Biopharmaceuticals Contract Manufacturing Market Overview:
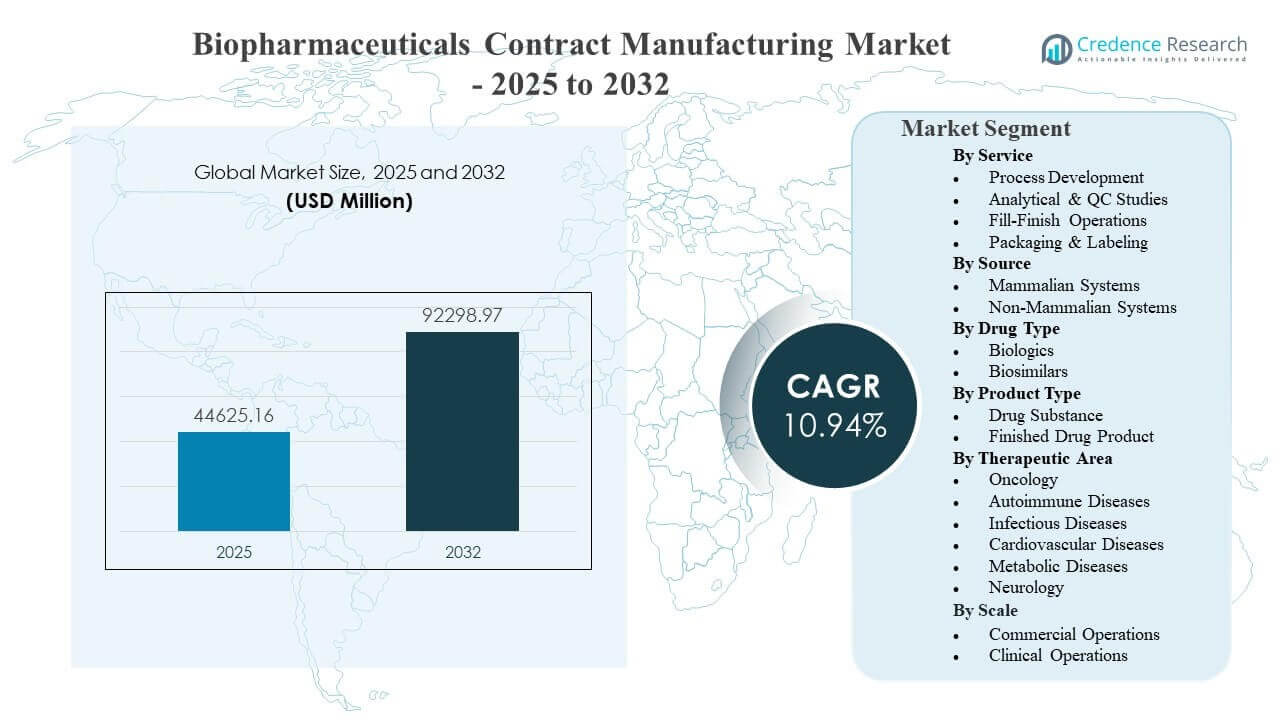
The global Biopharmaceuticals Contract Manufacturing Market size was estimated at USD 44,625.16 million in 2025 and is expected to reach USD 92,298.97 million by 2032, growing at a CAGR of 10.94% from 2025 to 2032. Demand is primarily driven by biopharma sponsors increasing outsourcing to accelerate development and scale-up while managing capital intensity, compliance burdens, and time-to-market risks across complex biologic modalities. Capacity expansion in large-scale biologics manufacturing and the growing preference for end-to-end CDMO partnerships further reinforce market growth, with outsourcing decisions increasingly tied to speed of tech transfer, analytics depth, and reliable commercial supply execution.
| REPORT ATTRIBUTE |
DETAILS |
| Historical Period |
2020-2024 |
| Base Year |
2025 |
| Forecast Period |
2026-2032 |
| Biopharmaceuticals Contract Manufacturing Market Size 2025 |
USD 44,625.16 million |
| Biopharmaceuticals Contract Manufacturing Market, CAGR |
10.94% |
| Biopharmaceuticals Contract Manufacturing Market Size 2032 |
USD 92,298.97 million |
Key Market Trends & Insights
- Process Development accounted for the largest share of 37.8% in 2025, reflecting strong demand for tech transfer, scale-up, and process robustness services.
- Mammalian Systems held 58.3% share in 2025, supported by broad use in complex biologics requiring human-like post-translational modifications.
- Oncology represented 32.4% share in 2025, driven by sustained biologics activity and high manufacturing intensity across development and supply.
- North America contributed 35.80% share in 2025, supported by a deep innovator base and strong commercial biologics production requirements.
- Biosimilars are projected to expand at 12.25% CAGR over the forecast period, reflecting cost-driven adoption and expanding pipeline execution needs.
Segment Analysis
The market continues to shift toward outsourcing models that reduce fixed manufacturing burdens while improving speed and flexibility across clinical and commercial programs. Sponsors increasingly prefer providers that can deliver integrated workflows spanning development, analytics, and manufacturing handoffs with minimal friction, particularly where process performance and regulatory consistency are closely scrutinized. This strengthens demand for service packages that combine process design, analytical control strategies, and execution readiness for scale-up.
Operational decision-making is also shaped by capacity access, quality track record, and the ability to manage variability across complex biologic products. Buyers place high emphasis on robust analytical and QC frameworks that support release confidence and comparability, especially when transitioning from clinical to commercial operations. As pipelines widen and molecule diversity grows, CDMOs benefit from demand for modular capacity, standardized platforms, and streamlined technology transfer pathways.
Access crucial information at unmatched prices!
Request your sample report today & start making informed decisions powered by Credence Research Inc.!
Download Sample
By Service Insights
Process Development accounted for the largest share of 37.8% in 2025. This leadership is supported by the need to define scalable, reproducible processes early to reduce downstream risk and prevent costly failures at later stages. Sponsors increasingly rely on CDMOs for platform process expertise, scale-up troubleshooting, and optimization that improves yields and impurity control. Integrated development-to-manufacturing pathways also reduce delays by shortening the handoff between lab-scale work and GMP production execution.
By Source Insights
Mammalian Systems accounted for the largest share of 58.3% in 2025. Mammalian platforms remain preferred for many biologics where complex protein expression and human-like glycosylation profiles are critical for efficacy and safety. High monoclonal antibody and recombinant protein volumes keep mammalian capacity utilization elevated across both clinical and commercial operations. CDMOs with proven mammalian platforms and strong analytical depth are better positioned to win long-duration supply agreements and multi-product portfolios.
By Drug Type Insights
Biologics continue to represent the foundational volume base for contract manufacturing because large portions of clinical pipelines and commercial portfolios depend on robust biologic production capabilities. Sponsors increasingly seek partners that can support multiple molecule types with consistent quality systems and repeatable manufacturing performance. Biosimilars add momentum through cost-focused programs that require efficient manufacturing execution, strong comparability planning, and disciplined analytical control strategies. Competitive positioning in this dimension often depends on the ability to deliver both cost efficiency and consistent compliance across scale transitions.
By Product Type Insights
Drug Substance outsourcing remains closely tied to demand for bioreactor capacity, upstream and downstream expertise, and process control disciplines that can be difficult to replicate in-house at comparable speed. Finished Drug Product requirements reinforce demand for sterile operations, container-closure integrity, and high-consequence quality assurance processes that support commercial distribution readiness. Many sponsors prioritize partners that can coordinate drug substance to fill-finish workflows to reduce transfer risk, improve schedule reliability, and strengthen release confidence. The push toward integrated offerings also supports simpler governance models and fewer operational handoffs across the supply chain.
By Therapeutic Area Insights
Oncology accounted for the largest share of 32.4% in 2025. A high level of biologics development activity in oncology sustains demand for complex manufacturing programs with stringent analytical and quality requirements. Programs often require flexible capacity planning due to multiple indications, evolving dose requirements, and changing timelines across trial phases. CDMOs that can scale rapidly and maintain stable quality performance are favored, particularly where sponsors need consistent supply across multi-region development and early commercial launch windows.
By Scale Insights
Commercial Operations are typically supported by longer-duration outsourcing contracts where supply assurance, validated processes, and regulatory readiness are decisive purchasing criteria. Clinical Operations growth is reinforced by the breadth of pipeline assets that require flexible batch sizing, rapid turnaround, and adaptable scheduling. Sponsors increasingly structure outsourcing strategies to reserve capacity for expected program advancement while avoiding overcommitment during early clinical risk periods. Scale decisions are also influenced by modality mix, with specialized biologics often requiring tailored execution and higher service intensity.
Biopharmaceuticals Contract Manufacturing Market Drivers
Expanding biologics pipelines and complexity of manufacturing
Biopharmaceutical pipelines continue to broaden across monoclonal antibodies and other complex biologic formats that require sophisticated manufacturing controls and compliance discipline. The operational burden of process development, analytical strategy design, and scale-up execution drives outsourcing to specialized partners. CDMOs benefit when they can standardize execution through platform processes while still supporting molecule-specific requirements. This combination improves speed, reduces failure risk, and supports repeatable quality across multiple programs.
- For instance, WuXi Biologics stated that its 4th-generation WuXia TrueSite TI cell line platform achieves average monoclonal antibody titers above 8.0 g/L, maintains stable expression in over 99% of clonal cell lines after 60 passages, and supports scale-up beyond 20,000 L, demonstrating how platformized systems can still handle complex molecule demands at industrial scale.
Acceleration needs across development and tech transfer timelines
Sponsors increasingly prioritize speed-to-clinic and speed-to-market, which elevates the value of experienced partners that can execute rapid tech transfer and scale-up. CDMOs that offer integrated development, analytics, and GMP manufacturing reduce handoff delays and governance complexity. Faster transitions between stages also improve portfolio throughput and allow sponsors to allocate internal resources to R&D priorities. This driver is amplified where clinical timelines are competitive and product differentiation depends on execution speed.
Quality, compliance, and analytical control requirements
Manufacturing of biologics requires stringent control of impurities, process variability, and product comparability, reinforcing demand for strong analytical and QC capabilities. Outsourcing supports access to established quality systems, validated methods, and experienced regulatory readiness practices. Sponsors also prefer partners with proven performance in deviation management, documentation rigor, and audit readiness. The emphasis on consistent quality performance supports repeat business and longer-term commercial supply relationships.
Capacity access and scalable commercial supply assurance
Commercial biologics require reliable access to large-scale capacity and stable execution performance. Building or expanding internal capacity is capital-intensive and time-consuming, encouraging sponsors to outsource to established providers with flexible manufacturing footprints. CDMOs that expand capacity and modernize facilities strengthen their ability to support long-duration contracts. This driver is reinforced by the need to manage supply continuity and reduce the risk of launch delays or stock-outs.
- For instance, Samsung Biologics reported that Plant 4 adds 240,000 liters of capacity and increases total capacity at Bio Campus I to 604,000 liters, illustrating how large-scale footprint expansion strengthens long-duration commercial supply support. CDMOs that expand capacity and modernize facilities strengthen their ability to support long-duration contracts.
Biopharmaceuticals Contract Manufacturing Market Challenges
Capacity constraints and long lead times remain a challenge as sponsors compete for qualified biologics manufacturing slots and validated fill-finish capabilities. Even when capacity exists, aligning schedules to program milestones can be difficult due to variability in clinical outcomes and shifting demand profiles. These constraints can increase pricing pressure and complicate outsourcing governance, especially for mid-sized sponsors. Execution risk is elevated when multiple tech transfers occur across sites or when manufacturing networks are rebalanced.
- For example, FUJIFILM Diosynth Biotechnologies’ Hillerød expansion which doubled drug substance capacity to 12 × 20,000L bioreactors only scheduled the commencement of fill-finish production for mid-2025 as a separate phase, underscoring how validated fill-finish capability lags behind upstream capacity additions and widens the scheduling gap for sponsors requiring end-to-end slots
Regulatory and quality complexity also creates challenges, particularly when sponsors require consistent compliance performance across multi-region supply chains. Variability in documentation expectations, inspection readiness, and change control processes can slow timelines and increase operational overhead. Process deviations or analytical comparability concerns can lead to rework, delays, or additional studies. Maintaining consistent quality while scaling rapidly remains a central operational and reputational risk for both sponsors and CDMOs.
Biopharmaceuticals Contract Manufacturing Market Trends and Opportunities
The market is trending toward integrated, end-to-end CDMO partnerships where sponsors prioritize fewer vendors and more streamlined supply chain execution. Demand is rising for providers that can link process development, analytical control, GMP manufacturing, and fill-finish operations under a unified quality framework. This reduces handoff friction and supports predictable scale transitions. Providers with strong project management and standardized tech transfer models are gaining advantage.
- For instance, Mabion, a biologics-focused CDMO, demonstrated the tangible impact of this integrated model when a fully aligned, end-to-end manufacturing approach spanning upstream process development through GMP batch release under a single quality framework reduced the estimated time to release of the first GMP batch by 40%, while achieving a threefold decrease in deviations attributable to tighter control over critical quality attributes and in-process analytics across the entire development-to-release continuum.
Opportunities are also increasing in advanced analytics, digital process monitoring, and manufacturing modernization that improves batch consistency and operational visibility. Sponsors value partners that can shorten investigations, support faster release decision-making, and provide stronger lifecycle management support. Modular facilities and flexible manufacturing configurations expand addressable programs by enabling faster changeovers and better utilization. These trends support differentiation through speed, transparency, and consistent execution.
Regional Insights
North America
North America accounted for 35.80% share in 2025, supported by high biologics innovation density and strong commercial manufacturing requirements. The region benefits from established regulatory and quality ecosystems that reinforce outsourcing for high-compliance programs. Buyers often prioritize track record, scale assurance, and integrated service capabilities across development and manufacturing. Strong demand from large sponsors and a broad clinical pipeline sustain capacity utilization and long-duration contracts.
Europe
Europe held 26.70% share in 2025, supported by mature CDMO footprints, strong GMP capability, and established biologics manufacturing clusters. Regional strength is reinforced by high standards in quality systems, experienced regulatory operations, and strong technical talent pools. Outsourcing demand is sustained across both clinical and commercial programs, particularly where specialized capabilities are required. Competitive positioning often hinges on reliability, compliance performance, and multi-site manufacturing networks.
Asia Pacific
Asia Pacific represented 29.10% share in 2025, supported by expanding large-scale capacity and increasing outsourcing momentum for both development and manufacturing. The region benefits from ongoing investments in modern facilities, process standardization, and supply chain integration. Sponsors leverage Asia Pacific to improve scalability and time-to-capacity access while maintaining strong quality expectations. Growth is reinforced by capacity additions, competitive economics, and expanding biologics activity across regional hubs.
Latin America
Latin America accounted for 5.40% share in 2025, reflecting a smaller but growing role in regional manufacturing support and selected fill-finish or packaging needs. Demand growth is tied to localized supply strategies, regional access requirements, and gradual capability expansion. Sponsors may engage regional partners where proximity and regional distribution advantages are meaningful. Continued investment in quality and compliance readiness remains critical for scaling higher-value biologics work.
Middle East & Africa
Middle East & Africa captured 3.00% share in 2025, supported by selective capacity development and increasing focus on strengthening regional healthcare supply chains. Outsourcing demand is emerging in targeted areas such as packaging, labeling, and localized manufacturing support. Growth is influenced by policy-driven investments, infrastructure expansion, and improving quality frameworks. Scaling higher-complexity biologics manufacturing will depend on continued capability build-out and workforce development.
Competitive Landscape
Competition is shaped by the ability to deliver reliable capacity, strong compliance performance, and integrated service breadth across development, analytics, and manufacturing. Providers differentiate through scale, technology platforms, speed of tech transfer, and execution consistency across multi-site networks. Long-term contracts often reflect a blend of commercial supply assurance and lifecycle management capability. CDMOs that can align governance, transparency, and operational discipline with sponsor expectations are better positioned to capture repeat business.
Lonza Group remains a prominent participant with a strong focus on scalable biologics manufacturing and integrated development-to-commercial services. The company’s positioning benefits from deep process expertise, quality system maturity, and the ability to support complex biologics programs across multiple stages. Its approach is strengthened by capacity planning discipline and emphasis on operational reliability for long-duration supply relationships. This profile aligns well with sponsor demand for predictable execution and reduced supply chain risk.
The industry research and growth report includes detailed analyses of the competitive landscape of the market and information about key companies, including:
- Lonza Group
- Thermo Fisher Scientific, Inc.
- Samsung Biologics
- WuXi Biologics
- Boehringer Ingelheim GmbH
- Catalent, Inc.
- FUJIFILM Diosynth Biotechnologies
- AGC Biologics
- Rentschler Biotechnologie GmbH
- AbbVie Inc.
- Merck KGaA
- Inno Biologics Sdn Bhd
Qualitative and quantitative analysis of companies has been conducted to help clients understand the wider business environment as well as the strengths and weaknesses of key industry players. Data is qualitatively analyzed to categorize companies as pure play, category-focused, industry-focused, and diversified; it is quantitatively analyzed to categorize companies as dominant, leading, strong, tentative, and weak.
Shape Your Report to Specific Countries or Regions & Enjoy 30% Off!
Recent Developments
- In March 2026, Catalent entered a global partnership with GelMEDIX for the development and clinical manufacturing of induced pluripotent stem cell, or iPSC-derived, therapies, with Catalent also providing GMP-compliant iPSC lines and support for process development, analytical development, scale-up, and GMP manufacturing through clinical stages.
- In February 2026, Samsung Biologics partnered with CEPI to strengthen outbreak-ready vaccine production, and the agreement included an initial budget of up to US$20 million to build a scalable rapid-response manufacturing process for recombinant-protein vaccines using Samsung Biologics’ mammalian cell-based infrastructure.
- In February 2026, Catalent and S.Biomedics announced a strategic partnership to support the development and manufacturing of TED-A9, which the announcement described as S.Biomedics’ allogeneic pluripotent stem cell therapy program.
- In December 2025, AGC Biologics partnered with ATUM to integrate the Leap-In Transposase expression platform into its cell line development offerings, a move aimed at increasing yields and shortening development timelines for monoclonal antibodies, multispecifics, and other complex biologics.
Report Scope
| Report Attribute |
Details |
| Market name |
Biopharmaceuticals Contract Manufacturing Market |
| Base year and size |
2025; USD 44,625.16 million |
| Forecast year and size |
2032; USD 92,298.97 million |
| CAGR and period |
10.94% (2025–2032) |
| Units |
USD million |
| Segment coverage |
Service, Source, Drug Type, Product Type, Therapeutic Area, Scale |
| Regional coverage |
North America; Europe; Asia Pacific; Latin America; Middle East & Africa |
| Companies covered |
Lonza Group; Thermo Fisher Scientific, Inc.; Samsung Biologics; WuXi Biologics; Boehringer Ingelheim GmbH; Catalent, Inc.; FUJIFILM Diosynth Biotechnologies; AGC Biologics; Rentschler Biotechnologie GmbH; AbbVie Inc.; Merck KGaA; Inno Biologics Sdn Bhd |
| Estimated pages |
340 |
Segmentation
By Service
- Process Development
- Analytical & QC Studies
- Fill-Finish Operations
- Packaging & Labeling
By Source
- Mammalian Systems
- Non-Mammalian Systems
By Drug Type
By Product Type
- Drug Substance
- Finished Drug Product
By Therapeutic Area
- Oncology
- Autoimmune Diseases
- Infectious Diseases
- Cardiovascular Diseases
- Metabolic Diseases
- Neurology
By Scale
- Commercial Operations
- Clinical Operations
By Region
- North America
- Europe
- Germany
- France
- U.K.
- Italy
- Spain
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- South-east Asia
- Rest of Asia Pacific
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Middle East & Africa
- GCC Countries
- South Africa
- Rest of the Middle East and Africa




