Market Overview:
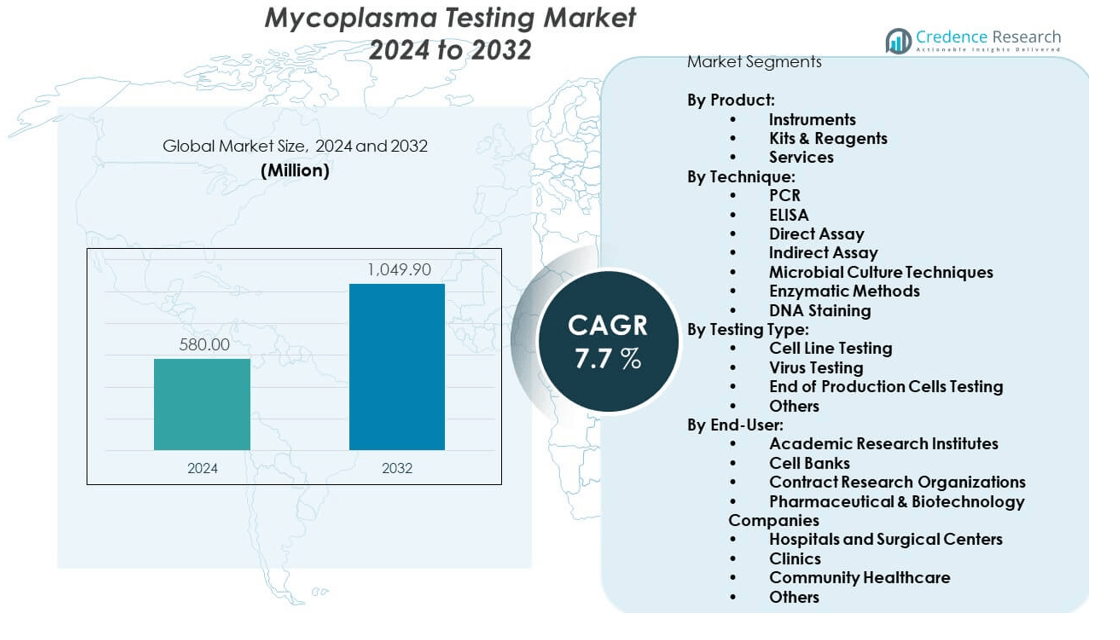
The Mycoplasma testing market was valued at USD 580 million in 2024 and is projected to reach USD 1,049.9 million by 2032, expanding at a CAGR of 7.7% during the forecast period.
| REPORT ATTRIBUTE |
DETAILS |
| Historical Period |
2020-2023 |
| Base Year |
2024 |
| Forecast Period |
2025-2032 |
| Mycoplasma Testing Market Size 2024 |
USD 580 million |
| Mycoplasma Testing Market, CAGR |
7.7% |
| Mycoplasma Testing Market Size 2032 |
USD 1,049.9 million |
Market growth is fueled by the rising prevalence of mycoplasma contamination in cell culture-based research and the need to ensure quality control in biologics manufacturing. Increasing adoption of advanced detection techniques, such as PCR-based assays and kits, is improving accuracy and speed, reducing operational risks for biopharmaceutical firms. Stringent regulatory guidelines issued by agencies, including the FDA and EMA, are compelling manufacturers to adopt routine testing, while expanding R&D activities in cell and gene therapies further accelerate adoption.
From a geographic perspective, North America leads the mycoplasma testing market due to its strong biopharmaceutical industry, advanced R&D infrastructure, and strict regulatory oversight. Europe follows closely, supported by robust biotechnology research and a well-established healthcare framework. Asia-Pacific is emerging as the fastest-growing region, fueled by expanding pharmaceutical manufacturing in countries such as China and India, rising healthcare investments, and increasing adoption of biologics. Meanwhile, Latin America and the Middle East & Africa show gradual adoption, with growing biotechnology sectors and improving regulatory frameworks supporting future opportunities.
Market Insights:
- The Mycoplasma testing market was valued at USD 580 million in 2024 and is projected to reach USD 1,049.9 million by 2032, growing at a CAGR of 7.7%.
- Demand is rising due to increased biologics and biosimilar production, where contamination control is critical.
- Regulatory requirements by agencies such as the FDA and EMA drive adoption of validated testing methods.
- High costs of advanced testing technologies and infrastructure limit adoption in smaller laboratories.
- North America leads the market, supported by strong biopharmaceutical infrastructure and strict compliance standards.
- Europe follows with robust biotechnology research and harmonized safety regulations across member countries.
- Asia-Pacific is the fastest-growing region, fueled by pharmaceutical manufacturing expansion in China and India.
Access crucial information at unmatched prices!
Request your sample report today & start making informed decisions powered by Credence Research Inc.!
Download Sample
Market Drivers:
Growing Reliance on Cell-Based Research and Biopharmaceutical Production Increases Testing Demand:
The global rise in cell-based research, biologics, and biosimilars has intensified the need for strict contamination control. The Mycoplasma testing market benefits from the fact that undetected contamination can compromise drug development and safety. Researchers and manufacturers integrate testing into routine workflows to safeguard product quality. It strengthens compliance with international guidelines and minimizes production losses. Strong investments in biopharmaceutical production facilities fuel demand for reliable testing kits. It also aligns with the increasing global focus on advanced therapeutics. The growing adoption of vaccines, monoclonal antibodies, and regenerative therapies makes contamination detection essential. Rising industry awareness further accelerates adoption.
- For instance, Thermo Fisher Scientific’s MycoSEQ Plus Mycoplasma Detection Kit achieves a sensitivity level below 10 genome copies per milliliter (GC/mL), meeting regulatory requirements for detection in complex bioproduction samples.
Regulatory Mandates from Global Agencies Drive Adoption of Testing Technologies:
International regulatory bodies, including the FDA and EMA, have established strict requirements for mycoplasma detection. The Mycoplasma testing market responds directly to these regulations, with mandatory testing in biologics and biosimilar production. Manufacturers adopt PCR-based, ELISA, and culture methods to ensure compliance. It prevents drug recalls and supports product approvals across diverse markets. The role of testing expands further with global harmonization of safety standards. Strong oversight in the United States and Europe continues to lead adoption trends. Emerging economies are also tightening regulations to align with international quality benchmarks. This environment creates consistent demand for validated testing solutions worldwide.
- For instance, Roche Diagnostics’ MycoTOOL real-time PCR mycoplasma test delivers reliable detection results within five hours, replacing traditional culture-based methods and enabling faster regulatory submissions.
Technological Advancements Improve Accuracy, Speed, and Sensitivity of Detection:
Advances in molecular biology and diagnostic technology are reshaping contamination detection practices. The Mycoplasma testing market gains momentum from innovations like real-time PCR, luminescence assays, and next-generation sequencing. These methods provide faster turnaround times and superior sensitivity. It reduces the risks associated with false negatives and improves decision-making. Automated testing systems help laboratories scale operations while reducing human error. Innovation also broadens applications in both small laboratories and large manufacturing plants. The growing trend toward miniaturized, user-friendly kits enables wider adoption across regions. It ensures laboratories and manufacturers adopt modern solutions to keep pace with industry demands.
Expansion of R&D Activities in Advanced Therapies Fuels Testing Adoption:
The development of cell and gene therapies continues to transform the healthcare industry. The Mycoplasma testing market supports these innovations by ensuring safe, contamination-free processes. Contamination can compromise high-value cell lines and delay approvals. It becomes essential for laboratories to maintain strict quality controls. Research organizations are increasing investments in advanced cell culture facilities. Pharmaceutical and biotechnology companies actively expand pipelines of gene-based therapies. Academic institutes and contract research organizations also accelerate R&D spending. It creates sustained demand for high-quality detection technologies to safeguard therapeutic innovation.
Market Trends:
Integration of Artificial Intelligence and Automation in Testing Platforms:
Automation is reshaping laboratory practices, and the Mycoplasma testing market is no exception. Advanced platforms integrate AI-driven algorithms to improve sensitivity and reduce turnaround times. Automated workflows minimize manual interventions and reduce operator variability. It enables consistent results and faster regulatory submissions. Robotics further streamline culture testing in high-throughput environments. Pharmaceutical companies deploy automation to handle rising sample volumes efficiently. AI-enhanced analytics provide predictive contamination insights for preventive action. It strengthens industry confidence in modern solutions and sets a new trend in advanced testing.
- For example, Thermo Fisher Scientific’s AutoMate Express instrument automates nucleic acid extraction for the MycoSEQ Mycoplasma detection workflow, delivering DNA and RNA recovery in under two hours with minimal hands-on time, boosting lab efficiency.
Growing Use of Mycoplasma Detection in Contract Research and Manufacturing:
Contract research organizations (CROs) and contract development and manufacturing organizations (CDMOs) are expanding services. The Mycoplasma testing market gains from this outsourcing trend, as companies delegate quality control to specialized providers. CROs adopt advanced kits to meet global compliance needs. It reduces the financial burden of maintaining in-house laboratories for smaller firms. The role of CDMOs expands with biopharmaceutical growth and outsourcing of R&D. Flexible service models allow tailored testing solutions for diverse clients. High-volume testing capabilities make CROs strategic partners in global supply chains. It reinforces their position as key stakeholders in advancing testing solutions worldwide.
- For instance, Bio-Rad Laboratories leverages its Droplet Digital PCR technology to enhance the sensitivity and specificity of mycoplasma detection during biotherapeutic development, processing high sample volumes with precise molecular quantification.
Shift Toward Point-of-Use and Rapid Detection Kits Across Facilities:
The demand for rapid and on-site testing solutions is increasing globally. The Mycoplasma testing market aligns with this shift by introducing portable, user-friendly kits. These tools allow laboratories and manufacturers to confirm contamination risks quickly. It minimizes downtime in production facilities and reduces financial risks. Rapid kits are adopted in clinical trials, vaccine production, and smaller laboratories. Pharmaceutical firms prioritize faster turnaround to streamline decision-making. The global demand for scalable and reliable point-of-use solutions continues to rise. It highlights a strong trend in decentralizing contamination detection across industries.
Adoption of Cloud-Connected Testing Platforms and Digital Recordkeeping:
Digital transformation extends into laboratory testing and monitoring environments. The Mycoplasma testing market is adopting cloud-based platforms to enhance traceability and data management. These systems provide real-time access to testing results across global facilities. It allows regulatory bodies and manufacturers to ensure compliance remotely. Digital records improve transparency and reduce the risk of documentation errors. Cloud integration also enables collaborative research in multinational projects. Pharmaceutical and biotech companies benefit from secure, centralized platforms for audits. It signals a trend toward digitization of testing records as part of industry modernization.
Market Challenges Analysis:
High Costs of Advanced Testing and Infrastructure Limit Wider Adoption:
The cost of mycoplasma detection technologies poses challenges for smaller laboratories and manufacturers. The Mycoplasma testing market requires investments in advanced PCR systems, culture facilities, and skilled personnel. It creates financial barriers for startups, academic centers, and small-scale biopharma firms. High maintenance costs of infrastructure also add pressure. Limited budgets often restrict the adoption of validated kits in resource-constrained settings. Technology transfer remains complex, requiring ongoing training for laboratory professionals. This disparity creates an uneven adoption landscape between developed and emerging markets. It reduces overall accessibility and hinders market penetration in cost-sensitive regions.
Complexity of Testing Methods and Risk of False Results Create Operational Barriers:
Testing methods such as culture assays and PCR require precision and technical expertise. The Mycoplasma testing market faces challenges due to variability in results caused by operator error. It increases risks of false positives or negatives, leading to delays in drug approvals. Inadequate standardization across testing platforms creates further difficulties. Laboratories struggle to balance accuracy, speed, and cost in routine operations. Lack of harmonized global protocols complicates compliance efforts for multinational firms. Training gaps also slow down technology adoption in certain regions. It highlights the ongoing need for industry-wide consistency and skill development to reduce risks.
Market Opportunities:
Expanding Biopharmaceutical Production in Emerging Economies Creates Growth Potential:
The global rise in pharmaceutical manufacturing hubs is reshaping industry dynamics. The Mycoplasma testing market benefits as emerging economies invest heavily in biologics and biosimilar facilities. It creates strong demand for validated contamination detection methods. Governments support infrastructure development to attract international biopharmaceutical firms. Increasing R&D spending in countries such as China, India, and South Korea strengthens adoption. Academic institutions also expand capacity for research in advanced cell therapies. It positions emerging economies as critical growth engines for modern testing solutions.
Rising Adoption of Personalized Medicine and Regenerative Therapies Boosts Demand:
Healthcare systems are shifting toward precision therapies, requiring stringent contamination controls. The Mycoplasma testing market gains opportunities from regenerative medicine, stem cell research, and gene therapies. It becomes essential for laboratories to ensure contamination-free cell cultures in clinical use. Strong clinical pipelines in oncology and rare diseases further expand testing requirements. Biotech startups invest in portable and cost-effective kits to support personalized care. Partnerships between global firms and regional laboratories accelerate availability of advanced tools. It strengthens the long-term growth prospects of testing in advanced therapy applications.
Market Segmentation Analysis:
By Product
The Mycoplasma testing market by product is divided into instruments, kits & reagents, and services. Kits & reagents dominate the segment, driven by their recurring use in routine laboratory testing. Instruments contribute steadily, supported by advancements in automated detection technologies. Services are expanding as pharmaceutical companies and research institutes increasingly outsource testing needs to specialized providers for efficiency and compliance.
- For example, InvivoGen’s PlasmoTest offers a visual, colorimetric detection kit with a sensitivity threshold reaching fewer than 10 CFU/mL, optimized for routine culture testing in labs worldwide.
By Technique
The market by technique includes PCR, ELISA, direct assay, indirect assay, microbial culture techniques, enzymatic methods, and DNA staining. PCR holds the largest share due to its high sensitivity, accuracy, and faster turnaround time. ELISA and microbial culture techniques remain relevant in regulatory validation. Direct and indirect assays, alongside enzymatic and DNA staining methods, continue to support specific research and niche applications. It shows that laboratories adopt multiple techniques depending on regulatory standards and project needs.
- For instance, Thermo Fisher Scientific’s MycoSEQ system supports both probe-based and dye-based PCR assays, consistently achieving detection limits below 10 CFU/mL and validated by in-house and third-party studies.
By Testing Type
The market by testing type covers cell line testing, virus testing, end of production cells testing, and others. Cell line testing dominates owing to its critical role in biologics and biosimilar production. Virus testing has strong relevance in vaccine development and gene therapy pipelines. End of production cells testing ensures compliance and product safety in large-scale manufacturing. Other testing applications extend support to broader diagnostic and laboratory research.
By End-User
The market by end-user includes academic research institutes, cell banks, CROs, pharmaceutical & biotechnology companies, hospitals and surgical centers, clinics, community healthcare, and others. Pharmaceutical and biotechnology companies lead demand due to their large-scale biologics production and strict compliance requirements. CROs and academic institutes expand adoption through outsourced testing and early-stage research. Hospitals, clinics, and community healthcare add to the base, supported by rising applications of cell-based therapies. It demonstrates wide adoption across both research and healthcare ecosystems.
Segmentation:
By Product:
- Instruments
- Kits & Reagents
- Services
By Technique:
- PCR
- ELISA
- Direct Assay
- Indirect Assay
- Microbial Culture Techniques
- Enzymatic Methods
- DNA Staining
By Testing Type:
- Cell Line Testing
- Virus Testing
- End of Production Cells Testing
- Others
By End-User:
- Academic Research Institutes
- Cell Banks
- Contract Research Organizations (CROs)
- Pharmaceutical & Biotechnology Companies
- Hospitals and Surgical Centers
- Clinics
- Community Healthcare
- Others
By Region
- North America
- Europe
- Germany
- France
- UK.
- Italy
- Spain
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- South-east Asia
- Rest of Asia Pacific
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Middle East & Africa
- GCC Countries
- South Africa
- The Rest of the Middle East and Africa
Regional Analysis:
North America
North America holds the largest share of the Mycoplasma testing market, accounting for over 40% of global revenue. The region benefits from advanced biopharmaceutical infrastructure, strong regulatory oversight, and a high volume of biologics and vaccine production. The presence of leading pharmaceutical companies and specialized CROs further strengthens its dominance. It is supported by government-backed research funding and a well-established healthcare framework. High adoption of advanced techniques, such as PCR-based kits and automated platforms, drives continuous growth. The United States leads the regional market, with Canada contributing through expanding biotechnology investments.
Europe
Europe represents nearly 30% of the global market share, supported by a strong biotechnology sector and harmonized safety regulations. Countries such as Germany, the United Kingdom, and France are leading contributors, supported by extensive R&D activity. It benefits from the region’s universal healthcare systems and emphasis on quality control in pharmaceutical manufacturing. Regulatory agencies enforce strict contamination detection guidelines, which encourage consistent adoption of validated testing technologies. Academic and clinical research facilities further drive demand across multiple applications. The region continues to expand capacity in advanced therapies, strengthening long-term demand for mycoplasma testing solutions.
Asia-Pacific
Asia-Pacific captures around 20% of the Mycoplasma testing market and is the fastest-growing region. Expanding pharmaceutical and biotechnology manufacturing hubs in China, India, and South Korea drive adoption of contamination detection solutions. It is supported by rising healthcare expenditure, government-led infrastructure investments, and a growing pipeline of biologics and biosimilars. Regional players are increasingly collaborating with international firms to enhance testing capabilities. Growing academic research activities and a shift toward advanced therapies add further momentum. The region’s rapid industrial growth and emphasis on innovation make it a critical future growth driver for the global market.
Shape Your Report to Specific Countries or Regions & Enjoy 30% Off!
Key Player Analysis:
- Thermo Fisher Scientific Inc.
- Roche Diagnostics
- Bio-Rad Laboratories
- GenBio
- InvivoGen
- Charles River Laboratories International
- Lonza Group Ltd.
- Merck KGaA
- American Type Culture Collection (ATCC)
- Bionique Testing Laboratories Inc.
- PromoCell GmbH
- Biological Industries
- Eurofins Scientific
- Sartorius AG
Competitive Analysis:
The Mycoplasma testing market is characterized by the presence of leading global players and regional specialists competing across products and services. Companies such as Thermo Fisher Scientific, Roche Diagnostics, Bio-Rad Laboratories, Merck KGaA, and Lonza Group Ltd. dominate with advanced testing kits, reagents, and instruments. It is highly competitive due to continuous R&D investments and growing demand for rapid detection methods. Firms expand their offerings through acquisitions, partnerships, and service innovations to strengthen global reach. Specialized providers like Charles River Laboratories and Bionique Testing Laboratories focus on outsourcing solutions, reinforcing their niche presence. The market is driven by technological advancements and strict regulatory standards, creating an environment where quality, reliability, and compliance remain key differentiators.
Recent Developments:
- In February 2024, Thermo Fisher Scientific expanded its GMP laboratory service offerings to include biosafety testing such as mycoplasma testing at its Middleton, Wisconsin facility. This addition aims to ensure biopharmaceutical products are contaminant-free, supporting safer medicines for patients.
- In July 2024, Bio-Rad Laboratories showcased advances with its Droplet Digital PCR solutions that enhance precision monitoring of molecular residual disease in cancer patients, indirectly supporting mycoplasma testing research and biopharmaceutical development through improved molecular diagnostics technologies.
- In October 2024, InvivoGen launched the PlasmoTest™, a cellular assay for visual, colorimetric detection of mycoplasma contamination in cell cultures. This test offers a quick and reliable method for routine mycoplasma detection with high sensitivity and ease of use.
Report Coverage:
The research report offers an in-depth analysis based on product, technique, testing type, and end-user. It details leading market players, providing an overview of their business, product offerings, investments, revenue streams, and key applications. Additionally, the report includes insights into the competitive environment, SWOT analysis, current market trends, as well as the primary drivers and constraints. Furthermore, it discusses various factors that have driven market expansion in recent years. The report also explores market dynamics, regulatory scenarios, and technological advancements that are shaping the industry. It assesses the impact of external factors and global economic changes on market growth. Lastly, it provides strategic recommendations for new entrants and established companies to navigate the complexities of the market.
Future Outlook:
- Growing adoption of PCR-based technologies will enhance accuracy and reduce turnaround times.
- Rising demand from cell and gene therapy development will boost adoption across laboratories.
- Outsourcing of mycoplasma testing to CROs and CDMOs will expand further.
- AI-driven automation in testing platforms will improve efficiency and reduce errors.
- Expansion of biopharmaceutical manufacturing in Asia-Pacific will create significant opportunities.
- Increasing collaborations between global firms and regional players will drive technology access.
- Regulatory tightening worldwide will ensure consistent adoption of validated methods.
- Portable and rapid kits will see rising demand in decentralized testing environments.
- Digital recordkeeping and cloud-based testing platforms will transform compliance monitoring.
- Continuous R&D investments will sustain innovation in novel detection techniques.




