Biochip Product Market Overview:
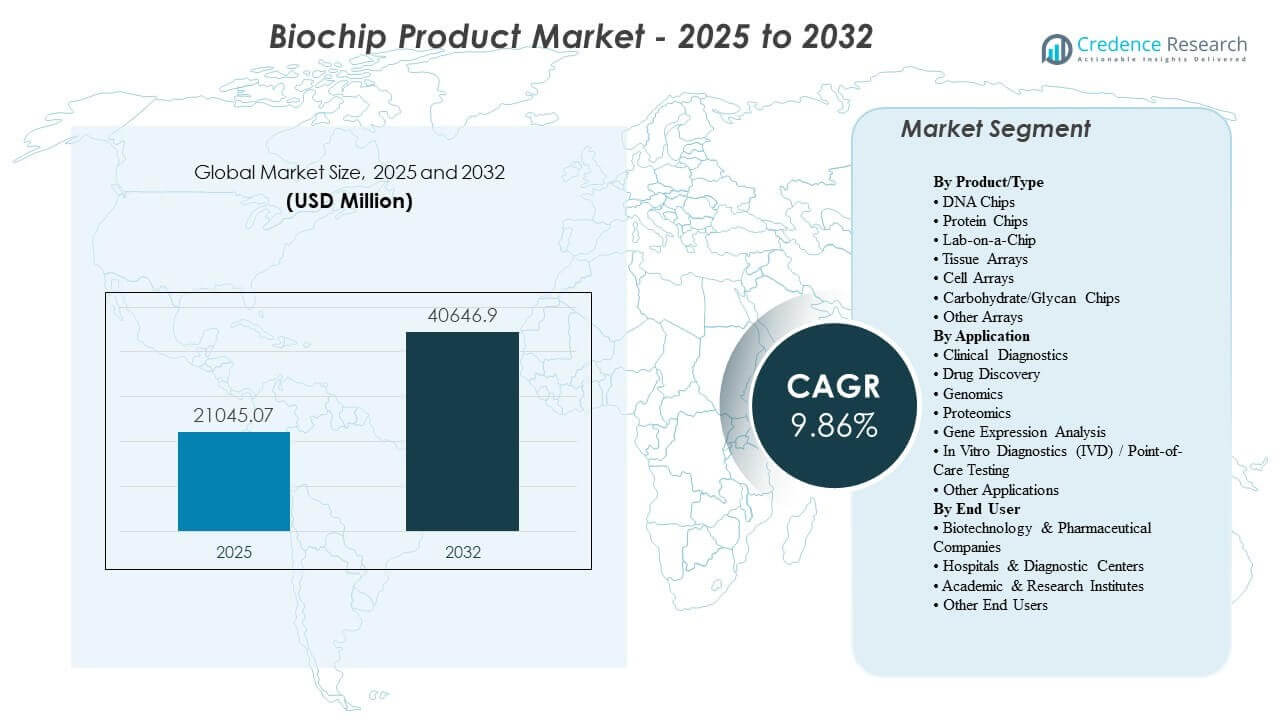
The global Biochip Product Market size was estimated at USD 21045.07 million in 2025 and is expected to reach USD 40646.9 million by 2032, growing at a CAGR of 9.86% from 2025 to 2032. Biochip Product Market growth is primarily driven by the rising need for faster, multiplexed, and more standardized testing workflows that shorten diagnostic turnaround times and improve decision-making across clinical and research settings. Biochip Product Market adoption is also supported by expanding healthcare and life-science infrastructure in emerging economies, alongside continued innovation in microfluidics, assay miniaturization, and integrated platform automation.
| REPORT ATTRIBUTE |
DETAILS |
| Historical Period |
2020-2024 |
| Base Year |
2025 |
| Forecast Period |
2026-2032 |
| Biochip Product Market Size 2025 |
USD 21045.07 Million |
| Biochip Product Market, CAGR |
9.7% |
| Biochip Product Market Size 2032 |
USD 40646.9 Million |
Key Market Trends & Insights
- North America accounted for 38.4% share in 2025, supported by high testing intensity, strong reimbursement pathways, and advanced laboratory infrastructure.
- Asia Pacific represented 32.1% share in 2025, driven by expanding diagnostics capacity and accelerating genomics and biopharma investments.
- Lab-on-a-Chip accounted for the largest share of 52.6% in 2025, reflecting the shift toward integrated, miniaturized workflows for rapid testing.
- Clinical Diagnostics held 38.7% share in 2025, supported by sustained demand for infectious disease testing, oncology profiling, and multiplex panels.
- Hospitals & Diagnostic Centers captured 42.3% share in 2025, led by operational need for faster time-to-result and scalable routine testing.
Segment Analysis
Biochip Product Market demand is shaped by procurement preferences that prioritize consistent performance under real operating conditions, reduced hands-on time, and simplified workflow integration across sample preparation, detection, and reporting. Buyers increasingly evaluate total cost of ownership through consumable utilization rates, calibration frequency, service uptime, and compatibility with existing laboratory information systems. Standardized cartridges and assay-ready formats are gaining traction because they reduce variability and enable broader deployment across multi-site laboratory networks. Biochip Product Market purchasing decisions also reflect the growing importance of analytics and software layers that improve interpretability, traceability, and quality management.
Biochip Product Market segmentation continues to diversify as healthcare systems balance centralized high-throughput laboratories with decentralized testing environments. Integrated platforms that combine microfluidics with multiplex detection are improving efficiency for complex panels, while array-based formats remain relevant for high-content profiling in genomics, proteomics, and gene expression workflows. Biochip Product Market end-user adoption is reinforced by biopharma screening needs, translational research programs, and hospital demand for rapid rule-in/rule-out testing that improves patient flow. Competitive differentiation increasingly depends on menu breadth, workflow simplicity, regulatory readiness, and supply reliability for consumables.
Access crucial information at unmatched prices!
Request your sample report today & start making informed decisions powered by Credence Research Inc.!
Download Sample
By Product/Type Insights
Lab-on-a-Chip accounted for the largest share of 52.6% in 2025. Lab-on-a-Chip leads because integrated microfluidic processing reduces reagent use and manual handling, improving operational efficiency in both clinical and research workflows. Lab-on-a-Chip platforms support rapid multiplex testing and are well-suited for near-patient and decentralized environments where turnaround time is critical. Lab-on-a-Chip adoption is also strengthened by platform ecosystems that bundle instruments, cartridges, and software, helping laboratories standardize results across sites.
By Application Insights
Clinical Diagnostics accounted for the largest share of 38.7% in 2025. Clinical Diagnostics leads because multiplexed biochip formats support faster time-to-result and improved clinical decision support in infectious disease and oncology-related testing pathways. Clinical Diagnostics utilization benefits from standardized assay menus that reduce variability and enable repeatable results across different operators and facilities. Clinical Diagnostics demand is also supported by hospital workflow pressure to shorten patient stays and improve triage efficiency through rapid, high-confidence results.
By End User Insights
Hospitals & Diagnostic Centers accounted for the largest share of 42.3% in 2025. Hospitals & Diagnostic Centers lead because routine testing volumes and operational constraints create strong incentives for integrated platforms that reduce manual steps and speed reporting. Hospitals & Diagnostic Centers also benefit from standardized consumable-driven workflows that simplify training and improve quality control across shifts. Hospitals & Diagnostic Centers adoption is reinforced by the growing use of syndromic panels and rapid molecular testing that supports timely treatment decisions and infection control protocols.
Biochip Product Market Drivers
Expansion of multiplex molecular and omics-based testing in clinical workflows
Biochip Product Market growth benefits from increased clinical use of multiplex testing that consolidates multiple targets into a single run. Healthcare systems are seeking faster diagnostic clarity for infectious diseases and oncology pathways, which supports higher utilization of integrated testing formats. Laboratories also value reduced retesting and fewer sequential assays, improving throughput and operational predictability. Biochip Product Market solutions that combine assay menus with streamlined reporting are gaining preference in high-volume settings.
- For instance, bioMérieux’s BIOFIRE BCID2 Panel is designed to detect 43 targets associated with bloodstream infections, including 10 antimicrobial resistance genes, with results available in about one hour from positive blood culture, demonstrating the clinical value of consolidated multiplex testing in time-sensitive care pathways.
Platform automation and miniaturization improving lab productivity
Biochip Product Market adoption is supported by automation and miniaturization that lowers hands-on time and improves repeatability. Integrated microfluidic designs reduce sample and reagent volumes, which can lower per-test costs and improve sustainability outcomes. Laboratories increasingly prioritize systems that reduce operator dependency and improve inter-site comparability. Biochip Product Market procurement often favors platforms that simplify maintenance and enable higher instrument uptime.
- For instance, Bio-Rad’s ddSEQ 3′ Single-Cell RNA-Seq workflow supports whole-transcriptome analysis of 500 to 5,000 individual cells, completes encapsulation for four samples in less than 5 minutes, and reduces hands-on time by up to 50% versus other kits, highlighting how microfluidic miniaturization can materially improve lab productivity.
Rising biopharma R&D intensity and translational research demand
Biochip Product Market demand is reinforced by expanding biopharma pipelines that require scalable screening, biomarker discovery, and assay development. Multi-omics and gene-expression workflows support wider use of arrays and integrated analytical tools for target validation and patient stratification. Research organizations also prefer standardized consumable ecosystems that reduce batch-to-batch variability across studies. Biochip Product Market suppliers that offer robust assay development support and workflow compatibility gain advantage in these environments.
Increasing standardization, quality requirements, and data integration needs
Biochip Product Market growth is driven by greater emphasis on standard operating procedures, quality management, and traceability. Hospitals and large laboratory networks increasingly require integration with digital quality systems and laboratory information environments to reduce error rates. Standardized cartridges and validated assay menus can simplify audit readiness and improve consistency across operators. Biochip Product Market platforms that align with regulated workflows and provide strong service support benefit from these requirements.
Biochip Product Market Challenges
Biochip Product Market expansion is constrained by cost sensitivity and procurement complexity, particularly where budgets prioritize core analyzers over specialized platforms. High upfront instrument costs, recurring consumable expenses, and service contracts can slow adoption in smaller laboratories. Laboratories may also face switching friction when existing workflows are optimized around incumbent instruments and validated protocols. Biochip Product Market adoption can therefore require strong evidence of throughput gains, menu relevance, and operational savings.
- For instance, Luminex’s MAGPIX analyzer is priced at approximately $27,500 roughly half the cost of the Luminex 200 system and was specifically engineered to lower the entry barrier for smaller laboratories, offering simultaneous measurement of up to 50 analytes per microplate well with a plate read time of 60 minutes.
Biochip Product Market growth also faces technical and operational barriers related to assay performance consistency across diverse sample types and operating environments. Cross-reactivity risk, sample preparation complexity, and the need for calibration and quality controls can affect deployment at scale. Regulatory timelines and clinical validation requirements add time and cost for new assay menus, particularly for high-impact diagnostic claims. Biochip Product Market suppliers must also manage supply continuity for cartridges and reagents to protect customer uptime and trust.
Biochip Product Market Trends and Opportunities
Biochip Product Market trends increasingly favor integrated, menu-expandable platforms that support rapid syndromic testing and broader decentralized deployment. Hospitals are prioritizing fast turnaround and standardized workflows, which increases demand for cartridge-based systems and streamlined reporting tools. Software capabilities such as automated interpretation, connectivity, and audit-ready traceability are becoming central differentiators. Biochip Product Market vendors that deliver end-to-end workflow integration are well-positioned to improve retention and recurring revenue.
Biochip Product Market opportunities are expanding in precision medicine and translational research where multi-omics profiling is becoming more routine. Growing adoption of single-cell and high-content analysis strengthens demand for scalable sample processing and higher multiplex capacity. Emerging economies offer additional upside as diagnostics infrastructure and biopharma ecosystems scale, creating new demand pools for both clinical and research applications. Biochip Product Market providers that localize support, strengthen distribution, and ensure supply resilience can capture these growth pockets.
- For instance, 10x Genomics states that its Chromium GEM-X platform can process 1–3,072 samples, profile hundreds to millions of cells per run, generate 10,000+ gel beads-in-emulsion in 4 minutes, achieve up to 80% cell capture with up to 95% usable reads, and scale multiplexed studies to 384 samples or up to 100 million cells per week, which directly supports more routine single-cell and multi-omics research workflows.
Regional Insights
North America
Biochip Product Market in North America accounted for 38.4% share in 2025, supported by mature laboratory ecosystems and strong adoption of advanced diagnostics. The United States drives demand through high testing intensity across infectious disease, oncology, and specialized reference lab services. Research institutions and biopharma programs also support sustained uptake of high-throughput profiling tools. Biochip Product Market suppliers benefit from strong service infrastructure and established procurement frameworks that support platform expansion.
Europe
Biochip Product Market in Europe represented 21.7% share in 2025, supported by established healthcare systems and strong research networks. Demand is concentrated in major Western European markets where laboratory consolidation and standardization support integrated platforms. Regulatory and quality expectations reinforce adoption of validated workflows and reliable consumable supply. Biochip Product Market growth is also supported by public-private research initiatives that sustain genomics and proteomics tool demand.
Asia Pacific
Biochip Product Market in Asia Pacific reached 32.1% share in 2025, reflecting rapid expansion of diagnostics capacity and rising life-science investment. China, Japan, India, and South Korea contribute to growth through scaling hospital networks, biopharma R&D, and precision-medicine initiatives. Large population bases and increasing test volumes support higher utilization of multiplex and integrated workflows. Biochip Product Market players that expand local partnerships and strengthen distribution can improve penetration and retention.
Latin America
Biochip Product Market in Latin America accounted for 5.1% share in 2025, with adoption concentrated in leading metropolitan hospital systems and private diagnostic chains. Budget constraints and procurement variability can slow platform rollouts outside major cities. Demand tends to be strongest where rapid testing improves patient throughput and reduces repeat visits. Biochip Product Market growth benefits from gradual modernization of laboratory infrastructure and increased access to advanced diagnostic menus.
Middle East & Africa
Biochip Product Market in the Middle East & Africa held 2.7% share in 2025, reflecting early-stage penetration outside select high-capacity hubs. Adoption is strongest in GCC markets and South Africa, where reference laboratories and tertiary hospitals lead demand. Expansion is influenced by healthcare investment programs and the need for faster infectious disease and syndromic testing capabilities. Biochip Product Market growth in the region depends on distribution strength, service availability, and cost-effective deployment models.
Competitive Landscape
Biochip Product Market competition is characterized by platform ecosystems that combine instruments, consumables, and software, with differentiation driven by assay menu breadth, workflow simplicity, and operational reliability. Suppliers compete on clinical validation depth, speed of new assay introductions, and the ability to integrate results into laboratory information environments. Commercial strategies emphasize recurring consumable revenue, strong service coverage, and long-term customer retention through workflow lock-in. Biochip Product Market leaders also invest in partnerships and acquisitions to strengthen upstream sample preparation and downstream analytics capabilities.
Thermo Fisher Scientific competes through broad life-science instrumentation coverage and the ability to bundle reagents, consumables, and analytical workflows to support high-throughput laboratories. Thermo Fisher Scientific benefits from scale in manufacturing and distribution, helping to maintain supply continuity for consumables and shorten delivery lead times. Thermo Fisher Scientific also emphasizes workflow compatibility across genomics and applied markets, which supports cross-selling into established customer bases. Biochip Product Market positioning is strengthened by ongoing expansion of assay options and platform usability improvements.
The industry research and growth report includes detailed analyses of the competitive landscape of the market and information about key companies, including:
- Thermo Fisher Scientific
- Illumina, Inc.
- Agilent Technologies, Inc.
- F. Hoffmann-La Roche Ltd.
- QIAGEN
- Bio-Rad Laboratories, Inc.
- Abbott Laboratories
- Standard BioTools Inc.
- Cepheid
- bioMérieux
- Revvity
- Randox Laboratories Ltd.
Qualitative and quantitative analysis of companies has been conducted to help clients understand the wider business environment as well as the strengths and weaknesses of key industry players. Data is qualitatively analyzed to categorize companies as pure play, category-focused, industry-focused, and diversified; it is quantitatively analyzed to categorize companies as dominant, leading, strong, tentative, and weak.
Shape Your Report to Specific Countries or Regions & Enjoy 30% Off!
Recent Developments
- In January 2025, bioMérieux announced an agreement to acquire SpinChip Diagnostics ASA, a Norwegian diagnostics company whose small benchtop immunoassay platform is designed for near-patient testing and can deliver results from a whole-blood sample in about 10 minutes.
- In January 2025, Coherent Corp launched its new pin-hole array biochips for medical diagnostics, with the company highlighting gene sequencing and diagnostic applications as key use cases for the technology.
- In April 2025, CN Bio established a strategic partnership with Pharmaron to validate CN Bio’s PhysioMimix organ-on-a-chip technology, integrate organ-on-a-chip tools into Pharmaron’s global R&D platform, and jointly develop new applications for drug discovery and development.
- In September 2025, Advanced Biomed Inc. launched the A+PerfusC system, an integrated perfusion 3D cell culture platform for precision medicine and drug discovery, and said the product is intended for uses such as oncology research, in vitro drug screening, organoid studies, and stem cell research
Report Scope
| Report Attribute |
Details |
| Market size value in 2025 |
USD 21045.07 million |
| Revenue forecast in 2032 |
USD 40646.9 million |
| Growth rate (CAGR) |
9.86% (2025–2032) |
| Base year |
2025 |
| Forecast period |
2026–2032 |
| Quantitative units |
USD million |
| Segments covered |
By Product/Type, By Application, By End User |
| Regional scope |
North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
| Key companies profiled |
Thermo Fisher Scientific; Illumina, Inc.; Agilent Technologies, Inc.; F. Hoffmann-La Roche Ltd.; QIAGEN; Bio-Rad Laboratories, Inc.; Abbott Laboratories; Standard BioTools Inc.; Cepheid; bioMérieux; Revvity; Randox Laboratories Ltd. |
| No. of Pages |
334 |
Segmentation
By Product/type
- DNA Chips
- Protein Chips
- Lab-on-a-Chip
- Tissue Arrays
- Cell Arrays
- Carbohydrate/Glycan Chips
- Other Arrays
By Application
- Clinical Diagnostics
- Drug Discovery
- Genomics
- Proteomics
- Gene Expression Analysis
- In Vitro Diagnostics (IVD) / Point-of-Care Testing
- Other Applications
By End User
- Biotechnology & Pharmaceutical Companies
- Hospitals & Diagnostic Centers
- Academic & Research Institutes
- Other End Users
By Region
- North America
- Europe
- Germany
- France
- U.K.
- Italy
- Spain
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- South-east Asia
- Rest of Asia Pacific
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Middle East & Africa
- GCC Countries
- South Africa
- Rest of the Middle East and Africa




