Alexipharmic Drugs Market Overview:
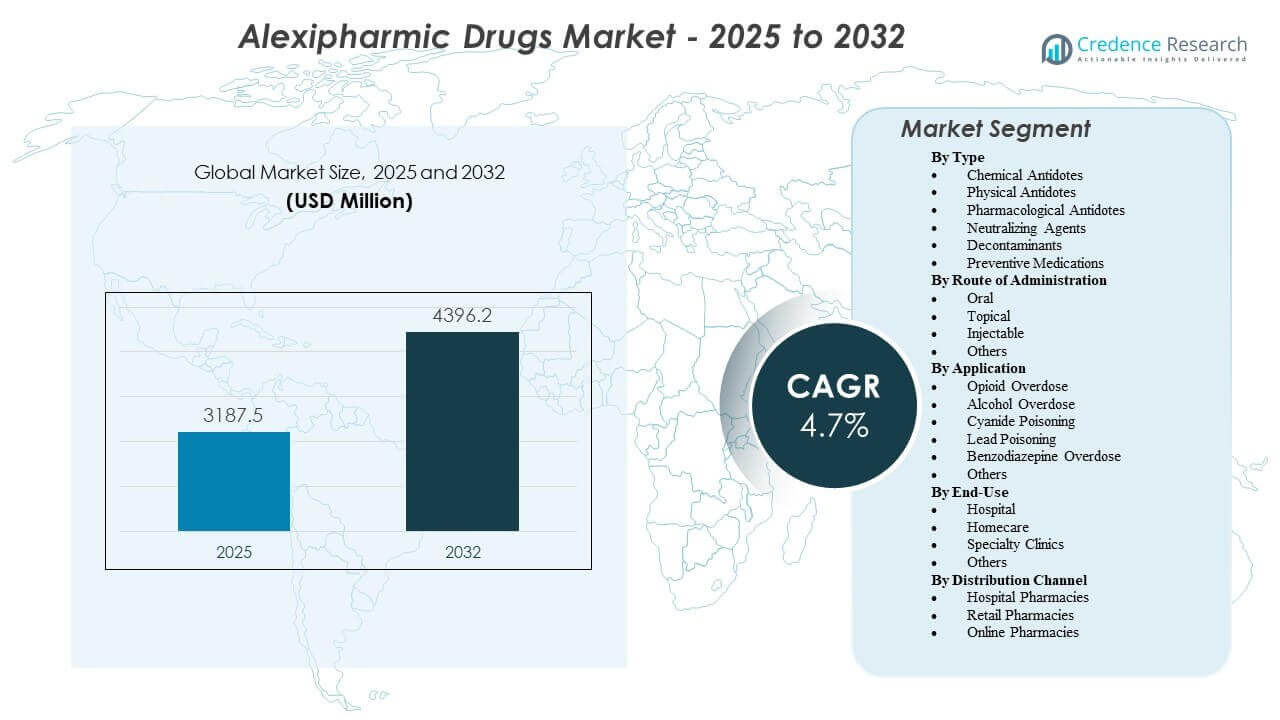
The Alexipharmic Drugs Market is projected to grow from USD 3,187.5 million in 2025 to an estimated USD 4,396.2 million by 2032, with a compound annual growth rate (CAGR) of 4.7% from 2025 to 2032.
| RT ATTRIBUTE |
DETAILS |
| Historical Period |
2020-2023 |
| Base Year |
2024 |
| Forecast Period |
2025-2032 |
| Alexipharmic Drugs Market Size 2025 |
USD 3,187.5 million |
| Alexipharmic Drugs Market, CAGR |
4.7% |
| Alexipharmic Drugs Market Size 2032 |
USD 4,396.2 million |
Alexipharmic Drugs Market Insights:
- Rising cases of opioid overdoses, alcohol toxicity, chemical exposure, and heavy metal poisoning are significantly driving demand for fast-acting antidotes and emergency detoxification treatments worldwide.
- Government initiatives promoting wider availability of lifesaving drugs such as naloxone, along with advancements in injectable and combination formulations, are accelerating product adoption across hospitals and emergency care settings.
- Limited awareness in low-income regions, stringent regulatory approvals, and the high cost associated with certain specialized antidotes act as key restraints to faster market penetration.
- North America leads the market due to strong emergency healthcare infrastructure and high overdose incidence, while Europe maintains steady growth; Asia-Pacific is emerging rapidly with expanding healthcare access in China and India, and Latin America and the Middle East & Africa are witnessing gradual development supported by improving medical facilities.
Alexipharmic Drugs Market Drivers
Growing Incidence of Toxic Exposures and Increased Reliance on Rapid Antidote Therapies
Rising exposure to industrial chemicals and household toxins strengthens demand for fast-acting counteragents across the Alexipharmic Drugs Market. Emergency centers treat higher poisoning cases that require structured detoxification. It supports wider adoption of targeted antidotes across critical care units. Hospitals expand poison response programs to manage emergency loads with better accuracy. Strong emphasis on early reversal pushes healthcare teams to invest in updated protocols. Awareness programs help communities understand poisoning symptoms with more clarity. Clinicians depend on evidence-based regimens that improve recovery speed. The Alexipharmic Drugs Market benefits from structured readiness across emergency departments.
Access crucial information at unmatched prices!
Request your sample report today & start making informed decisions powered by Credence Research Inc.!
Download Sample
Expansion of Pharmaceutical Innovation for Broad-Spectrum and Toxin-Specific Antidotes
Drug developers invest in advanced molecules that neutralize varied toxins with higher precision. New discovery platforms support faster identification of antidote candidates. It improves response capability during complex poisoning events. Firms enhance formulation stability to extend usability in resource-limited environments. Hospitals adopt advanced delivery systems that support faster onset. Research groups work on mechanism-specific solutions to improve patient outcomes. Strong funding support encourages deeper R&D pipelines focused on toxicology. The Alexipharmic Drugs Market gains steady momentum from expanding innovation streams.
- For instance, Indivior PLC recently launched OPVEE® (nalmefene), which utilizes a high-affinity mu-opioid receptor antagonist to provide a rapid onset of action and a long 11.4-hour half-life, specifically engineered to counteract the prolonged effects of synthetic opioids like fentanyl which traditional antidotes may fail to suppress.
Strengthening Government Mandates for Poison Preparedness and Emergency Stockpiles
Regulatory bodies implement mandates that ensure hospitals maintain essential antidote inventories. Policy upgrades aim to reduce treatment gaps during toxin-related emergencies. It drives coordinated purchasing across public health networks. Emergency teams follow standardized response plans that improve patient safety. Strong enforcement encourages transparent inventory audits. National poison centers promote structured training for healthcare staff. Governments highlight the importance of antidotes in high-risk industrial zones. The Alexipharmic Drugs Market aligns with broader public health safety plans.
- For instance, the U.S. Strategic National Stockpile maintains federally controlled antidotes such as pralidoxime and cyanide kits, supported by recurring procurement contracts to guarantee national emergency readiness.
Rising Awareness of Occupational Hazards and Industrial Chemical Risks
Manufacturing zones report higher exposure to chemicals used in metalworking, agriculture, and cleaning operations. Safety teams educate workers on early symptoms linked to toxic inhalation or ingestion. It supports faster reporting that reduces medical complications. Employers invest in medical preparedness programs that include antidote access. Hazard control guidelines push adoption of emergency kits in factories. Training programs help teams respond with greater confidence during accidents. Worker protection agencies endorse structured antidote availability. The Alexipharmic Drugs Market grows with rising industrial safety standards.
Alexipharmic Drugs Market Trends
Integration of Digital Poison Management Platforms and Real-Time Treatment Guidance Tools
Hospitals deploy digital decision systems that guide clinicians during toxin exposure events. AI-supported tools review symptoms to recommend antidote choices with better accuracy. It helps reduce errors linked to delayed treatment. Poison centers use teleconsultation models to support remote clinicians. Health systems share digital playbooks to unify emergency responses. Automated alerts speed up antidote identification in complex cases. Data integration strengthens national monitoring for toxic exposures. The Alexipharmic Drugs Market gains from improved clinical intelligence adoption.
- For instance, Baebies, Inc. integrated its FINDER® platform with digital health networks to provide rapid phenotyping of toxic enzyme deficiencies, enabling clinicians to receive actionable diagnostic results in less than 15 minutes from a single drop of blood to guide precise antidote dosing.
Shift Toward Ready-to-Use Formulations and Rapid-Onset Delivery Mechanisms
Manufacturers develop pre-filled syringes and autoinjectors for faster response times. Ready formulations reduce preparation time in emergencies. It improves safety across high-pressure environments. Drug firms optimize bioavailability for quicker toxin neutralization. Compact packaging supports wider use in ambulances and field units. Hospitals prefer formats that reduce dosing errors. Prehospital teams benefit from stable shelf-life designs. The Alexipharmic Drugs Market moves toward simplified administration pathways.
Growing Pipeline of Targeted Molecules Designed for Niche and Rare Poisoning Cases
Research teams focus on antidotes for less common but severe toxin exposures. Development programs explore new mechanisms for venoms, heavy metals, and synthetic compounds. It drives diversification of therapeutic classes. Toxicology labs collaborate with biotech firms to accelerate molecule validation. Grants support rare poisoning research with stronger urgency. Regulators fast-track niche antidotes with high clinical need. Hospitals prepare for unconventional threats with more structured protocols. The Alexipharmic Drugs Market expands its therapeutic breadth through specialized innovation.
- For instance, Opiant Pharmaceuticals (acquired by Indivior) developed OPVEE®, a nalmefene nasal spray that demonstrated a median 15-minute Tmax in FDA-reviewed clinical studies, along with an extended 11-hour half-life that supports sustained reversal of potent synthetic fentanyl analogues that outlast standard naloxone.
Rising Adoption of Collaborative Poison Surveillance Networks Across Regions
Public health agencies link monitoring systems to detect poisoning trends quickly. Shared databases improve situational awareness for hospitals. It strengthens coordinated intervention in multi-case events. Poison centers exchange insights to refine national response plans. Cross-border programs support knowledge sharing for emerging threats. Training modules standardize best practices. Surveillance upgrades help identify hazardous materials with higher accuracy. The Alexipharmic Drugs Market benefits from stronger global partnerships.
Alexipharmic Drugs Market Challenges Analysis
Complex Regulatory Pathways and High Evidence Requirements for Antidote Approvals
Regulators demand robust safety data before approving antidotes due to high-risk use cases. Developers face strict validation protocols that lengthen development timelines. It increases pressure on small firms with limited resources. Toxicology studies require extensive modeling to prove effectiveness. Manufacturing standards demand precise control that raises production cost. Clinical testing remains difficult due to unpredictable poisoning events. Firms struggle to secure large sample groups for outcome measurement. The Alexipharmic Drugs Market must address regulatory hurdles to maintain innovation flow.
Limited Accessibility in Low-Resource Regions and Uneven Emergency Preparedness Levels
Rural centers struggle to maintain antidote stockpiles due to tight budgets and supply gaps. Training gaps reduce clinician confidence in administering antidotes. It delays treatment in critical exposure cases. Distribution networks fail to supply stable inventory during emergency demand spikes. Hospitals lack cold-chain infrastructure for certain formulations. Public health teams highlight uneven readiness across regions. Awareness levels remain low in high-risk communities. The Alexipharmic Drugs Market faces persistent access barriers that slow growth.
Alexipharmic Drugs Market Opportunities
Expansion of Public-Private Partnerships to Strengthen Poison Response Infrastructure
Governments seek partnerships that improve antidote distribution and hospital readiness. Firms gain opportunities to supply national poison preparedness programs. It supports long-term contracts for stocking essential antidotes. Health agencies fund training modules to improve clinician capability. Emergency teams receive structured support to upgrade response workflows. Manufacturers engage in multi-year collaboration projects for supply resilience. Centers develop integrated treatment networks that speed up intervention. The Alexipharmic Drugs Market benefits from widening institutional partnerships.
Advancement in Novel Therapeutic Platforms and Cross-Disciplinary Toxicology Research
Biotech firms explore monoclonal antibodies, nanoparticle carriers, and enzyme therapies for targeted toxin neutralization. Research collaborations push discovery pipelines with stronger precision. It drives interest in platform-based antidotes with scalable design. Universities partner with chemical safety groups to study emerging toxins. Labs use high-throughput tools to identify new therapeutic targets. Investment groups support early-stage programs with strong clinical potential. Specialized formulations aim to support field use in disaster zones. The Alexipharmic Drugs Market gains fresh potential through scientific breakthroughs.
Alexipharmic Drugs Market Segmentation Analysis:
By Type
Chemical antidotes support strong demand across the Alexipharmic Drugs Market due to their wide use in metal toxicity and reactive chemical exposures. Hospitals rely on chelating and reactive agents that provide targeted detoxification. It helps clinicians manage high-risk emergencies with structured precision. Physical antidotes gain traction where dilution or absorption offers fast symptomatic relief. Pharmacological antidotes expand with enzyme-based and receptor-based therapies designed for complex poison interactions. Neutralizing agents support broader coverage against corrosive and irritant substances. Decontaminants remain essential for rapid external exposure control. Preventive medications gain interest from high-risk occupational groups.
By Route of Administration
Oral formulations hold wide adoption due to convenience and suitability for mild to moderate poison cases across the Alexipharmic Drugs Market. Topical products serve targeted exposure scenarios where skin contact drives symptom severity. Injectable formulations retain dominance in emergency settings because of rapid onset. It supports critical care teams that require reliable dosing during urgent detoxification. Other administration routes remain relevant for niche clinical needs. Hospitals choose routes based on toxin type and patient stability. Diverse formats improve treatment flexibility across care levels. Broad availability strengthens readiness for varied poisoning events.
- For instance, Chemet® (succimer), produced by Arbor Pharmaceuticals, is an FDA-approved oral heavy-metal chelator used nationwide for pediatric lead poisoning and remains the only U.S.-approved oral chelation modality in routine clinical use.
By Application
Opioid overdose treatments remain central to demand due to rising misuse trends across several regions within the Alexipharmic Drugs Market. Alcohol overdose treatment maintains steady use in emergency departments worldwide. Cyanide poisoning solutions serve industrial zones and high-risk work environments. It requires fast-acting agents that stabilize patients within short therapeutic windows. Lead poisoning treatment depends on structured chelation therapies. Benzodiazepine overdose intervention supports psychiatric and emergency centers with focused reversal agents. Other applications cover a wide mix of accidental and intentional exposures. Broad clinical diversity sustains long-term adoption.
By End-Use
Hospitals dominate end-use due to their role in acute poisoning management within the Alexipharmic Drugs Market. Emergency units require well-stocked antidote inventories for high-severity cases. Homecare settings grow with increased use of mild-case detoxification therapies. It supports patients who need monitored recovery outside hospitals. Specialty clinics handle occupational and chronic exposure cases that require structured diagnostics. Other settings cover community care centers that manage moderate-risk scenarios. Strong clinical dependency across all categories sustains steady market penetration. Each end-use group contributes distinct demand patterns.
- For instance, many U.S. hospitals maintain FDA-approved Cyanokit® (hydroxocobalamin) for cyanide poisoning, following national emergency guidelines that require 24/7 antidote availability in facilities handling industrial or fire-related exposure risks.
By Distribution Channel
Hospital pharmacies lead distribution due to their responsibility for stocking critical antidotes across the Alexipharmic Drugs Market. Retail pharmacies support non-emergency needs and mild case management. It expands patient access for preventive and low-risk antidote use. Online pharmacies gain traction with improved reach and rising interest in convenient access channels. Digital platforms support better access for remote regions. Structured distribution improves supply stability for all healthcare facilities. Broad channel availability strengthens resilience during emergency surges.
Segmentation:
By Type
- Chemical Antidotes
- Physical Antidotes
- Pharmacological Antidotes
- Neutralizing Agents
- Decontaminants
- Preventive Medications
By Route of Administration
- Oral
- Topical
- Injectable
- Others
By Application
- Opioid Overdose
- Alcohol Overdose
- Cyanide Poisoning
- Lead Poisoning
- Benzodiazepine Overdose
- Others
By End-Use
- Hospital
- Homecare
- Specialty Clinics
- Others
By Distribution Channel
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
By Region
- North America
- Europe
- Germany
- France
- U.K.
- Italy
- Spain
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- South-east Asia
- Rest of Asia Pacific
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Middle East & Africa
- GCC Countries
- South Africa
- Rest of the Middle East and Africa
Regional Analysis:
North America holds the largest share of the Alexipharmic Drugs Market at around 38% due to strong emergency care systems and high readiness levels for poisoning response. Hospitals maintain structured antidote inventories that support rapid intervention. It benefits from advanced toxicology research that drives new product development. Regulatory bodies promote strict preparedness guidelines that reinforce market growth. Rising opioid overdose cases strengthen demand for fast-acting reversal agents. Growing awareness campaigns expand the use of preventive solutions across communities.
Europe accounts for nearly 27% of the market due to well-established poison control networks and strong pharmaceutical manufacturing capabilities. Countries invest in standardized treatment protocols that support consistent antidote use across hospitals. It benefits from regulated chemical safety programs that drive preventive medication adoption. Industrial regions report structured preparedness plans for chemical and biological risks. Public health agencies support coordinated emergency response training. Increasing chronic exposure cases sustain demand for chelation and neutralizing agents.
Asia Pacific holds close to 24% share and remains the fastest-growing region due to rising industrial activities and expanding healthcare access across emerging economies. Hospitals upgrade emergency departments to manage higher toxin exposure cases. It gains momentum from large population density, which elevates accidental poisoning risks. Governments strengthen poison information centers to improve reporting and treatment accuracy. Domestic pharmaceutical firms increase production capacity for essential antidotes. Improved distribution networks support wider rural access. Latin America, Middle East, and Africa collectively account for the remaining 11% share, driven by improving emergency care infrastructure.
Shape Your Report to Specific Countries or Regions & Enjoy 30% Off!
Key Player Analysis:
- Pfizer Inc. (including Alpharma Pharmaceuticals)
- Fresenius Kabi
- Teva Pharmaceutical Industries Ltd.
- Ethypharm S.A.
- Emergent BioSolutions
- Bausch Health Companies Inc. (formerly Valeant)
- Hikma Pharmaceuticals
- Amneal Pharmaceuticals
- Apotex Inc.
- Accord Healthcare (Intas Pharmaceuticals)
- Mylan N.V. (Viatris)
- Bristol-Myers Squibb Company
- Johnson & Johnson
- Merck & Co., Inc.
- Sanofi S.A.
Competitive Analysis:
The Alexipharmic Drugs Market features a mix of global pharmaceutical leaders and specialized toxicology firms that compete through innovation, rapid-response formulations, and broad antidote portfolios. Companies invest in enzyme-based and receptor-targeted antidotes to strengthen therapeutic precision. It drives strong differentiation in high-severity treatment categories. Firms pursue regulatory clearances for expanded indications that support market penetration. Strategic partnerships with hospitals and poison centers enhance brand visibility and improve supply reliability. Manufacturers focus on stable formulations suitable for field use to serve emergency responders. Competition intensifies in segments such as opioid reversal agents and chelating therapies where multiple players offer similar solutions. Strong R&D pipelines and national stockpile contracts influence long-term positioning across key regions.
Recent Developments:
- In February 2026, Pfizer joined BaseLaunch as the newest partner among six leading pharma companies, gaining access to high-quality life sciences opportunities in central Europe and contributing expertise in biotech innovation and drug development startups.
- In November 2025, Pfizer Inc. acquired Metsera adding four clinical-stage incretin and amylin programs to bolster its obesity portfolio, alongside a $2 billion licensing deal for YP05002, an oral GLP-1 receptor agonist from YaoPharma, positioning it against leaders like Eli Lilly and Novo Nordisk in the $100B market.
Report Coverage:
The research report offers an in-depth analysis based on Type, Route of Administration, Application, End-Use, and Distribution Channel. It details leading market players, providing an overview of their business, product offerings, investments, revenue streams, and key applications. Additionally, the report includes insights into the competitive environment, SWOT analysis, current market trends, as well as the primary drivers and constraints. Furthermore, it discusses various factors that have driven market expansion in recent years. The report also explores market dynamics, regulatory scenarios, and technological advancements that are shaping the industry. It assesses the impact of external factors and global economic changes on market growth. Lastly, it provides strategic recommendations for new entrants and established companies to navigate the complexities of the market.
Future Outlook:
- Expanding emergency care capacity strengthens demand for advanced detoxification therapies across diverse clinical settings.
- Growing adoption of rapid-acting antidotes supports faster intervention during high-risk poisoning cases.
- Rising industrial exposure rates drive sustained need for toxin-neutralizing and preventive formulations.
- Increased awareness of opioid and alcohol overdoses boosts therapeutic uptake in acute care units.
- Evolving toxicology research encourages development of enzyme-based and receptor-targeted antidotes.
- Broader government preparedness programs improve stockpiling and distribution of essential antidotes.
- Expanding digital support tools improve poison management accuracy for frontline medical teams.
- Stronger pharmaceutical investment accelerates pipeline growth for niche and rare toxin treatments.
- Wider adoption of stable formulations strengthens field readiness for emergency responders.
- Growing healthcare access in emerging economies supports long-term expansion of the Alexipharmic Drugs Market.




