Angioplasty Balloons Market Overview:
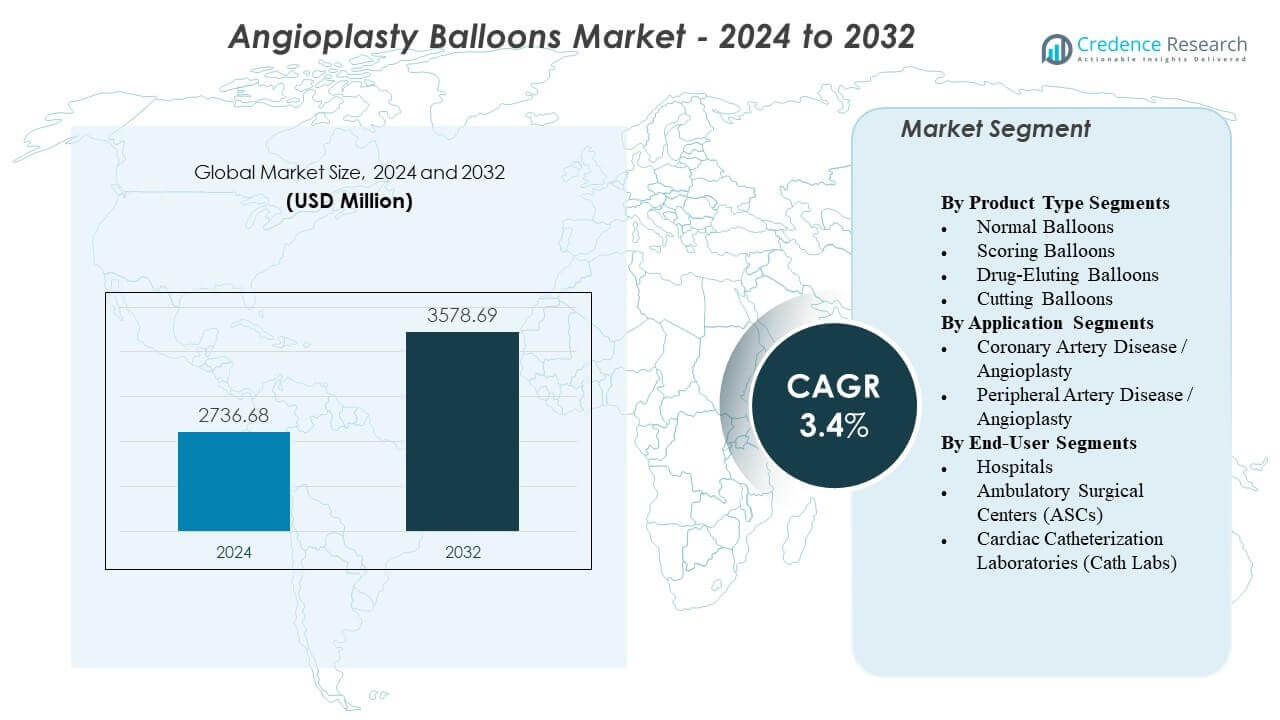
The Angioplasty Balloons Market is projected to grow from USD 2,736.68 million in 2024 to an estimated USD 3,578.69 million by 2032, with a compound annual growth rate (CAGR) of 3.4% from 2024 to 2032.
| REPORT ATTRIBUTE |
DETAILS |
| Historical Period |
2020-2023 |
| Base Year |
2024 |
| Forecast Period |
2025-2032 |
| Angioplasty Balloons Market Size 2024 |
USD 2,736.68 million |
| Angioplasty Balloons Market, CAGR |
3.4% |
| Angioplasty Balloons Market Size 2032 |
USD 3,578.69 million |
Market growth is driven by the rising burden of coronary and peripheral artery disease, prompting hospitals to adopt balloons that offer improved precision, deliverability, and vessel safety. Advancements in drug-coated, scoring, and high-pressure non-compliant balloons support better lesion preparation and reduce repeat interventions. Clinicians favor newer designs with refined profiles and controlled inflation systems that enhance predictability during complex procedures. Manufacturers focus on developing platforms tailored for calcified lesions, small vessels, and restenosis cases, supporting stronger adoption across interventional cardiology.
Regionally, North America leads due to advanced cardiovascular infrastructure, strong clinical adoption, and extensive use of high-performance balloon technologies. Europe follows with established interventional practices and steady integration of specialty balloons engineered for complex anatomy. Asia Pacific is emerging rapidly as rising healthcare investment, expanding cath lab networks, and growing awareness of early cardiovascular treatment fuel broader adoption. Latin America and the Middle East show gradual growth as cardiac programs strengthen and access to modern angioplasty systems improves across major medical centers.
Angioplasty Balloons Market Insights:
- The Angioplasty Balloons Market is projected to rise from USD 2,736.68 million in 2024 to USD 3,578.69 million by 2032, advancing at a 3.4% CAGR.
- Growing demand for minimally invasive cardiovascular procedures and improved balloon technologies supports strong market momentum.
- Limited access to advanced cath lab infrastructure and challenges in treating heavily calcified lesions restrain wider adoption in several regions.
- North America leads due to advanced interventional systems and high procedural volumes, while Europe follows with established clinical practice patterns.
- Asia Pacific emerges as the fastest-expanding region driven by rising healthcare investment and increasing availability of modern angioplasty solutions.
Access crucial information at unmatched prices!
Request your sample report today & start making informed decisions powered by Credence Research Inc.!
Download Sample
Angioplasty Balloons Market Drivers
Growing Burden of Cardiovascular Disorders and Rising Preference for Minimally Invasive Interventions
The rising global burden of coronary artery blockages strengthens demand for angioplasty procedures that support quick recovery. The Angioplasty Balloons Market gains strong traction due to the need for precision-driven tools that reduce trauma during treatment. Healthcare systems prefer minimally invasive solutions that shorten hospital stay and improve procedural safety. Physicians support these balloons due to consistent performance during lesion dilation. It encourages adoption in high-volume cardiac centers. Hospitals continue to expand cath lab capabilities to meet rising procedure counts. Manufacturers respond with designs tailored for complex anatomy. Patients benefit from faster outcomes and reduced complications.
Rapid Advancements in Balloon Technologies Improving Clinical Success and Workflow Efficiency
Manufacturers introduce newer balloon platforms with stronger pushability and reliable inflation control that support efficient navigation. It helps specialists handle calcified and tortuous vessels with improved confidence. The Angioplasty Balloons Market expands due to improved coating technologies that support controlled drug delivery. Hospitals value designs that reduce restenosis cases and increase procedural durability. Clinical teams adopt balloons with refined compliance levels to match lesion requirements. Companies focus on product safety to support demanding interventional settings. Regulatory approvals encourage innovation activity within major markets. Newer systems gain preference in centers treating complex cardiovascular profiles.
- For instance, Medtronic launched the Prevail Drug-Coated Balloon (DCB), which utilizes a PowerTrac technology that enhances pushability by 2 times compared to previous generations, allowing for better deliverability in highly tortuous 0.014-inch wire systems.
Growing Adoption of Image-Guided Procedures and Expanding Interventional Cardiology Capabilities Worldwide
Cath labs integrate advanced imaging systems that increase accuracy during angioplasty. These tools enable cardiologists to position balloons with enhanced clarity across multiple vessel structures. The Angioplasty Balloons Market benefits from imaging upgrades that improve treatment planning. Hospitals invest in digital platforms to support better patient outcomes. It strengthens procedural success across both primary and advanced treatments. Healthcare providers rely on updated guidance tools to reduce complications. Training programs expand to support rising device usage. Developing nations increase investment in cardiovascular units to meet growing clinical demand.
- For instance, Philips Healthcare integrated its Azurion image-guided therapy platform with specialized balloon titration tools, which has been shown to reduce procedural time by 17% and decrease radiation exposure by up to 50% during complex balloon angioplasty cases.
Increasing Use of Drug-Coated and Specialty Balloons Targeting Complex Vascular Lesions
Drug-coated balloons gain wider use due to their ability to reduce recurrence of vessel narrowing. Hospitals adopt these devices in cases where stents show limited suitability. The Angioplasty Balloons Market sees rising penetration of balloons designed for bifurcations, small vessels and restenosis cases. Clinicians rely on targeted formulations that support better healing at the treatment site. It supports strong usage trends within advanced care centers. Manufacturers build products with uniform drug transfer for reliable performance. Healthcare providers choose platforms suited for high-risk lesion types. Research supports expansion of specialized balloons that improve long-term results.
Angioplasty Balloons Market Trends
Shift Toward High-Pressure and Non-Compliant Balloons for Complex Lesion Preparation
Interventional teams now prefer high-pressure balloons for calcified and resistant plaques where standard tools struggle. The Angioplasty Balloons Market reflects this shift due to growing clinical need for strong lesion modification. Hospitals see higher success rates with non-compliant designs that maintain diameter stability. It improves precision during pre-stent preparation. Cardiologists adopt these devices to manage challenging anatomy. Demand rises for balloons that limit slippage during inflation. Manufacturers improve material strength to support higher inflation thresholds. Adoption remains strong in specialized cardiovascular centers.
- For instance, Boston Scientific’s NC Emerge™ non-compliant balloon delivers up to 20 atm rated burst pressure, providing controlled expansion for calcified lesions that resist standard balloons. Its engineered polymer layers maintain diameter stability during aggressive lesion preparation.
Strong Uptake of Ultra-Low-Profile Balloons Supporting Small Vessel and Peripheral Procedures
Device makers develop balloons with reduced profiles to reach tight and narrow vessels safely. The Angioplasty Balloons Market sees growing usage of these balloons in small vessel interventions. Physicians value improved crossability when dealing with delicate anatomy. It reduces vessel trauma and supports smoother navigation. Hospitals integrate these devices for both coronary and peripheral applications. Clinical teams prefer designs that maintain flexibility without losing structural strength. Demand extends into regions expanding cardiac programs. Product portfolios grow to match diverse lesion types.
Expansion of Digital Integration, Including Smart Inflation Systems and Workflow-Supporting Platforms
Smart inflation tools help clinicians control pressure levels with improved accuracy. The Angioplasty Balloons Market benefits from digital systems that support consistent performance and reduce operator error. Hospitals integrate displays that track inflation parameters in real time. It strengthens procedural reliability for complex interventions. Teams adopt platforms that record data for quality improvement. Device makers invest in systems that link balloons with predictive tools. Clinical outcomes improve due to enhanced standardization. Adoption rises in advanced cardiac networks.
- For instance, the NuMED Pre-SION™ digital inflation device delivers high-accuracy pressure readings typically within ±1–2% of full-scale, allowing clinicians to manage inflation cycles with tighter control during high-pressure NC balloon procedures.
Rising Preference for Personalized Balloon Selection Driven by Data-Supported Treatment Planning
Providers adopt platforms that analyze vessel dimensions and lesion characteristics to guide balloon choice. The Angioplasty Balloons Market aligns with this trend through wider product variety. Physicians rely on accurate sizing to reduce complications. It supports stronger postoperative outcomes. Hospitals value tools that match balloon compliance with patient-specific needs. Companies develop tailored product families to meet varied anatomical demands. Clinical studies encourage structured balloon selection pathways. The trend grows with improved diagnostic imaging integration.
Angioplasty Balloons Market Challenges Analysis
Regulatory Complexity, Cost Pressures and Limited Access in Underdeveloped Cardiac Care Systems
High regulatory requirements increase development timelines for balloon platforms that target advanced cardiovascular conditions. The Angioplasty Balloons Market faces burden from strict testing standards that extend approval phases. Hospitals in developing nations struggle to maintain purchasing budgets for premium devices. It increases disparity between advanced and emerging care systems. Supply chain delays disrupt product availability in remote regions. Training shortages reduce procedural success in newer cardiac units. Clinical teams need consistent device quality, yet variations remain across manufacturers. Cost sensitivity continues to restrict adoption in low-resource settings.
Rising Clinical Preference for Alternative Technologies and Increased Focus on Long-Term Outcomes
Alternative vascular treatment methods reduce dependence on balloons for some lesion types. Hospitals evaluate long-term success rates before expanding device procurement. The Angioplasty Balloons Market experiences pressure from providers seeking tools with extended durability. It creates demand for systems that outperform conventional balloons in recurrence reduction. Complexity increases when treating heavily calcified lesions with limited balloon response. Clinicians require improved safety profiles for high-risk patients. Product differentiation becomes difficult in a market with many similar offerings. Regulatory bodies emphasize outcome-based assessments that challenge slower-moving manufacturers.
Angioplasty Balloons Market Opportunities
Technical Innovations, Expanding Cath Lab Networks and Rising Demand for Value-Driven Balloon Platforms
New designs featuring improved inflation control, enhanced coatings and specialized structures create strong growth potential worldwide. The Angioplasty Balloons Market benefits from hospitals expanding cath lab capacity in developing regions. It increases access to interventional cardiology services. Manufacturers introduce cost-efficient models to support broader adoption within budget-constrained systems. Digital tools elevate product placement accuracy and drive stronger clinical confidence. Partnerships with healthcare providers strengthen training and validation processes. Demand grows for balloons optimized for recurrent lesions. Companies gain advantage through material advances that support better durability.
Growing Opportunity in Drug-Coated Balloons, Niche Lesion Categories and Personalized Therapy Planning
Drug-coated balloons create new value streams for treating restenosis without stent placement. The Angioplasty Balloons Market gains opportunity in segments focusing on bifurcation lesions and challenging vessel types. It accelerates adoption in centers handling complex procedural loads. Providers seek specialized balloons that match unique anatomical and diagnostic requirements. Product designers invest in controlled-release coatings that support improved healing. Clinical networks expand research to validate next-generation formulations. Global focus on reducing post-procedure complications drives interest in innovative balloon therapy pathways.
Angioplasty Balloons Market Segmentation Analysis:
By Product Type
Normal balloons hold strong use in routine angioplasty and support consistent vessel dilation. Scoring balloons provide higher precision for resistant plaque and help clinicians treat fibrotic lesions with control. Drug-eluting balloons gain traction for restenosis cases where stent placement offers limited advantage. Cutting balloons support controlled lesion modification in tight or calcified vessels. The Angioplasty Balloons Market grows with wider adoption of specialized designs that meet complex cardiovascular needs. It benefits from strong clinical demand for tools that fit varied anatomical and procedural conditions. Hospitals and cardiac centers prefer product portfolios that ensure flexibility. Manufacturers respond with expanded technology options that support better outcomes.
- For instance, BIOTRONIK’s Passeo-18 Lux drug-eluting balloon demonstrated a >60% reduction in restenosis at 12 months, supported by paclitaxel-coated matrix technology that ensures consistent drug transfer.
By Application
Coronary artery angioplasty remains the dominant segment due to high procedure volumes for treating narrowed coronary vessels. Clinicians rely on balloons that deliver predictable expansion during pre-stent or stand-alone interventions. Peripheral artery angioplasty expands quickly with increasing detection of limb ischemia and demand for minimally invasive treatments. The Angioplasty Balloons Market aligns with both clinical settings through device designs suited for distinct vessel diameters. It supports treatment planning where durability and safety guide product choice. Hospitals value balloons that manage restenosis and complex lesions in peripheral pathways. Adoption grows with rising awareness across vascular care programs. Manufacturers continue refining products that elevate procedural success.
By End User
Hospitals lead due to advanced cath labs, high cardiac patient flow and broader access to trained specialists. These facilities handle complex angioplasty cases that require varied balloon types. Ambulatory surgical centers expand usage with shorter-stay treatments and streamlined workflows. It supports care models focused on efficiency and rapid recovery. Cardiac catheterization laboratories play a central role because they manage diagnostic and interventional workloads. The Angioplasty Balloons Market benefits from increased lab capacity and investment in imaging and inflation technologies. Specialized labs prefer balloons that maintain reliability during demanding procedures. Growth spreads across regions upgrading cardiovascular treatment infrastructure.
- For instance, Philips’ Azurion cath lab platform improved workflow efficiency by up to 17%, allowing hospitals to manage higher angioplasty volumes with optimized imaging and process automation.
Segmentation:
By Product Type Segments
- Normal Balloons
- Scoring Balloons
- Drug-Eluting Balloons
- Cutting Balloons
By Application Segments
- Coronary Artery Disease / Angioplasty
- Peripheral Artery Disease / Angioplasty
By End-User Segments
- Hospitals
- Ambulatory Surgical Centers (ASCs)
- Cardiac Catheterization Laboratories (Cath Labs)
By Region
- North America
- Europe
- Germany
- France
- U.K.
- Italy
- Spain
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- South-east Asia
- Rest of Asia Pacific
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Middle East & Africa
- GCC Countries
- South Africa
- Rest of the Middle East and Africa
Regional Analysis:
North America holds the largest share of the Angioplasty Balloons Market due to high cardiovascular procedure volumes and strong adoption of advanced interventional tools. The region benefits from established cath lab networks that support consistent demand across hospitals and specialty centers. It maintains a leading position due to strong reimbursement systems and continuous product upgrades. The market gains strength from widespread use of drug-eluting and specialty balloons. North America continues to set performance benchmarks that influence global device strategies. The Angioplasty Balloons Market grows steadily here, and it supports innovation cycles shaped by clinical evidence.
Europe secures the second-largest market share supported by mature healthcare systems and structured cardiovascular care pathways. Hospitals adopt a wide mix of compliant, non-compliant and scoring balloons tailored to complex lesions. It benefits from strong clinical preference for evidence-validated devices and rising usage of drug-coated balloons. Peripheral artery disease interventions expand steadily in this region, driving higher procedure counts. European countries maintain strong quality standards that shape procurement trends. The region strengthens global competitiveness through active research networks and technology adoption.
Asia Pacific records the fastest growth and expands its market share with rising procedure volumes and improving cardiac care infrastructure. The Angioplasty Balloons Market gains momentum in countries investing in new cath labs and cardiovascular treatment programs. It benefits from rising awareness of coronary disease and wider access to minimally invasive procedures. Hospitals adopt newer balloon platforms to meet high patient demand. Manufacturers target this region with cost-efficient and high-performance designs. Growth accelerates as training programs expand and private hospitals upgrade interventional capabilities.
Shape Your Report to Specific Countries or Regions & Enjoy 30% Off!
Key Player Analysis:
- Medtronic
- Boston Scientific Corporation
- Terumo Corporation
- BIOTRONIK
- Abbott Laboratories
- Braun Melsungen AG
- BD (Becton, Dickinson and Company)
- Cook Medical
- Johnson & Johnson
- Teleflex Incorporated
- AngioDynamics
- Cardinal Health
- Koninklijke Philips N.V.
Competitive Analysis:
The Angioplasty Balloons Market reflects strong competition driven by product innovation, clinical performance and geographic expansion. Leading companies such as Medtronic, Boston Scientific Corporation, Terumo Corporation, BIOTRONIK, and Abbott Laboratories focus on delivering advanced balloon platforms that support complex cardiovascular interventions. It drives companies to refine material strength, coating technologies and inflation control systems to strengthen clinical outcomes. Firms such as B. Braun Melsungen AG, BD, Cook Medical, and Johnson & Johnson compete by expanding product portfolios that address both coronary and peripheral applications. Companies also enhance training programs to support physicians handling advanced cases. Strong distribution networks help firms such as Teleflex Incorporated, AngioDynamics, Cardinal Health, and Koninklijke Philips N.V. reach diverse healthcare settings. The competitive landscape continues to evolve as firms integrate digital tools and imaging compatibility into their platforms. Innovation strength, regulatory success and clinical validation shape long-term positioning across global markets.
Recent Developments:
- In February 2025, Teleflex announced the acquisition of BIOTRONIK’s vascular intervention business for approximately EUR 760 million (USD 820 million). This deal enhances Teleflex’s interventional cardiology portfolio by adding drug-coated balloons (DCBs), drug-eluting stents, and other angioplasty-related devices from BIOTRONIK.
- In January 2025, Boston Scientific acquired Bolt Medical for USD 664 million, entering the intravascular lithotripsy segment with acoustic-pressure balloon technology designed for treating calcified arterial disease, relevant to angioplasty procedures.
Report Coverage:
The research report offers an in-depth analysis based on Product Type Segments, Application Segments, End-User Segments and Region Segments. It details leading market players, providing an overview of their business, product offerings, investments, revenue streams, and key applications. Additionally, the report includes insights into the competitive environment, SWOT analysis, current market trends, as well as the primary drivers and constraints. Furthermore, it discusses various factors that have driven market expansion in recent years. The report also explores market dynamics, regulatory scenarios, and technological advancements that are shaping the industry. It assesses the impact of external factors and global economic changes on market growth. Lastly, it provides strategic recommendations for new entrants and established companies to navigate the complexities of the market.
Future Outlook:
- The market will see strong adoption of high-pressure and non-compliant balloons that support precise lesion preparation in complex coronary and peripheral cases.
- Drug-eluting balloons will gain wider use due to their ability to manage restenosis without permanent implants, strengthening their role in repeat interventions.
- Ultra-low-profile balloons will expand in clinical use as providers seek devices that navigate tight and tortuous vessels with minimal trauma.
- Hospitals will increase investments in digital inflation control systems that improve consistency and help streamline interventional workflows.
- Product innovation will focus on advanced materials that deliver controlled expansion and higher durability during demanding procedures.
- Personalized therapy planning will influence balloon selection, supported by imaging tools that guide sizing and compliance choice.
- Emerging regions will accelerate adoption due to expanding cath lab networks and rising awareness of early cardiovascular treatment.
- Manufacturers will strengthen collaborations with cardiology centers to validate new balloon platforms and enhance training programs.
- Competition will shift toward differentiated technologies that address niche lesion types and reduce complications after intervention.
- Regulatory pathways will support safer and more effective devices, pushing companies to refine product design and clinical performance.




