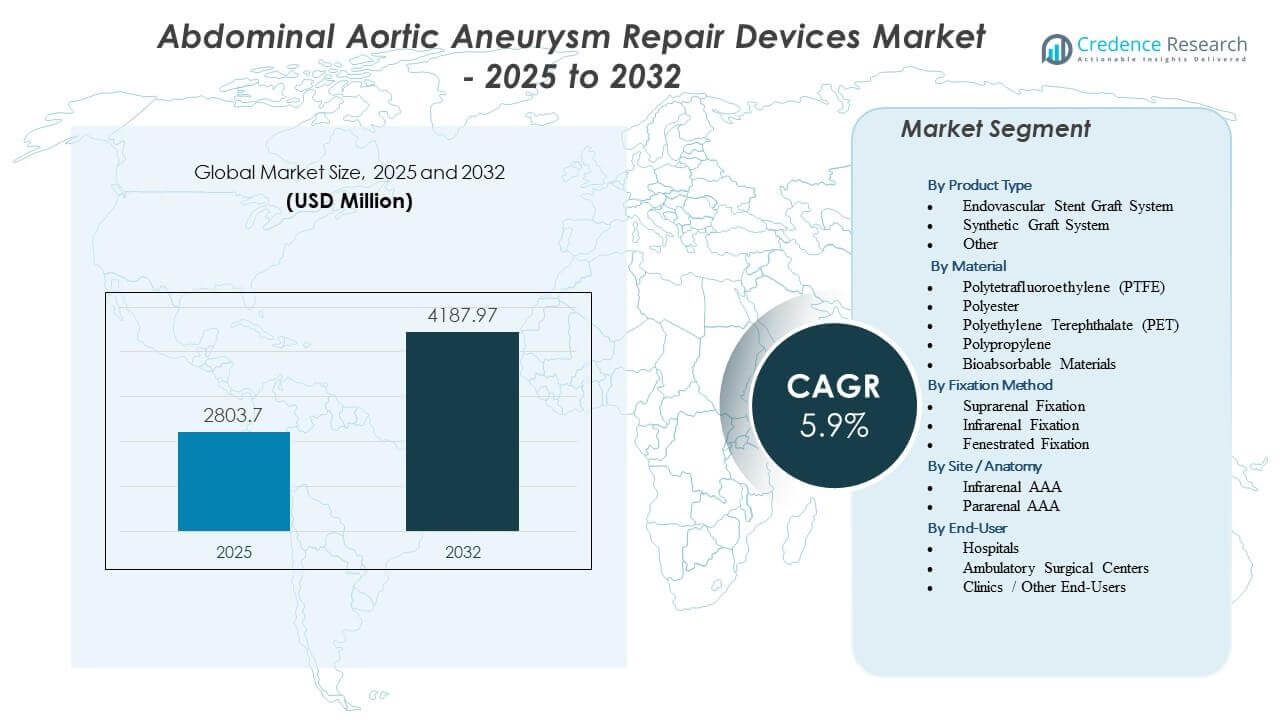
The Abdominal Aortic Aneurysm Repair Devices Market is projected to grow from USD 2803.7 million in 2025 to an estimated USD 4187.97 million by 2032, with a compound annual growth rate (CAGR) of 5.9% from 2025 to 2032.
| RT ATTRIBUTE |
DETAILS |
| Historical Period |
2020-2023 |
| Base Year |
2024 |
| Forecast Period |
2025-2032 |
| Abdominal Aortic Aneurysm Repair Devices Market Size 2025 |
USD 2803.7 million |
| Abdominal Aortic Aneurysm Repair Devices Market, CAGR |
5.9% |
| Abdominal Aortic Aneurysm Repair Devices Market Size 2032 |
USD 4187.97 million |
Abdominal Aortic Aneurysm Repair Devices Market Insights:
- Rising adoption of EVAR, improved stent graft designs, and increased screening in aging populations strengthen market momentum.
- Market restraints include high device costs, limited access in low-resource settings, and the need for specialized surgical expertise.
- North America leads due to advanced facilities and high treatment rates, while Europe maintains strong uptake supported by clinical guidelines.
- Asia Pacific emerges as the fastest-growing region driven by expanding healthcare infrastructure and rising awareness of aneurysm risks.
Abdominal Aortic Aneurysm Repair Devices Market Drivers
Rising Prevalence of Aneurysms and Growing Adoption of Minimally Invasive Repair Techniques
The growing burden of abdominal aortic aneurysms strengthens demand for advanced repair devices. Many patients seek early intervention due to rising awareness of rupture risks. Surgeons prefer minimally invasive repair solutions because they shorten recovery time and reduce complications. Hospitals expand EVAR capability to treat high-risk patients with better procedural outcomes. Improved screening programs detect aneurysms earlier in older populations. The trend encourages device makers to introduce more durable grafts. Regulatory bodies support safer devices with enhanced reliability. The Abdominal Aortic Aneurysm Repair Devices Market benefits from wider clinical acceptance of minimally invasive repair methods.
Access crucial information at unmatched prices!
Request your sample report today & start making informed decisions powered by Credence Research Inc.!
Download Sample
Technological Innovations in Stent Grafts and Imaging Systems Enhancing Procedural Accuracy
New stent graft materials improve flexibility and durability during complex repairs. Physicians rely on advanced imaging tools to achieve precise placement during EVAR. These tools support higher success rates in anatomically difficult cases. Device makers design grafts that fit more patient anatomies with improved sealing zones. Hospitals invest in hybrid operating rooms to support image-guided interventions. Patients gain faster mobility due to refined device engineering. Technological progress boosts surgeon confidence in performing high-risk repairs. This trend strengthens device demand across global vascular centers.
- For instance, W. L. Gore & Associates launched the GORE EXCLUDER Conformable AAA Device featuring an ACTIVE CONTROL System, which allows surgeons to achieve 100% deployment control and adjust the graft’s position even after it has been partially unsheathed.
Growing Geriatric Population and Rising Risk Factors Such as Smoking and Hypertension
Older populations experience higher aneurysm incidence due to weakened vascular structures. Rising hypertension increases vulnerability among susceptible groups. Many countries report higher smoking rates in aging males, accelerating aneurysm development. These factors push demand for early diagnosis and timely surgical repair. Health systems expand vascular care services to manage high-risk patients. More individuals qualify for EVAR because device designs now support broader anatomies. Public-health campaigns highlight aneurysm screening importance. The Abdominal Aortic Aneurysm Repair Devices Market gains steady momentum from these demographic shifts.
Increasing Investments Toward Vascular Care Infrastructure and Specialized Treatment Centers
Governments expand funding for vascular surgery units to support advanced aneurysm repair. Hospitals adopt EVAR-focused programs to manage rising patient volumes. Training initiatives enhance surgeon capability and improve outcomes. Vendors launch educational programs to guide surgeons on new device systems. Reimbursement support in many regions encourages wider treatment access. Growth in multispecialty hospitals pushes adoption of complex aneurysm repair solutions. Health systems focus on lowering mortality linked to late-stage aneurysm presentation. Expanded infrastructure strengthens adoption of EVAR technology worldwide.
- For instance, Terumo Aortic has scaled its specialized manufacturing for the Fenestrated Anaconda device, a custom-built solution that enables the placement of up to 4 fenestrations to preserve blood flow to the renal arteries in complex cases with highly angulated necks.
Abdominal Aortic Aneurysm Repair Devices Market Trends
Shift Toward Next-Generation Fenestrated and Branched Stent Grafts for Complex Aneurysm Cases
Surgeons increasingly select fenestrated devices to treat anatomies unsuitable for standard EVAR. These grafts support precise alignment with renal and iliac vessels. Manufacturers design customizable options to match diverse patient structures. Increased use in tertiary hospitals expands procedural capability. The trend enhances outcomes for patients with short neck or tortuous aortic anatomy. Adoption grows due to successful long-term durability data. Hybrid centers support faster adaptation of complex repair techniques. The Abdominal Aortic Aneurysm Repair Devices Market observes rising demand for flexible graft designs.
Integration of AI-Driven Planning Tools and Advanced Navigation Systems for Precision Repair
AI tools help surgeons predict complications and plan graft placement accurately. These systems support detailed visualization before and during EVAR. Hospitals integrate real-time navigation platforms for better control. Vendors introduce software that simulates deployment across varied anatomies. Surgeons rely on predictive analytics to minimize endoleak risks. AI use expands across high-volume vascular centers. The trend supports more personalized aneurysm treatment planning. It improves confidence among less experienced operators.
- For instance, GE HealthCare’s EVAR Assist imaging suite, integrated within the IGS platform, is FDA-cleared and has demonstrated measurable contrast and radiation reduction in EVAR planning and guidance across published clinical evaluations.
Growing Preference for Outpatient EVAR Procedures Supported by Reduced Hospital Stay
Improved device designs lower post-procedure complications. Many institutions shift eligible patients toward outpatient EVAR. Faster discharge reduces healthcare cost burdens and improves patient experience. Better sealing technology lowers readmission risk. Surgeons rely on simplified graft deployment systems to shorten operation time. Hospitals adopt fast-track recovery protocols with structured monitoring. The trend grows in regions with strong reimbursement support. It promotes wider adoption of minimally invasive repair approaches.
- For instance, studies on the Gore Excluder AAA Endoprosthesis report median discharge within 24 hours for selected low-risk patients, supporting broader outpatient EVAR adoption in vascular programs.
Rising Focus on Post-Market Surveillance and Long-Term Performance Tracking for Device Reliability
Regulators push firms to present more long-term safety data. Hospitals track EVAR outcomes to ensure graft durability. Real-world evidence helps refine next-generation graft engineering. Vendors expand follow-up programs to detect endoleaks earlier. The trend improves transparency in device performance. Data-driven assessment supports better procurement decisions. Patients gain confidence in long-term device safety. The Abdominal Aortic Aneurysm Repair Devices Market benefits from structured performance monitoring guidelines.
Market Challenges Analysis
Complex Anatomical Limitations, Procedural Risks, and High Reintervention Rates in Specific Patient Groups
Many patients present anatomies that fall outside standard EVAR instructions-for-use, limiting repair choices. Complex aneurysm shapes raise risks during graft placement. Surgeons face complications linked to sealing zones and migration. High-risk groups often require secondary procedures due to endoleaks. Hospitals must invest in skilled teams to manage such cases effectively. Device makers struggle to design universal grafts for wide anatomical variation. Reintervention risk increases healthcare costs and burdens treatment centers. The Abdominal Aortic Aneurysm Repair Devices Market faces constraints from these anatomical challenges.
High Device Costs, Limited Access in Low-Resource Regions, and Reimbursement Barriers
Many countries experience financial constraints that limit access to advanced grafts. High device pricing discourages adoption in rural and low-income settings. Some insurance systems provide partial reimbursement, raising patient out-of-pocket burden. Hospitals hesitate to upgrade technology due to capital limitations. Limited availability of skilled vascular surgeons restricts procedure growth. Training programs remain concentrated in urban centers. Market expansion slows in regions without strong vascular care infrastructure. Cost barriers continue to influence global adoption rates.
Abdominal Aortic Aneurysm Repair Devices Market Opportunities
Expanding Demand for Personalized Graft Designs and Next-Generation Minimally Invasive Repair Options
Customized grafts can address anatomies that currently fall outside standard EVAR criteria. Manufacturers explore modular systems that adapt to patient-specific needs. Hospitals show rising interest in devices suited for complex aortic repairs. Improved imaging allows better matching of grafts to individual structures. New EVAR accessories support higher accuracy during deployment. Growth in hybrid operating rooms enhances adoption of advanced repair tools. Surgeons view personalized repair techniques as a way to improve outcomes. The Abdominal Aortic Aneurysm Repair Devices Market gains momentum from this personalization trend.
Opportunities in Emerging Markets Through Infrastructure Growth and Wider Screening Programs
Developing countries invest in vascular centers to manage rising aneurysm incidence. Public screening identifies more patients earlier, increasing treatment volumes. Governments upgrade hospitals to support image-guided EVAR. Vendors expand training programs to support regional surgeon capability. Local distributors help extend device access across rural areas. Reimbursement reforms support adoption of minimally invasive repair options. Market players see strong growth potential in regions with aging populations. Broader awareness encourages earlier intervention and higher procedural demand.
Abdominal Aortic Aneurysm Repair Devices Market Segmentation Analysis:
By Product Type
Endovascular stent graft systems dominate due to strong adoption of minimally invasive EVAR procedures. These systems offer shorter recovery time and lower complication rates, which strengthens demand across high-volume vascular centers. Surgeons prefer these grafts for diverse anatomies and improved long-term durability. Synthetic graft systems hold steady use in open surgical repair where full arterial reconstruction is required. Polyester and PTFE-based grafts support predictable handling during OSR. Other products, including delivery systems and ancillary tools, provide support for accurate placement and procedural safety. Hospitals upgrade inventories to include advanced graft deployment tools. The Abdominal Aortic Aneurysm Repair Devices Market sees strong product innovation focused on precise sealing and migration resistance.
- For instance, the Medtronic Endurant™ Stent Graft System demonstrated a 98.6% freedom from aneurysm-related mortality at 5 years in the ENGAGE Registry, confirming strong long-term performance across more than 1,200 real-world EVAR patients.
By Material
PTFE remains widely used due to excellent biocompatibility and reduced thrombosis risk. Polyester maintains demand in open repair due to strong tensile strength and ease of suturing. PET grafts provide durability and maintain lumen shape across long-term follow-up. Polypropylene finds limited yet targeted use where flexibility is required in graft components. Bioabsorbable materials gain attention for future devices targeting improved endothelial integration. Manufacturers evaluate hybrid material blends to improve sealing capability. Hospitals seek materials that reduce reintervention rates in complex anatomies. The Abdominal Aortic Aneurysm Repair Devices Market benefits from advancements that enhance graft longevity.
By Fixation Method
Suprarenal fixation leads due to strong anchoring capability above the renal arteries, improving stability in short or angulated necks. Surgeons rely on it to prevent graft migration in challenging anatomies. Infrarenal fixation maintains use where patients present adequate neck length and low angulation. These grafts support straightforward placement and low renal interaction risk. Fenestrated fixation grows rapidly because it addresses cases that fall outside standard IFU ranges. These devices align with critical branch vessels for precise sealing. Hospitals increase procedural capacity for fenestrated EVAR as training improves. The Abdominal Aortic Aneurysm Repair Devices Market gains traction from expanding fixation solutions for complex repairs.
- For instance, suprarenal fixation in the Endurant™ system demonstrated a graft migration rate below 1% at 5 years in the ENGAGE Registry, reinforcing its stability in hostile neck anatomies.
By Site / Anatomy
Infrarenal AAA cases dominate due to high prevalence across aging populations. EVAR adoption remains high here because most patients qualify for standard grafts. Hospitals streamline workflows to handle large volumes of infrarenal repairs with predictable outcomes. Pararenal AAA cases require advanced grafts, including fenestrated and branched systems. Surgeons treat these patients in specialized centers equipped for complex procedures. The complexity drives demand for customizable graft designs. Treatment decisions rely heavily on anatomical imaging to select correct fixations. The Abdominal Aortic Aneurysm Repair Devices Market expands through improved solutions for anatomically difficult presentations.
By End-User
Hospitals remain the primary end-users due to availability of vascular surgeons, hybrid operating rooms, and advanced imaging. Large centers handle high procedural volumes and support both EVAR and OSR. Ambulatory surgical centers increase use of EVAR for eligible patients due to faster discharge pathways. These centers focus on cost-efficient care supported by streamlined device inventories. Clinics and other end-users contribute through screening, referral, and long-term patient follow-up. They support early detection, which improves intervention timing. Growing patient awareness boosts demand for specialized vascular evaluation. The Abdominal Aortic Aneurysm Repair Devices Market benefits from expanded care delivery across multiple facility types.
Segmentation:
By Product Type
- Endovascular Stent Graft System
- Synthetic Graft System
- Other
By Material
- Polytetrafluoroethylene (PTFE)
- Polyester
- Polyethylene Terephthalate (PET)
- Polypropylene
- Bioabsorbable Materials
By Fixation Method
- Suprarenal Fixation
- Infrarenal Fixation
- Fenestrated Fixation
By Site / Anatomy
- Infrarenal AAA
- Pararenal AAA
By End-User
- Hospitals
- Ambulatory Surgical Centers
- Clinics / Other End-Users
By Region
- North America
- Europe
- Germany
- France
- U.K.
- Italy
- Spain
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- South-east Asia
- Rest of Asia Pacific
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Middle East & Africa
- GCC Countries
- South Africa
- Rest of the Middle East and Africa
Regional Analysis:
North America holds the largest share of the global market with around 38%, supported by strong EVAR adoption, advanced vascular care infrastructure, and high diagnosis rates. Large hospital networks perform high procedural volumes, which strengthens demand for next-generation grafts. Leading manufacturers maintain strong distribution footprints across the United States and Canada. The region benefits from wide availability of hybrid operating rooms and image-guided systems. Regulatory approvals move steadily, enabling faster integration of device innovation. The Abdominal Aortic Aneurysm Repair Devices Market remains well established in North America due to strong patient awareness and high screening penetration.
Europe follows with approximately 30% market share driven by favorable clinical guidelines and widespread acceptance of minimally invasive aneurysm repair. Surgeons across Germany, the UK, France, and the Nordics adopt advanced fenestrated and branched grafts for complex cases. Most countries maintain structured screening programs targeting older populations. Hospitals invest in EVAR-focused training centers that support procedural efficiency. Market players collaborate with regional societies to strengthen device evaluation. Europe maintains steady growth as healthcare systems prioritize early intervention.
Asia Pacific emerges as the fastest-growing region with nearly 22% share, driven by rising healthcare investments and a rapidly expanding elderly population. China, Japan, India, and South Korea increase adoption of modern stent graft systems as imaging access improves. Hospitals upgrade vascular surgery departments to manage higher aneurysm detection rates. Emerging economies expand reimbursement pathways that support wider EVAR uptake. Manufacturers focus on local partnerships to improve product reach. The Abdominal Aortic Aneurysm Repair Devices Market gains momentum across Asia Pacific with growing clinical expertise and broader patient eligibility.
Shape Your Report to Specific Countries or Regions & Enjoy 30% Off!
Key Player Analysis:
- Medtronic plc
- Cook Medical Inc.
- L. Gore & Associates, Inc.
- Terumo Corporation
- MicroPort Scientific Corporation
- Artivion Inc.
- Lombard Medical
- Cardinal Health, Inc.
- Endologix LLC
- Boston Scientific Corporation
Competitive Analysis:
The Abdominal Aortic Aneurysm Repair Devices Market features strong competition among global medical device companies focused on improving graft flexibility, sealing performance, and long-term durability. Leading players include Medtronic, Cook Medical, W. L. Gore & Associates, Terumo, Endologix, and Cardinal Health. Each competitor expands its product portfolio with advanced stent graft designs suited for standard and complex anatomies. Companies invest in clinical registries to validate durability, reduce migration risk, and improve branch vessel integration. Hybrid operating room technologies strengthen vendor differentiation as hospitals seek seamless image-guided deployment. Competitive strategies center on physician training, expanded IFU approvals, and long-term surveillance programs. Firms work to reduce reintervention rates while improving ease of use to support broader EVAR adoption. The market moves toward graft systems with advanced fixation and enhanced sealing properties that support safer and more predictable outcomes.
Recent Developments:
- In May 2024, Endologix LLC announced clinical progress of its ChEVAS (Chimney Endovascular Aneurysm Sealing) system after new study results showed improved sealing performance in challenging anatomies. The development aligned with Endologix’s strategy to address patients who fall outside conventional EVAR treatment ranges.
- In February 2024, W. L. Gore & Associates reported new clinical milestones for the GORE® EXCLUDER® Conformable AAA Endoprosthesis with ACTIVE CONTROL System after successful U.S. commercial rollout. The company highlighted broader physician adoption driven by improved repositionability and enhanced sealing outcomes in hostile neck anatomies.
- In January 2024, Medtronic announced the continued expansion of its Endurant™ II/IIs stent graft system clinical data through new long-term outcomes published from the ENGAGE Registry. The update reinforced the device’s strong durability profile in both standard and complex AAA anatomies, supporting broader clinical confidence in EVAR adoption across major vascular centers.
Report Coverage:
The research report offers an in-depth analysis based on Product Type, Material, Fixation Method, Site / Anatomy, and End-User. It details leading market players, providing an overview of their business, product offerings, investments, revenue streams, and key applications. Additionally, the report includes insights into the competitive environment, SWOT analysis, current market trends, as well as the primary drivers and constraints. Furthermore, it discusses various factors that have driven market expansion in recent years. The report also explores market dynamics, regulatory scenarios, and technological advancements that are shaping the industry. It assesses the impact of external factors and global economic changes on market growth. Lastly, it provides strategic recommendations for new entrants and established companies to navigate the complexities of the market.
Future Outlook:
- The Abdominal Aortic Aneurysm Repair Devices Market will advance with broader EVAR adoption supported by improved sealing technologies and greater anatomical compatibility.
- Growth will expand as hospitals invest in hybrid operating rooms that enhance accuracy during complex aneurysm repair.
- Device manufacturers will focus on expanding indications to treat patients who fall outside standard eligibility profiles.
- AI-driven planning tools and imaging integration will support more precise graft placement across varied anatomies.
- Minimally invasive repair options will strengthen demand in regions with rising screening participation.
- New graft materials will target improved flexibility, durability, and safer long-term outcomes.
- Companies will increase investment in post-market surveillance programs to support long-term data transparency.
- Outpatient EVAR pathways will gain momentum as clinical outcomes continue to favor early discharge.
- Training programs will expand to support surgeon proficiency in fenestrated and branched graft systems.
- Strategic partnerships will rise as firms work to integrate imaging, navigation, and deployment innovations into unified platforms.




