Artificial Blood Vessels Market Overview:
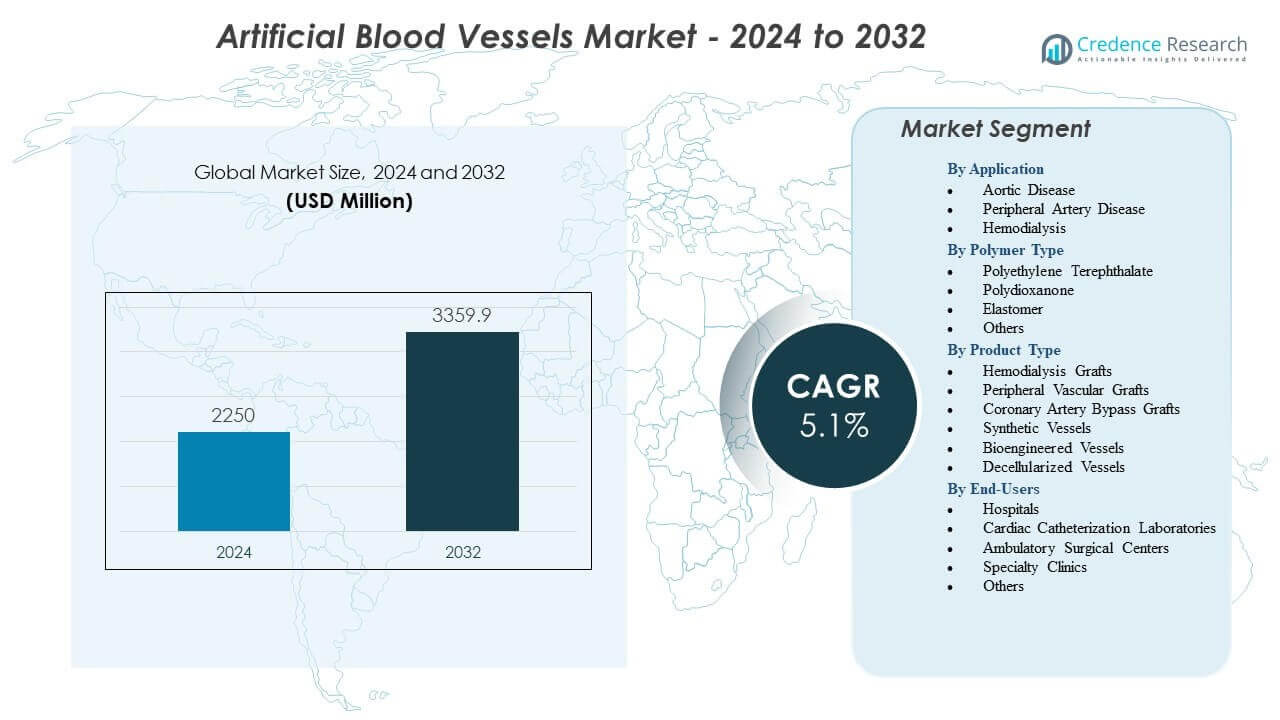
The Artificial Blood Vessels Market is projected to grow from USD 2,250 million in 2024 to an estimated USD 3,359.9 million by 2032, with a compound annual growth rate (CAGR) of 5.1% from 2024 to 2032.
| REPORT ATTRIBUTE |
DETAILS |
| Historical Period |
2020-2023 |
| Base Year |
2024 |
| Forecast Period |
2025-2032 |
| Artificial Blood Vessels Market Size 2024 |
USD 2,250 million |
| Artificial Blood Vessels Market, CAGR |
5.1% |
| Artificial Blood Vessels Market Size 2032 |
USD 3,359.9 million |
Market drivers reflect growing need for durable grafts in complex cardiovascular, peripheral artery, and dialysis-related procedures. Surgeons prefer engineered vessels that offer consistent performance, longer patency, and reduced infection risk. Innovations in biomaterials enhance flexibility, healing, and biocompatibility, supporting broader use in trauma repair and reconstructive vascular surgery. Dialysis access care increases demand for durable grafts due to rising chronic kidney disease cases. Regulatory approvals for next-generation solutions further strengthen clinical adoption. Collectively, these factors support steady expansion of the Artificial Blood Vessels Market.
Regional growth varies across major healthcare markets. North America leads due to advanced surgical infrastructure, high procedure rates, and early adoption of synthetic and bioengineered graft technologies. Europe follows with strong presence of vascular device manufacturers and structured clinical pathways that support long-term graft evaluation. Asia Pacific emerges rapidly as investments in cardiac and dialysis care rise across China, India, and Japan, supported by expanding hospital networks. Latin America and the Middle East show gradual improvement driven by growing access to specialized vascular treatment facilities and trained surgical teams.
Artificial Blood Vessels Market Insights:
- The Artificial Blood Vessels Market is projected to grow from USD 2,250 million in 2024 to USD 3,359.9 million by 2032 at a 5.1% CAGR, supported by rising demand for engineered graft solutions.
- Market growth is driven by increasing cardiovascular cases, higher need for durable grafts in complex surgeries, and wider adoption of advanced biomaterial technologies.
- Restraints include stringent regulatory requirements, limited long-term clinical data for newer bioengineered vessels, and high material development costs.
- North America leads the market due to strong surgical capacity and faster integration of advanced graft technologies across hospitals.
- Asia Pacific shows rapid expansion, while Europe maintains steady adoption; emerging regions gain traction as specialized vascular centers continue to expand.
Access crucial information at unmatched prices!
Request your sample report today & start making informed decisions powered by Credence Research Inc.!
Download Sample
Artificial Blood Vessels Market Drivers
Growing Burden of Cardiovascular Diseases Driving Demand for Advanced Vascular Replacement Solutions
Growing cardiovascular cases increase the need for artificial grafts in complex treatments. Hospitals prefer engineered vessels due to their strong performance in high-risk surgeries. It supports better outcomes when natural vessels fail to meet clinical needs. Surgeons choose synthetic options for stability during long procedures. Demand rises when aging populations report higher arterial blockages. New biomaterials extend durability in graft placements. The Artificial Blood Vessels Market benefits when providers adopt next-generation vascular tools. Research teams continue to refine polymers that offer strong healing responses.
Rise in Minimally Invasive Procedures Supporting Broader Adoption Across Surgical Settings
Minimally invasive procedures influence demand for engineered vascular grafts. Surgeons rely on smaller and flexible designs that reduce patient discomfort. It reduces recovery time when compared with traditional open surgeries. Hospitals upgrade operating units to support advanced vascular implants. Product developers focus on thin-wall grafts that maintain strong flow capacity. Growing patient preference for low-risk interventions boosts adoption. Training programs equip specialists with better device handling skills. The Artificial Blood Vessels Market gains traction when providers streamline treatment pathways.
- For instance, Medtronic’s Endurant II stent graft system demonstrated a 99% technical success rate in EVAR procedures and reduced operative time by up to 25% in post-market surveillance, supporting broader adoption.
Rapid Innovation in Polymer and Biocompatible Materials Enhancing Clinical Performance
Material innovation shapes new directions in vascular graft development. Manufacturers launch products that reduce immune response and support natural healing. It improves long-term graft stability across varied patient groups. Research centers test hybrid structures that blend strength with flexibility. Hospitals evaluate new designs to improve success rates in critical surgeries. Surgeons adopt biofriendly polymers when natural vessels fail. Development pipelines expand due to rising investment in biomaterials. The Artificial Blood Vessels Market observes steady progress in material science.
- For instance, Abbott’s Supera peripheral stent, constructed from interwoven nitinol wires, demonstrated a 12-month patency rate of 86.6% in the SUPERB trial.
Growing Use of Artificial Vessels in Trauma, Dialysis, and Complex Surgical Applications
Trauma care units depend on engineered vessels for emergency repairs. Dialysis centers require durable access grafts that reduce treatment disruption. It supports safer outcomes when patients undergo multiple interventions. Complex surgeries need synthetic vessels with controlled flow resistance. Critical care hospitals adopt grafts that reduce procedure delays. Device makers refine designs to match varied clinical pressures. Growth in vascular injuries increases the need for strong implant options. The Artificial Blood Vessels Market expands when specialized procedures rise globally.
Artificial Blood Vessels Market Trends
Adoption of Tissue-Engineered Vascular Grafts Shaping Next-Generation Clinical Solutions
Tissue-engineered grafts gain interest due to their natural healing behavior. Research groups test cell-seeded designs for improved integration. It creates pathways for new treatment standards across hospitals. Surgeons review these grafts for pediatric and adult repairs. Long-term studies help validate performance across age groups. Customizable graft structures support tailored patient care. Manufacturers explore automated platforms for large-scale production. The Artificial Blood Vessels Market reports rising interest in regenerative vascular technology.
- For instance, Humacyte’s Human Acellular Vessel (HAV) demonstrated 91% primary patency at six months in trauma studies and showed zero implant-related infections across Phase II data,
Shift Toward Customizable and Patient-Specific Vascular Implants Across Surgical Units
Customizable grafts support better clinical outcomes in complex cases. Surgeons value implants tailored to patient anatomy. It improves precision when graft placement demands high accuracy. Healthcare systems evaluate digital design tools for better sizing. Advances in fabrication enable rapid prototyping of graft models. Specialists use imaging data to plan ideal vessel geometry. Interest grows when patient-specific implants reduce revision rates. The Artificial Blood Vessels Market benefits from growth in personalized surgical care.
Growing Integration of Smart Materials and Functional Coatings in Modern Vascular Grafts
Smart materials improve graft flexibility and reduce infection risks. Developers add surface coatings to reduce clot formation. It improves flow behavior across varying pressure conditions. Hospitals adopt coated grafts to reduce complications after surgery. Research units design sensors for monitoring graft health. Functional materials support better resistance to mechanical stress. New formulations help reduce inflammation in early healing stages. The Artificial Blood Vessels Market reflects improved device performance through smart materials.
- For instance, W. L. Gore’s CBAS Heparin Surface uses covalent bonding that retains antithrombotic activity for the full device lifespan and demonstrated a 37% reduction in early thrombosis versus conventional PTFE grafts.
Expansion of Hybrid Vascular Designs Combining Synthetic Strength and Biological Adaptability
Hybrid grafts blend synthetic durability with biological compatibility. Developers design structures that match natural vessel movement. It improves stability when patients undergo high-strain activities. Surgeons rely on hybrid options for complex reconstructions. Clinical trials evaluate long-term safety and performance. Hospitals use hybrid solutions for trauma care and high-flow surgeries. Product portfolios expand with new composite formulations. The Artificial Blood Vessels Market tracks rising adoption of hybrid vascular systems.
Artificial Blood Vessels Market Challenges Analysis
Regulatory Complexity and Limited Long-Term Clinical Data Slowing Wider Adoption
Regulatory pathways require extensive testing and delay new product entry. Long-term performance data remains limited across several graft types. It reduces confidence when hospitals evaluate high-risk procedures. Manufacturers invest heavily in trials that require long study periods. Approval timelines vary across countries and slow global rollout. Surgeons hesitate when durability data does not meet expectations. The Artificial Blood Vessels Market faces slower penetration in conservative clinical environments. Market players work to improve evidence availability across patient groups.
High Material Costs and Production Difficulties Restricting Broad Market Access
Advanced biomaterials increase manufacturing costs for developers. Complex production steps limit large-scale output. It challenges smaller firms that lack adequate resources. Hospitals hesitate when procurement budgets face pressure. Price sensitivity in emerging markets restricts adoption. Scaling up production without losing quality remains difficult. The Artificial Blood Vessels Market encounters barriers when cost-effective solutions are limited. Industry stakeholders explore new methods to simplify fabrication.
Artificial Blood Vessels Market Opportunities
Rising Demand in Emerging Healthcare Systems Creating Expansion Pathways for Vascular Solutions
Emerging regions increase investment in cardiac and trauma care. Hospitals upgrade surgical units to support complex vascular procedures. It promotes adoption when physicians gain access to modern graft options. Governments strengthen training for vascular specialists. Patient awareness of advanced treatments rises steadily. Infrastructure improvements expand procedure volumes. The Artificial Blood Vessels Market gains new prospects when developing nations boost healthcare spending. Manufacturers explore partnerships to improve regional distribution.
Rapid Progress in Regenerative Medicine and Biofabrication Opening New Product Pipelines
Regenerative medicine supports breakthroughs in tissue-based vascular products. Biofabrication tools create grafts with controlled shape and strength. It improves design flexibility for varied clinical needs. Research centers test engineered vessels for next-generation therapies. Hospitals monitor early success from advanced regenerative trials. Investments grow in bioprinting platforms that support precision manufacturing. The Artificial Blood Vessels Market gains strong opportunity from the rise of living graft constructs. Industry teams explore scalable models for commercial production.
Artificial Blood Vessels Market Segmentation Analysis:
By Application
Aortic disease remains a major focus due to the complexity of vascular repairs that require durable grafts. Surgeons prefer engineered options that maintain strength under high pressure. Peripheral artery disease drives steady demand where flexible vessels support improved limb outcomes. Hemodialysis access care needs grafts that tolerate repeated use without failure. It supports stable growth in the Artificial Blood Vessels Market due to rising chronic kidney conditions. Hospitals evaluate each option to match varied clinical risks across patient groups.
- For instance, W. L. Gore’s TAG Thoracic Branch Endoprosthesis demonstrated 98% patency at 12 months in clinical evaluations
By Polymer Type
Polyethylene terephthalate maintains strong use due to its durability and long clinical record. Polydioxanone offers biodegradability that supports interest in temporary scaffolds. Elastomer materials improve flexibility where vessel movement demands adaptive performance. Other polymers gain attention when surgeons look for grafts that balance flow control and healing response. It encourages steady product diversification in the Artificial Blood Vessels Market as developers refine material blends for broader applications.
By Product Type
Hemodialysis grafts hold steady demand due to the rising dialysis population. Peripheral vascular grafts support limb-saving procedures that rely on strong blood flow. Coronary artery bypass grafts remain vital in cardiac surgery where reliability defines performance. Synthetic vessels dominate due to proven strength, while bioengineered vessels gain momentum for their natural integration. Decellularized vessels attract interest for reduced immune response. It strengthens product variety in the Artificial Blood Vessels Market.
- For instance, CryoLife’s decellularization process removes over 99% of donor cellular material to reduce immune response.
By End-Users
Hospitals lead due to high procedure volume and advanced surgical capability. Cardiac catheterization laboratories support growing use of grafts in interventional care. Ambulatory surgical centers expand adoption with improved vascular tools for day-care procedures. Specialty clinics focus on complex vascular disorders that need specialized grafts. Other facilities integrate these solutions to support broader treatment access. It reinforces stable demand across the Artificial Blood Vessels Market.
Segmentation:
By Application
- Aortic Disease
- Peripheral Artery Disease
- Hemodialysis
By Polymer Type
- Polyethylene Terephthalate
- Polydioxanone
- Elastomer
- Others
By Product Type
- Hemodialysis Grafts
- Peripheral Vascular Grafts
- Coronary Artery Bypass Grafts
- Synthetic Vessels
- Bioengineered Vessels
- Decellularized Vessels
By End-Users
- Hospitals
- Cardiac Catheterization Laboratories
- Ambulatory Surgical Centers
- Specialty Clinics
- Others
By Region
- North America
- Europe
- Germany
- France
- U.K.
- Italy
- Spain
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- South-east Asia
- Rest of Asia Pacific
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Middle East & Africa
- GCC Countries
- South Africa
- Rest of the Middle East and Africa
Regional Analysis:
North America holds the largest share of the Artificial Blood Vessels Market at nearly 40%, supported by strong surgical volumes and widespread acceptance of advanced graft technologies. Hospitals invest heavily in high-performance vascular implants for cardiac, peripheral, and dialysis procedures. It benefits from mature healthcare systems that support rapid integration of new biomaterials. The region maintains strong research activity that accelerates product testing and clinical evaluations. Leading companies operate major facilities that enhance distribution speed and device availability. Regulatory clarity also helps providers adopt safer graft options.
Europe accounts for roughly 30% of the market and shows steady demand for artificial grafts across cardiac and vascular centers. Countries adopt engineered vessels to reduce procedure complications and improve long-term durability. It benefits from structured clinical programs that track outcomes and refine usage guidelines. Strong presence of established manufacturers supports continuous innovation in both synthetic and bioengineered vessels. Providers integrate these solutions into routine practice when natural vessels are unsuitable. Growing focus on minimally invasive vascular repair strengthens regional demand.
Asia Pacific holds nearly 22% share and grows faster than other regions due to rising cases of cardiovascular and kidney disorders. Large populations in China and India create high demand for vascular implants in both public and private hospitals. It expands further when nations invest in specialized cardiac and dialysis infrastructure. Surgeons adopt artificial grafts to address rising amputation and bypass procedure needs. Improvements in training and product availability support broader usage across urban centers. Latin America and Middle East & Africa together hold the remaining 8%, supported by gradual improvement in specialty care.
Shape Your Report to Specific Countries or Regions & Enjoy 30% Off!
Key Player Analysis:
- Braun Melsungen
- Becton Dickinson (BD)
- Terumo Corporation
- L. Gore & Associates
- LeMaitre Vascular
- Cook Medical
- Medtronic
- Humacyte Inc.
- Jotec GmbH
- Boston Scientific
Competitive Analysis:
Competition in the Artificial Blood Vessels Market centers on material innovation, long-term performance, and product differentiation across surgical applications. Companies pursue stronger biocompatible polymers that reduce complications and improve healing behavior. It drives investment in synthetic and biological graft technologies that support varied clinical needs. Established players defend their position through broad product portfolios and strong clinical evidence. Emerging firms introduce bioengineered vessels that target next-generation vascular repair. Competitive pressure increases when hospitals demand durable grafts for cardiac, dialysis, and peripheral artery procedures. Firms build strategic partnerships to improve global reach and accelerate regulatory pathways. Market competition continues to intensify as developers refine performance data and pursue advanced manufacturing methods.
Recent Developments:
- In February 2026, Medtronic plc announced its intent to acquire CathWorks, following a 2022 strategic partnership, to bolster its interventional cardiology portfolio with AI-powered coronary physiology tools that support vascular interventions including artificial blood vessel contexts.
- In February 2025, Humacyte launched commercial shipments of Symvess following FDA batch release review, with product sales reaching $703,000 by Q3 2025 across 25 Value Analysis Committee approvals covering 92 civilian hospitals.
- In February 2025, Teleflex Incorporated announced a definitive agreement to acquire substantially all of BIOTRONIK’s Vascular Intervention business for approximately €760 million, adding drug-coated balloons, drug-eluting stents, and other vascular intervention devices relevant to artificial blood vessel applications.
Report Coverage:
The research report offers an in-depth analysis based on Application, Polymer Type, End-Users, Product Type, and region. It details leading market players, providing an overview of their business, product offerings, investments, revenue streams, and key applications. Additionally, the report includes insights into the competitive environment, SWOT analysis, current market trends, as well as the primary drivers and constraints. Furthermore, it discusses various factors that have driven market expansion in recent years. The report also explores market dynamics, regulatory scenarios, and technological advancements that are shaping the industry. It assesses the impact of external factors and global economic changes on market growth. Lastly, it provides strategic recommendations for new entrants and established companies to navigate the complexities of the market.
Future Outlook:
- Rising demand for engineered grafts will strengthen long-term adoption across cardiac, peripheral, and dialysis procedures.
- Material innovation will support new designs that mimic natural vessels and improve clinical confidence.
- Bioengineered and decellularized vessels will gain traction due to stronger healing behavior in complex surgeries.
- Hybrid grafts will expand use in trauma care and reconstructive procedures that require flexible structures.
- Digital planning tools will improve sizing accuracy and support better patient-specific graft selection.
- Hospitals will integrate advanced grafts as minimally invasive procedures become more common across regions.
- Regulatory pathways will support broader adoption when manufacturers secure stronger clinical data.
- Growth in specialized cardiac and vascular centers across emerging markets will widen global access.
- Industry partnerships will accelerate innovation pipelines and expand geographic distribution networks.
- The Artificial Blood Vessels Market will benefit from rising clinical focus on durability, safety, and long-term treatment outcomes.




