Abdominal Stent Graft System Market Overview:
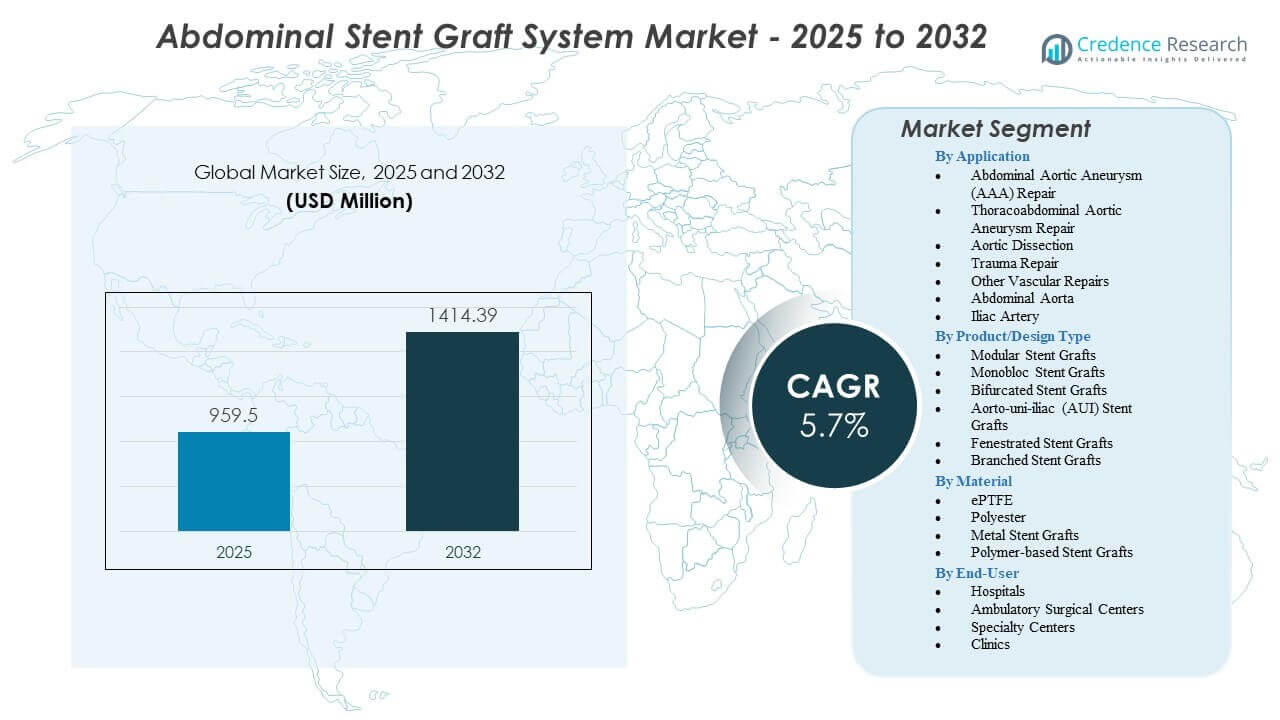
The Abdominal Stent Graft System Market is projected to grow from USD 959.5 million in 2025 to an estimated USD 1,414.39 million by 2032, with a compound annual growth rate (CAGR of 5.7%) from 2025 to 2032.
| REPORT ATTRIBUTE |
DETAILS |
| Historical Period |
2020-2023 |
| Base Year |
2024 |
| Forecast Period |
2025-2032 |
| Abdominal Stent Graft System Market Size 2025 |
USD 959.5 Million |
| Abdominal Stent Graft System Market, CAGR |
5.7% |
| Abdominal Stent Graft System Market Size 2032 |
USD 1,414.39 Million |
Abdominal Stent Graft System Market Insights:
- Demand strengthens due to rising AAA detection, growing EVAR adoption, and continuous improvement in graft materials and delivery systems that support safer and faster procedures.
- Market restraints include complex anatomical challenges, strict regulatory requirements, and the need for long-term durability data that influences device selection across clinical settings.
- North America leads the market due to strong clinical capacity and high procedural adoption, while Europe maintains stable growth supported by structured screening and advanced EVAR practices.
- Asia Pacific emerges as the fastest-growing region, driven by expanding vascular centers, rising diagnostic awareness, and increasing investment in minimally invasive endovascular repair.
Abdominal Stent Graft System Market Drivers
Growing Preference for Minimally Invasive Repair Techniques
Minimally invasive EVAR procedures continue to drive strong growth in the Abdominal Stent Graft System Market due to shorter recovery time and reduced surgical trauma. Surgeons prefer EVAR for high-risk patients because it lowers complication rates and improves long-term outcomes. Hospitals upgrade vascular units to support advanced endovascular tools, encouraging wider use of next-generation grafts. Screening programs help detect aneurysms earlier, which increases the number of eligible patients. Device makers invest in lower-profile delivery systems to support difficult anatomies. Clinical studies confirm better durability for updated graft designs, reinforcing physician confidence. Awareness programs push more patients toward elective repair instead of emergency surgery. Strong technology adoption in developed regions accelerates demand.
Access crucial information at unmatched prices!
Request your sample report today & start making informed decisions powered by Credence Research Inc.!
Download Sample
Increasing Global Burden of Abdominal Aortic Aneurysms
Rising AAA prevalence fuels steady adoption in the Abdominal Stent Graft System Market because screening identifies more cases that need structured follow-up. Ageing populations raise the number of patients at risk, which strengthens procedural demand. Lifestyle-linked risk factors increase aneurysm formation across multiple regions. Health systems expand diagnostic programs to support early detection, encouraging treatment before rupture risk grows. Patients receive guidance on elective repair timelines, which increases the number of planned EVAR procedures. Advancements in imaging improve detection accuracy and reduce diagnostic delays. Hospitals emphasize early intervention to avoid emergency events. Vascular surgeons adopt newer grafts that offer stronger sealing in complex anatomies.
Technological Advancements in Stent Graft Design and Materials
Continuous innovation strengthens confidence in the Abdominal Stent Graft System Market by supporting safer and more precise deployment. Manufacturers introduce grafts with improved flexibility to help surgeons navigate tortuous vessels. Lower-profile systems give clinicians capacity to treat patients previously considered unsuitable for EVAR. Modern graft fabrics improve durability and reduce the chance of postoperative complications. Advanced fixation technologies help maintain long-term stability. Patients benefit from shorter hospital stays, which supports wider preference for EVAR over open repair. Research teams conduct long-term follow-up studies to validate outcomes of new devices. Improvements in imaging and guidance tools enhance procedural accuracy.
- For instance, Gore’s Excluder Conformable device features a ≤15 Fr delivery system that enabled access in patients with narrow iliac pathways during trial assessments.
Expansion of Specialized Vascular Centers Worldwide
Growing investment in vascular infrastructure supports stable demand in the Abdominal Stent Graft System Market due to improved access to trained specialists. Countries expand EVAR programs to strengthen emergency preparedness for aneurysm rupture cases. Hospitals build hybrid operating rooms that integrate imaging and surgical capabilities. Surgeons receive advanced procedural training, which improves EVAR adoption in emerging regions. Patients choose specialized centers because they offer safer outcomes and shorter recovery periods. Health systems promote elective repair guidelines to reduce rupture-related deaths. Procurement teams prioritize next-generation grafts to support complex anatomies. Expanding referral networks increase the number of treated patients each year.
- For instance, Siemens Healthineers reported global adoption of more than 1,500 ARTIS hybrid OR systems designed to support high-precision EVAR procedures.
Market Trends
Shift Toward Customizable and Anatomy-Adaptive Graft Systems
New design strategies shape trends in the Abdominal Stent Graft System Market through grafts that adapt to varied patient anatomies. Manufacturers introduce grafts with enhanced conformability for challenging neck angles. Personalized planning tools improve pre-operative decision making. Surgeons use advanced sizing software to match grafts to complex vascular dimensions. Customizable systems help reduce procedural complications. Flexible materials enhance device behavior during deployment. Hospitals integrate planning platforms into routine workflows. Clinicians favor grafts that support long-term durability across diverse patient groups.
- For instance, Gore’s conformable EXCLUDER device demonstrated precise wall apposition across angulations up to 90 degrees during clinical evaluations.
Integration of Digital Imaging and AI-Supported Planning Tools
Digital platforms influence the Abdominal Stent Graft System Market by supporting accurate pre-procedure mapping through AI-based measurements. Surgeons use automated vessel analysis tools for faster planning. Software improves precision in graft placement and reduces sizing errors. Hospitals adopt integrated platforms for EVAR workflow optimization. Image-guided navigation supports safer outcomes. Training programs highlight the value of digital planning in complex cases. AI tools help predict anatomical risks. Health systems move toward standardized digital EVAR workflows.
Growing Adoption of Low-Profile Delivery Systems
Next-generation low-profile systems impact the Abdominal Stent Graft System Market by offering improved access through narrow or calcified vessels. Surgeons prefer these systems for patients who face challenges with standard delivery tools. Manufacturers redesign catheters to reduce insertion trauma. Patients benefit from reduced operative time. Hospitals report shorter recovery periods with updated systems. Lower-profile solutions support broader anatomical applicability. Device makers highlight reduced post-procedural complications. Demand grows rapidly in regions with rising minimally invasive surgery capacity.
- For instance, Medtronic’s Endurant II delivery system features profiles as low as 14 Fr, enabling treatment in patients previously unsuitable for EVAR.
Increasing Post-Market Surveillance and Long-Term Outcome Studies
Long-term data programs influence the Abdominal Stent Graft System Market because they strengthen trust in device performance. Manufacturers track device behavior across diverse patient groups. Surveillance studies highlight durability trends that support regulatory approvals. Clinicians use real-world findings to refine procedural strategies. Health systems prioritize grafts with strong evidence backing. Research teams expand global registries to compare outcomes. Surgeons gain deeper insight into device complications. Long-term reporting supports continuous product refinement.
Abdominal Stent Graft System Market Challenges Analysis
Complex Anatomies and Procedural Limitations
Complex patient anatomies create strong challenges for the Abdominal Stent Graft System Market due to limited suitability for certain graft designs. Surgeons encounter difficulty when treating short necks, severe angulation, or calcified vessels. These cases require advanced planning and higher surgical skill. Some anatomies still favor open repair, which limits EVAR adoption. Device makers work to improve sealing technologies, yet anatomical restrictions continue to impact decision making. Clinical outcomes depend heavily on surgeon experience. Hospitals face pressure to expand training programs. Lack of uniform screening reduces early identification of complex cases.
Regulatory Requirements and Device Durability Concerns
Strict global regulatory expectations influence the Abdominal Stent Graft System Market because they require extensive data on long-term durability. Manufacturers must demonstrate strong clinical performance before achieving approvals. Procurement teams evaluate durability metrics carefully. Hospitals seek grafts with proven performance histories to reduce reintervention risk. Durability concerns influence physician confidence when selecting newer devices. Regulatory delays slow market entry for updated designs. Long-term follow-up studies require significant investment. Variability in guidelines across regions creates added complexity.
Abdominal Stent Graft System Market Opportunities
Expansion of EVAR Access in Emerging Healthcare Markets
\Emerging regions create sizable opportunities for the Abdominal Stent Graft System Market due to expanding vascular care infrastructure. Governments invest in diagnostic programs to detect aneurysms earlier. Hospitals develop new hybrid operating rooms to support EVAR. Training programs strengthen surgeon capabilities. Patients gain access to minimally invasive repair options that were previously unavailable. Manufacturers enter partnerships to expand distribution. Early adoption grows as awareness campaigns improve screening rates. Rapid healthcare modernization supports long-term demand growth.
Development of Next-Generation Smart and Adaptive Graft Technologies
Technology development opens major opportunities for the Abdominal Stent Graft System Market through grafts that integrate advanced materials and improved fixation. Engineers explore designs that adapt to vessel movement for better durability. Precision imaging tools support enhanced deployment accuracy. Makers test new sealing mechanisms to reduce endoleaks. Hospitals value grafts that require fewer revisions. Research teams expand collaboration with imaging specialists. Surgeons prefer systems that improve procedural predictability. Innovation pipelines support broader anatomical suitability across global markets.
Market Segmentation Analysis:
By Application
Application segments shape demand in the Abdominal Stent Graft System Market, with AAA repair holding the largest share due to high diagnosis rates and strong EVAR adoption. Thoracoabdominal aneurysm repair grows steadily because surgeons rely on advanced grafts for complex cases. Aortic dissection treatment benefits from improved imaging and faster intervention strategies. Trauma repair gains traction where hybrid surgical centers support emergency EVAR use. Other vascular repairs support niche demand for graft adaptability. Abdominal aorta interventions maintain stable volume due to routine screening. Iliac artery repairs advance with low-profile systems that improve access in difficult anatomies.
- For instance, Cook Medical’s Zenith Fenestrated system reported a technical success rate above 94% in multicenter evaluations for complex aortic anatomies.
By Product/Design Type
Product design influences the Abdominal Stent Graft System Market, with modular grafts preferred for flexibility in varied anatomies. Monobloc grafts attract users who seek simplified deployment. Bifurcated grafts dominate due to strong compatibility with infrarenal AAA cases. AUI grafts serve patients with narrow or diseased iliac systems. Fenestrated grafts support repairs in short necks and complex vessel branches. Branched grafts help surgeons manage thoracoabdominal involvement with higher stability. Each design type offers a targeted clinical advantage that supports broader procedural capability.
- For example, the Aorfix™ stent graft is the only FDA-approved EVAR device rated for aortic neck angulations up to 90 degrees, enabled by its modular two-piece design and fishmouth proximal sealing ring. Its helical and circular nitinol ring structure improves conformability in hostile anatomies, with the PYTHAGORAS trial confirming comparable outcomes in both standard and severely angulated neck groups.
By Material
Material selection drives performance in the Abdominal Stent Graft System Market, with ePTFE valued for durability and smooth blood flow. Polyester remains widely used due to strong clinical evidence and long-term stability. Metal stent graft frames provide fixation strength and shape retention. Polymer-based grafts support innovation in sealing technology and adaptive designs. Each material group supports different durability and handling requirements that influence surgeon preference.
By End-User
End-users guide adoption in the Abdominal Stent Graft System Market, with hospitals leading due to high surgical capacity and advanced imaging access. Ambulatory surgical centers expand use where minimally invasive EVAR procedures fit outpatient models. Specialty centers adopt complex graft systems for high-acuity cases. Clinics support diagnostics and referral pathways that sustain procedural demand across broader healthcare networks.
Segmentation:
By Application
- Abdominal Aortic Aneurysm (AAA) Repair
- Thoracoabdominal Aortic Aneurysm Repair
- Aortic Dissection
- Trauma Repair
- Other Vascular Repairs
- Abdominal Aorta
- Iliac Artery
By Product/Design Type
- Modular Stent Grafts
- Monobloc Stent Grafts
- Bifurcated Stent Grafts
- Aorto-uni-iliac (AUI) Stent Grafts
- Fenestrated Stent Grafts
- Branched Stent Grafts
By Material
- ePTFE
- Polyester
- Metal Stent Grafts
- Polymer-based Stent Grafts
By End-User
- Hospitals
- Ambulatory Surgical Centers
- Specialty Centers
- Clinics
By Region
- North America
- Europe
- Germany
- France
- U.K.
- Italy
- Spain
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- South-east Asia
- Rest of Asia Pacific
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Middle East & Africa
- GCC Countries
- South Africa
- Rest of the Middle East and Africa
Regional Analysis:
North America holds the largest share of the Abdominal Stent Graft System Market, capturing roughly 38% due to strong EVAR adoption, early availability of advanced graft technologies, and wide access to vascular specialists. Hospitals in the region benefit from high diagnostic rates that support early AAA detection. Training programs strengthen surgeon capability and improve procedural outcomes. Regulatory pathways encourage timely product launches. It continues to lead because healthcare networks prioritize minimally invasive repair. High patient awareness supports steady procedural volumes across major centers.
Europe accounts for nearly 32% of the global market and maintains strong growth supported by mature screening programs and broad acceptance of endovascular repair. Clinical societies promote structured AAA surveillance, which increases elective repair rates. Countries with advanced imaging networks record higher detection accuracy and referral efficiency. Surgeons in leading markets adopt fenestrated and branched grafts for complex anatomies. Hospitals participate in long-term registries that help refine graft selection. Strong research environments reinforce device innovation and accelerate adoption.
Asia Pacific holds close to 22% and represents the fastest-growing region because healthcare systems invest heavily in vascular infrastructure. Rising diagnosis rates in China, India, and Japan increase procedure volume. Hospitals expand hybrid operating rooms to support advanced EVAR capability. Surgeons gain access to extensive training programs that strengthen procedural safety. It accelerates rapidly due to growing preference for minimally invasive repair. Latin America and the Middle East & Africa together capture the remaining 8%, supported by improving access to specialized care and broader clinical training across emerging vascular centers.
Shape Your Report to Specific Countries or Regions & Enjoy 30% Off!
Key Player Analysis:
- Medtronic
- L. Gore & Associates
- Terumo Corporation / Terumo Aortic
- Cook Medical
- Endologix
- MicroPort Scientific / Endovastec
- Becton Dickinson (BD)
- Jotec (part of CryoLife / Artivion)
- Bolton Medical (acquired by Terumo Aortic)
- Lombard Medical
- Cordis Corporation
- Artivion
- Sofmedica
- Cardinal Health
- Braile Biomédica
- Bentley InnoMed
Competitive Analysis:
Competitive dynamics in the Abdominal Stent Graft System Market are shaped by strong innovation pipelines, extensive clinical data, and expanding global distribution networks. Key players focus on lower-profile delivery systems, enhanced fixation mechanisms, and improved sealing technologies to strengthen device durability. Companies invest in post-market surveillance programs to validate long-term outcomes and build physician confidence. Hospitals prioritize grafts with proven reliability in complex anatomies, which increases competition for advanced fenestrated and branched systems. Partnerships with vascular centers help firms expand training and support services. It drives companies to emphasize product differentiation, regulatory compliance, and consistent clinical performance. Market leaders maintain advantage through strong brand reputation, ongoing device refinements, and broad geographical presence. New entrants face pressure to meet high evidence standards and compete with established technology platforms.
Recent Developments:
- In October 2025, Medtronic received FDA labeling approval for its Endurant™ stent graft system, the first and only to treat ruptured abdominal aortic aneurysms (rAAA).
Report Coverage:
The research report offers an in-depth analysis based on Application, Product/Design Type, Material, and End-User. It details leading market players, providing an overview of their business, product offerings, investments, revenue streams, and key applications. Additionally, the report includes insights into the competitive environment, SWOT analysis, current market trends, as well as the primary drivers and constraints. Furthermore, it discusses various factors that have driven market expansion in recent years. The report also explores market dynamics, regulatory scenarios, and technological advancements that are shaping the industry. It assesses the impact of external factors and global economic changes on market growth. Lastly, it provides strategic recommendations for new entrants and established companies to navigate the complexities of the market.
Future Outlook:
- Growing adoption of minimally invasive repair will strengthen demand for advanced graft designs across the Abdominal Stent Graft System Market.
- Expansion of vascular screening programs will support earlier detection and increase elective EVAR procedures.
- Surgeons will rely more on low-profile systems that enable access in narrow or complex anatomies.
- Digital planning platforms and AI-supported imaging will improve precision and reduce procedural errors.
- Hybrid operating rooms will accelerate adoption in regions upgrading surgical infrastructure.
- Next-generation fenestrated and branched grafts will gain traction for complex abdominal and thoracoabdominal repairs.
- Material innovations will improve graft durability and reduce long-term complications.
- Emerging markets will record faster growth due to rising healthcare investment and improved vascular care access.
- Long-term registries and post-market studies will guide device refinements and strengthen clinical confidence.
- Strategic partnerships between device makers and specialty centers will expand training and support broader procedural capability.




