Biobank Equipment Market Overview:
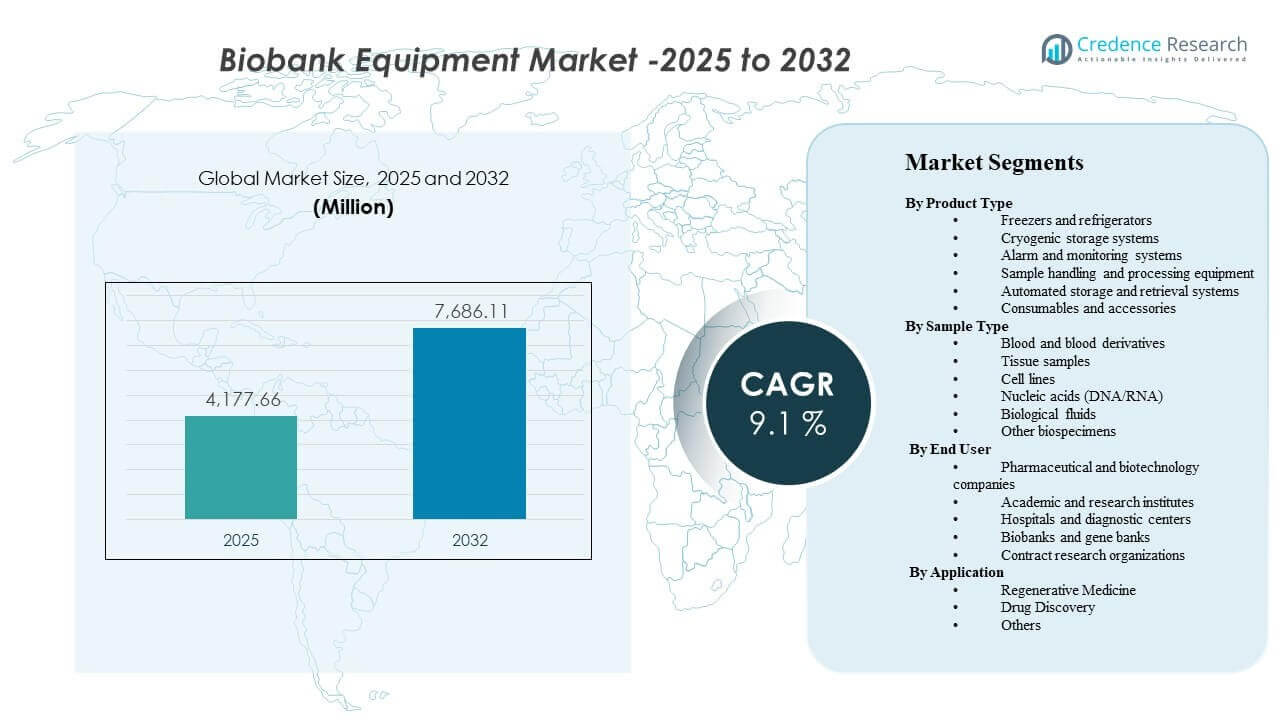
The global Biobank Equipment Market size was estimated at USD 4,177.66 million in 2025 and is expected to reach USD 7,686.11 million by 2032, growing at a CAGR of 9.1% from 2025 to 2032. Biobank Equipment Market growth is primarily supported by sustained expansion in biospecimen collection and long-term preservation needs across clinical research, genomics, and translational programs that require reliable ultra-low temperature performance and validated monitoring. Biobank Equipment Market demand is also reinforced by increased automation adoption in high-throughput repositories to improve chain-of-custody integrity, reduce manual handling risk, and standardize sample quality across multi-site studies.
| REPORT ATTRIBUTE |
DETAILS |
| Historical Period |
2020-2024 |
| Base Year |
2025 |
| Forecast Period |
2026-2032 |
| Biobank Equipment Market Size 2025 |
USD 4,177.66 million |
| Biobank Equipment Market, CAGR |
9.1% |
| Biobank Equipment Market Size 2032 |
USD 7,686.11 million |
Key Market Trends & Insights
- The Biobank Equipment Market is projected to expand from USD 4,177.66 million in 2025 to USD 7,686.11 million by 2032, reflecting a 9.1% CAGR over 2025–2032.
- Cryogenic storage systems accounted for the largest product share of 51.4% in 2025, reflecting sustained demand for long-term, ultra-stable preservation conditions.
- Regenerative Medicine accounted for 35.7% share in 2025, highlighting strong pull from cell and gene therapy workflows requiring controlled preservation and traceability.
- Biobanks and biorepositories represented 43.5% of end-user demand in 2025, supported by centralized repository expansion and scale-driven investments in storage capacity and automation.
- North America captured 36.6% of the Biobank Equipment Market in 2025, reflecting advanced research infrastructure and higher penetration of monitored cold storage systems.
Segment Analysis
Biobank Equipment Market demand is closely tied to the scale-up of biospecimen-driven research and the increasing operational complexity of biorepositories. Large repositories are prioritizing temperature stability, audit readiness, and operational resilience, which is accelerating adoption of integrated storage platforms that combine hardware performance with monitoring, access control, and documentation workflows. Automation is increasingly used to support higher sample volumes, reduce handling variability, and maintain chain-of-custody integrity across multi-site studies and longitudinal cohorts.
Biobank Equipment Market purchasing decisions are also shaped by the biospecimen mix and downstream use cases. Blood-related samples and biological fluids typically require high-throughput intake, aliquoting, and reliable cold storage cycles, whereas tissue samples and cell lines demand stricter contamination control and traceability. Genomics and multi-omics programs increase the need for standardized nucleic acid handling and stable long-term preservation environments. These dynamics collectively raise demand for robust equipment portfolios spanning storage, monitoring, and sample processing.
Access crucial information at unmatched prices!
Request your sample report today & start making informed decisions powered by Credence Research Inc.!
Download Sample
By Product Type Insights
Cryogenic storage systems accounted for the largest share of 51.4% in 2025. Cryogenic storage systems lead because cryogenic environments support long-term preservation and sample integrity requirements across high-value biospecimens used in advanced research. Cryogenic storage systems also align with repository-scale capacity needs and standardized archiving protocols that require stable ultra-low temperature performance. Cryogenic storage systems benefit from increased emphasis on operational continuity, where redundancy, monitoring, and alarm integration reduce risk of sample loss.
By Sample Type Insights
Blood and blood derivatives represent a central sample category for biobanking workflows due to broad use in clinical research, biomarker studies, and population health programs. Blood and blood derivatives commonly drive high-frequency intake and aliquoting needs, which increases demand for consistent cold-chain performance and standardized handling. Tissue samples and cell lines often require stricter chain-of-custody, contamination control, and multi-format storage, which reinforces investments in controlled processing and reliable long-term storage systems. Nucleic acids (DNA/RNA) support genomics and multi-omics programs that require repeatable preparation and stable preservation conditions to protect data quality.
By End User Insights
Biobanks and biorepositories accounted for the largest share of 43.5% in 2025. Biobanks and biorepositories lead because centralized repositories concentrate large sample volumes and typically require scalable storage capacity with robust monitoring and access controls. Biobanks and biorepositories also prioritize standardization to support multi-site studies, which drives procurement of integrated systems that improve traceability and reduce operational variability. Biobanks and biorepositories increasingly adopt automation to reduce manual handling burden and improve throughput without compromising sample integrity.
By Application Insights
Regenerative Medicine accounted for the largest share of 35.7% in 2025. Regenerative Medicine leads because cell and gene therapy pipelines require strict preservation conditions and traceability practices that protect viability and identity of advanced therapy materials. Regenerative Medicine programs often rely on long-term storage of cells, tissues, and intermediates under controlled conditions, increasing the need for reliable cryogenic and ultra-low temperature systems. Regenerative Medicine also strengthens demand for monitoring and alarm systems to maintain compliance and reduce the risk of temperature excursions across critical inventories.
Biobank Equipment Market Drivers
Expansion of biospecimen-driven research and biorepository scaling
Biobank Equipment Market growth is driven by expanding use of biospecimens in clinical research, genomics, and translational studies that require robust storage and documentation practices. Higher sample volumes increase pressure on repositories to expand capacity without increasing risk exposure. Equipment procurement increasingly prioritizes temperature stability, redundancy, and reliable monitoring to protect long-term sample value. Repository scale also supports greater investment in automation to improve throughput and reduce manual handling variability.
- For instance, Hamilton states that its BiOS automated -80°C platform can scale from 100,000 to 23 million samples, and that its battery-backed LN2 supply can maintain a -80°C storage temperature for 48 hours during a power outage, showing how large repositories are investing in both high-capacity storage and redundancy-led sample protection.
Increased demand from regenerative medicine and advanced therapy workflows
Biobank Equipment Market demand benefits from increased activity in cell and gene therapy programs that require strict preservation conditions and traceability. Advanced therapy materials often require controlled environments that protect viability and maintain identity throughout storage and movement. These requirements increase adoption of cryogenic systems, reliable monitoring, and controlled access workflows. The resulting quality and compliance expectations accelerate equipment upgrades and standardization across repositories.
- For instance, Azenta says its LN2-based CryoArc automated cryogenic systems operate at -190°C, with storage capacities of up to 8,800 2 mL vials in CryoArc Pico, 26,600 2 mL vials in CryoArc Deca, and 990 250 mL cryo cassettes in the cassette-configured CryoArc Tera, reflecting the strict viability and traceability needs of advanced therapy storage workflows.
Modernization of laboratory infrastructure and compliance expectations
Biobank Equipment Market purchasing is reinforced by modernization of laboratory infrastructure, particularly where repositories must meet quality management and audit readiness requirements. Temperature integrity, remote monitoring, and alarm validation are prioritized to reduce operational risk. Standardized processes for sample intake, labeling, and handling are increasingly embedded into equipment selection criteria. Modernization also supports replacement cycles where older storage units are upgraded for efficiency and reliability.
Growth of automation for productivity and risk reduction
Biobank Equipment Market adoption is increasingly supported by automation as repositories expand and staffing constraints intensify. Automated storage and retrieval systems reduce manual interaction with samples, lowering contamination risk and handling errors. Automation supports higher-density storage and improved traceability through integrated tracking and controlled access. These benefits align with repository objectives to improve operational resilience and consistency across large-scale studies.
Biobank Equipment Market Challenges
Biobank Equipment Market growth faces constraints from high upfront capital requirements and ongoing operating costs, particularly for ultra-low temperature and cryogenic systems. Cost sensitivity can slow adoption among smaller repositories and emerging-market institutions, where budgets prioritize essential clinical equipment over advanced storage upgrades. Procurement decisions also require careful evaluation of total cost of ownership, including energy use, maintenance, and service coverage. These cost and planning factors can extend purchasing cycles and delay modernization programs.
- For instance, PHCbi’s MDF-DU502VH VIP ECO is a 500 litre, -86°C ultra-low freezer rated at 6.70 kWh/day, and it combines inverter compressors, hydrocarbon refrigerants, VIP PLUS vacuum insulation, and logging of freezer temperature, door-opening times, and alarm history, illustrating why buyers closely assess power consumption, monitoring features, and lifecycle service requirements before approving storage upgrades.
Biobank Equipment Market adoption also encounters operational and integration challenges related to facility readiness and workflow standardization. Installation may require facility upgrades for space planning, power infrastructure, backup systems, and environmental controls. Integration with sample tracking, laboratory information systems, and monitoring platforms can be complex across multi-vendor environments. Inconsistent protocols across institutions can further limit seamless scaling and reduce the benefits of standardization and automation.
Biobank Equipment Market Trends and Opportunities
Biobank Equipment Market trends increasingly emphasize energy efficiency, temperature uniformity, and smarter monitoring capabilities. Buyers increasingly prioritize remote alarms, audit-ready reporting, and access control to strengthen compliance and reduce excursion risks. Repositories also favor systems that support better operational continuity through redundancy and proactive maintenance features. These preferences create opportunities for suppliers that can combine reliable storage performance with integrated monitoring and workflow tools.
- For instance, Thermo Fisher Scientific’s TSX Universal ultra-low temperature freezers specify less than 4°C temperature variation, 15-minute door recovery, 30-second re-entry after door opening, and 33% lower energy use than previous models, while also offering a 7-inch user interface, optional HID access, built-in monitoring outputs, and factory calibration and QC documentation that align with compliance-focused biobank operations.
Biobank Equipment Market opportunities are expanding in automation-enabled repositories and standardized sample processing ecosystems. Automated storage and retrieval platforms are being adopted to support higher sample volumes and reduce manual variability, particularly in centralized biorepositories and multi-site networks. Sample handling and processing equipment that improves repeatability and throughput is increasingly bundled into broader workflow upgrades. These shifts create openings for integrated solutions that span intake, processing, storage, and retrieval.
Regional Insights
North America
North America represented 36.6% of the Biobank Equipment Market in 2025. North America demand is supported by mature biobank networks, strong clinical research activity, and higher adoption of monitored cold storage and automated repository workflows. Investment priorities emphasize operational resilience, traceability, and compliance-ready storage environments. Replacement cycles also support ongoing demand for modernized ultra-low temperature systems and monitoring upgrades.
Europe
Europe accounted for 25.1% of the Biobank Equipment Market in 2025. Europe demand is supported by established biomedical research ecosystems and structured biorepository programs that prioritize standardization, sample quality, and documentation integrity. Procurement emphasizes reliability, validated monitoring, and operational continuity across repositories. Cross-institution collaboration also increases adoption of compatible storage and handling systems that support harmonized practices.
Asia Pacific
Asia Pacific captured 23.9% of the Biobank Equipment Market in 2025. Asia Pacific growth is supported by expanding life-science R&D, rising clinical trial activity, and continued investments in laboratory infrastructure. Repositories increasingly adopt scalable storage capacity and standardized handling practices as sample volumes increase. Automation adoption is rising in larger repositories seeking productivity improvements and reduced manual handling risk.
Latin America
Latin America accounted for 7.8% of the Biobank Equipment Market in 2025. Latin America demand is driven by selective modernization of laboratory infrastructure and increased participation in clinical research initiatives. Procurement tends to prioritize core storage systems and monitoring reliability as repositories mature. Growth opportunities improve where institutional funding supports repository expansion and standardized operating practices.
Middle East & Africa
Middle East & Africa represented 6.6% of the Biobank Equipment Market in 2025. Middle East & Africa demand is concentrated in select research hubs and healthcare modernization programs that invest in repository capacity and cold storage integrity. Procurement focuses on reliable storage performance, monitoring, and service availability to manage operational risk. Expansion potential improves as national genomics and research initiatives increase long-term biospecimen storage needs.
Shape Your Report to Specific Countries or Regions & Enjoy 30% Off!
Competitive Landscape
Biobank Equipment Market competition is shaped by performance differentiation in temperature stability, capacity scalability, automation readiness, and monitoring integration. Suppliers compete on reliability, service networks, and systems that support compliance workflows through alarms, access controls, and documentation features. Portfolio breadth across storage, sample handling, and automation is increasingly important as repositories seek end-to-end workflow standardization. Strategic focus is also placed on energy efficiency, remote management, and modular system configurations that reduce operating risk.
Azenta Life Sciences is positioned around automation-enabled storage and sample management workflows that support high-volume repositories requiring standardized retrieval and traceability. Azenta Life Sciences benefits from integrated approaches that connect storage infrastructure with controlled access and operational integrity requirements. Azenta Life Sciences also aligns with repository scale-up needs where automation reduces manual handling and improves throughput consistency. Azenta Life Sciences strategy reflects growing demand for centralized repositories that require resilient, audit-ready storage operations.
The industry research and growth report includes detailed analyses of the competitive landscape of the market and information about key companies, including:
- Azenta Life Sciences
- Becton, Dickinson and Company
- Chart Industries, Inc.
- Danaher Corporation
- Hamilton Company
- Merck KGaA
- PHC Holdings Corporation
- QIAGEN N.V.
- Tecan Group Ltd.
- Thermo Fisher Scientific Inc.
Qualitative and quantitative analysis of companies has been conducted to help clients understand the wider business environment as well as the strengths and weaknesses of key industry players. Data is qualitatively analyzed to categorize companies as pure play, category-focused, industry-focused, and diversified; it is quantitatively analyzed to categorize companies as dominant, leading, strong, tentative, and weak.
Recent Developments
- In a March 2026 acquisition update, Azenta, Inc. announced that its subsidiary Azenta UK Ltd acquired UK Biocentre Limited to expand biorepository capabilities in the UK and strengthen its wider European sample management footprint. Azenta said the acquisition will support broader automated sample storage, integrated processing services, and deployment of its BioArc Ultra platform for pharmaceutical, biotechnology, academic, and public health customers.
- In a February 2026 partnership update, Azenta Life Sciences announced a strategic partnership with Frontier Space, which was described as a pioneer in commercial space-based research systems. The announcement framed the deal as a strategic collaboration connected to advanced research and biobanking-related systems development.
- In a June 2025 partnership update, Elucidata and Sapien Biosciences announced a strategic collaboration to transform Sapien’s biobank assets into AI-ready, multimodal data products for drug and diagnostic development. Sapien Biosciences said its biobank includes more than 300,000 patient samples and over 2 million pathology samples, showing the scale of infrastructure tied to the broader biobanking ecosystem.
- In a May 2025 product launch update, Haier Biomedical introduced its Ultra Eco next-generation ultra-low temperature freezer, featuring 4.16 kWh/day energy consumption, low-GWP refrigerants and insulation materials, and advanced IoT connectivity. Haier positioned the product for laboratories and biobanks that need sustainable cold storage with lower long-term operating costs and strong sample protection performance
Report Scope
| Report Attribute |
Details |
| Market size value in 2025 |
USD 4,177.66 million |
| Revenue forecast in 2032 |
USD 7,686.11 million |
| Growth rate (CAGR) |
9.1% (2025–2032) |
| Base year |
2025 |
| Forecast period |
2026-2032 |
| Quantitative units |
USD million |
| Segments covered |
By Product Type; By Sample Type; By End User; By Application |
| Regional scope |
North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
| Key companies profiled |
Azenta Life Sciences, Becton, Dickinson and Company, Chart Industries, Inc., Danaher Corporation, Hamilton Company, Merck KGaA, PHC Holdings Corporation, QIAGEN N.V., Tecan Group Ltd., Thermo Fisher Scientific Inc. companies |
| No.of Pages |
335 |
Segmentation
By Product Type
- Freezers and refrigerators
- Cryogenic storage systems
- Alarm and monitoring systems
- Sample handling and processing equipment
- Automated storage and retrieval systems
- Consumables and accessories
By Sample Type
- Blood and blood derivatives
- Tissue samples
- Cell lines
- Nucleic acids (DNA/RNA)
- Biological fluids
- Other biospecimens
By End User
- Pharmaceutical and biotechnology companies
- Academic and research institutes
- Hospitals and diagnostic centers
- Biobanks and gene banks
- Contract research organizations
By Application
- Regenerative Medicine
- Drug Discovery
- Others
By Region
- North America
- Europe
- Germany
- France
- U.K.
- Italy
- Spain
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- South-east Asia
- Rest of Asia Pacific
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Middle East & Africa
- GCC Countries
- South Africa
- Rest of the Middle East and Africa




