Market Overview:
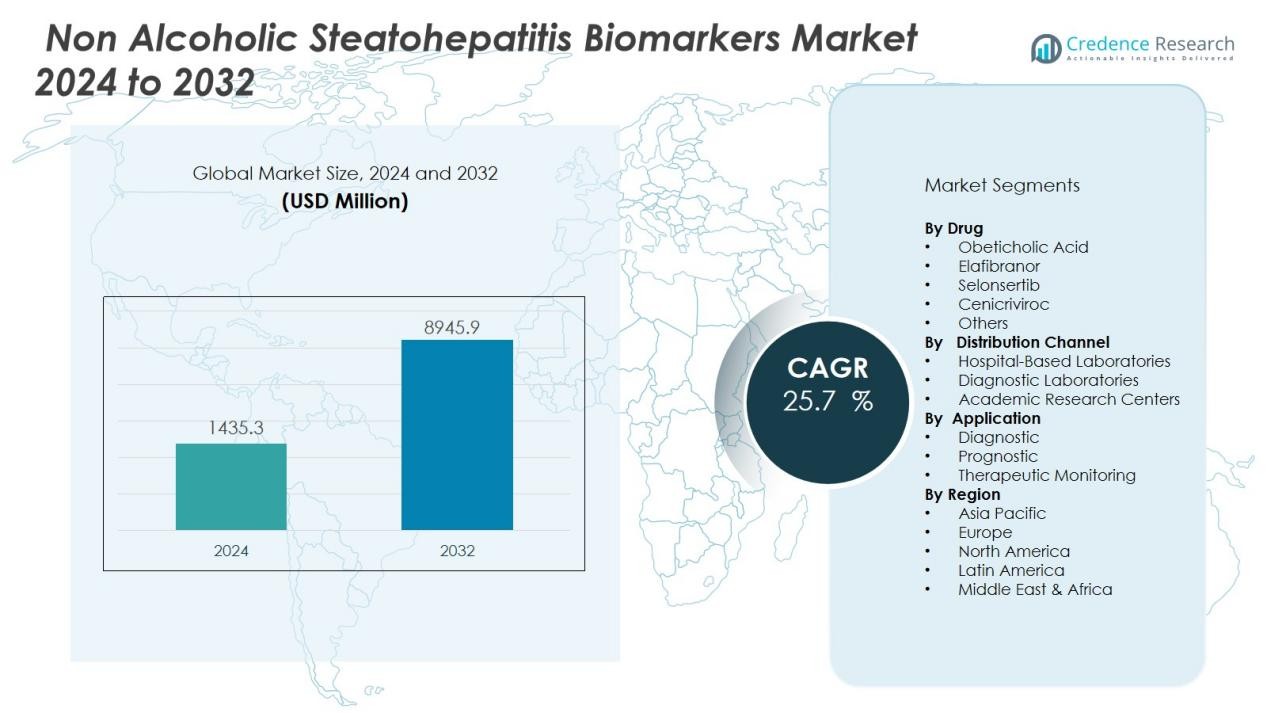
The Non Alcoholic Steatohepatitis Biomarkers Market size was valued at USD 1435.3 million in 2024 and is anticipated to reach USD 8945.9 million by 2032, at a CAGR of 25.7 % during the forecast period (2024-2032).
| REPORT ATTRIBUTE |
DETAILS |
| Historical Period |
2020-2024 |
| Base Year |
2024 |
| Forecast Period |
2025-2032 |
| Non Alcoholic Steatohepatitis Biomarkers Market Size 2024 |
USD 1435.3 Million |
| Non Alcoholic Steatohepatitis Biomarkers Market, CAGR |
25.7 % |
| Non Alcoholic Steatohepatitis Biomarkers Market Size 2032 |
USD 8945.9 Million |
Growth in this market is primarily fueled by the surge in obesity, diabetes, and metabolic syndrome cases, which are key risk factors for NASH. Traditional liver biopsy remains the gold standard but poses limitations such as invasiveness and patient discomfort. Consequently, healthcare providers and pharmaceutical companies are focusing on biomarker-based solutions to enhance accuracy, improve patient compliance, and support clinical trial processes for NASH therapeutics. Advancements in genomics, proteomics, and imaging biomarkers are further accelerating development.
Regionally, North America holds the dominant share of the NASH biomarkers market due to a high burden of lifestyle-related disorders, strong R&D infrastructure, and active pharmaceutical pipelines. Europe follows closely, supported by growing clinical research and supportive healthcare policies. Meanwhile, Asia-Pacific is emerging as the fastest-growing region, driven by increasing healthcare expenditure, rising obesity rates, and improving diagnostic adoption in China and India. Latin America and the Middle East & Africa are also gradually adopting biomarker technologies through government-led healthcare initiatives and expanding diagnostic networks.
Access crucial information at unmatched prices!
Request your sample report today & start making informed decisions powered by Credence Research Inc.!
Download Sample
Market Insights:
- The non alcoholic steatohepatitis biomarkers market was valued at USD 1435.3 million in 2024 and is projected to reach USD 8945.9 million by 2032, at a CAGR of 25.7%.
- Rising prevalence of obesity, diabetes, and metabolic syndrome is driving demand for NASH biomarkers.
- Non-invasive biomarker solutions are replacing invasive liver biopsies, improving patient comfort and compliance.
- Expanding pharmaceutical research and clinical trials are boosting biomarker adoption for drug development and monitoring.
- Technological advancements in genomics, proteomics, and imaging are enhancing biomarker accuracy and efficiency.
- Challenges include limited standardization, high development costs, regulatory hurdles, and reimbursement constraints.
- North America holds 42% market share, Europe accounts for 28%, and Asia-Pacific follows with 21% led by China, India, and Japan.
Market Drivers:
Rising Prevalence of Obesity, Diabetes, and Metabolic Disorders
The non alcoholic steatohepatitis biomarkers market is driven by the increasing burden of obesity, type 2 diabetes, and metabolic syndrome worldwide. These conditions are leading contributors to NASH progression, creating a strong demand for early detection and monitoring tools. It benefits from a global healthcare shift toward preventive strategies to reduce long-term complications. Growing awareness of lifestyle-related diseases further supports the adoption of biomarker-based diagnostic solutions.
- For instance, Roche’s cobas® c 513 analyzer is capable of delivering up to 400 HbA1c test results per hour, allowing hospitals to rapidly identify patients at risk of metabolic disorders that predispose to NASH.
Shift Toward Non-Invasive Diagnostic and Monitoring Methods
Growing limitations of liver biopsy, including its invasive nature and risk of complications, are steering the market toward non-invasive biomarker alternatives. Patients and clinicians prefer biomarker-based solutions for safer, quicker, and more accurate assessments. It supports clinical adoption by improving patient compliance and reducing diagnostic delays. The focus on personalized medicine further enhances the demand for advanced biomarker platforms.
Expanding Pharmaceutical Research and Clinical Trials in NASH Therapeutics
Intensifying R&D efforts in the pharmaceutical industry are creating significant opportunities for biomarker adoption. Biomarkers play a central role in patient selection, disease staging, and treatment response monitoring during NASH clinical trials. The non alcoholic steatohepatitis biomarkers market benefits from rising investments in drug pipelines and the need for reliable surrogate endpoints. Collaborations between diagnostic firms and pharmaceutical companies are accelerating technology development.
- For instance, Pfizer leveraged advanced imaging-based biomarkers in its NASH clinical trial for PF-06865571, enrolling over 230 participants to assess liver fat reduction outcomes using MRI-PDFF technology.
Technological Advancements in Genomics, Proteomics, and Imaging
Continuous innovation in genomics, proteomics, metabolomics, and imaging is enhancing the accuracy of NASH biomarker platforms. These advancements enable precise identification of disease progression and treatment outcomes. It supports the shift toward evidence-based diagnosis and strengthens the role of biomarkers in clinical practice. Increasing integration of artificial intelligence and digital health tools further accelerates the adoption of innovative biomarker technologies.
Market Trends:
Growing Adoption of Non-Invasive and Composite Biomarker Panels
The non alcoholic steatohepatitis biomarkers market is witnessing a clear trend toward non-invasive diagnostic solutions that minimize patient risk and improve compliance. Composite biomarker panels that combine serum, imaging, and genetic markers are gaining traction for their ability to provide comprehensive insights into disease stage and progression. It supports healthcare providers in moving away from invasive liver biopsies, aligning with patient-centric care models. Companies are increasingly developing multimodal diagnostic platforms to enhance accuracy and reduce variability in results. Growing preference for early detection through such approaches is reshaping diagnostic protocols. The integration of biomarker panels into routine clinical practice is expected to accelerate over the forecast period.
- For instance, Perspectum’s LiverMultiScan platform has been adopted in over 500 clinical centers globally, providing a non-invasive, quantitative imaging-based biomarker solution that helps clinicians assess and monitor NASH patients without biopsy.
Integration of Artificial Intelligence and Digital Health in Biomarker Development
Advanced digital technologies are emerging as key enablers in biomarker innovation, with artificial intelligence and machine learning improving predictive capabilities. It is driving the development of algorithms that analyze vast datasets, including imaging and molecular profiles, to identify reliable biomarkers. The non alcoholic steatohepatitis biomarkers market is benefiting from partnerships between digital health firms and research organizations to accelerate validation processes. Wearable devices and digital monitoring tools are also being explored to track disease progression in real time. Pharmaceutical companies are leveraging these technologies to optimize clinical trial outcomes and patient stratification. Growing use of AI-driven insights is shaping the future of biomarker-based diagnostics and personalized treatment strategies.
- For instance, IBM Watson for Genomics delivered a comprehensive interpretation of a whole-genome sequencing dataset in just 10 minutes per patient, compared to 160 human-analysis hours required previously 10 minutes.
Market Challenges Analysis:
Limited Standardization and Validation of Biomarkers Across Clinical Settings
The non alcoholic steatohepatitis biomarkers market faces significant challenges due to the lack of standardized validation protocols. Biomarkers often produce variable outcomes across laboratories and patient groups, limiting their reliability in routine practice. It creates barriers for regulatory approval and widespread adoption. Healthcare providers remain cautious about replacing established diagnostic methods without stronger clinical evidence. The absence of universal guidelines for biomarker use further complicates integration into healthcare systems. These limitations slow progress in achieving global acceptance of biomarker-based diagnostics.
High Development Costs and Regulatory Hurdles for Commercialization
Developing and validating NASH biomarkers requires extensive investment in clinical research, advanced technologies, and multi-center trials. It places a financial burden on diagnostic companies, especially smaller players. Regulatory pathways for biomarker approval are complex and demand robust clinical evidence, extending timelines and increasing costs. Pharmaceutical collaborations provide some support, but commercialization challenges remain high. Market entry is further hindered by reimbursement uncertainties, as many payers remain reluctant to cover advanced biomarker tests. These challenges collectively restrain the speed of adoption across healthcare markets.
Market Opportunities:
Expanding Role of Biomarkers in Drug Development and Clinical Trials
The non alcoholic steatohepatitis biomarkers market is positioned for growth through its integration into pharmaceutical R&D pipelines. Biomarkers are increasingly used to identify suitable patients, predict therapeutic outcomes, and monitor treatment responses in NASH clinical trials. It supports faster drug development by reducing trial costs and improving efficiency. Regulatory agencies are encouraging the use of surrogate endpoints, creating opportunities for biomarker-based validation. Partnerships between diagnostic developers and pharmaceutical companies are expanding, fostering innovation and commercial adoption. This trend enhances the relevance of biomarkers as essential tools in precision medicine.
Growing Demand for Early Detection and Personalized Healthcare Solutions
Rising awareness of the importance of early diagnosis creates strong potential for biomarker adoption in routine practice. It enables healthcare providers to detect NASH before irreversible liver damage occurs, improving treatment outcomes. The non alcoholic steatohepatitis biomarkers market benefits from the global shift toward personalized healthcare, where patient-specific data guide medical decisions. Integration of digital health platforms and artificial intelligence offers further scope for innovation in disease monitoring. Emerging economies with rising healthcare investments present untapped opportunities for biomarker penetration. Expanding adoption in clinical settings positions biomarkers as critical tools in addressing the growing NASH burden worldwide.
Market Segmentation Analysis:
By Drug:
The non alcoholic steatohepatitis biomarkers market is segmented by drug into obeticholic acid, elafibranor, selonsertib, cenicriviroc, and others. Biomarkers play a critical role in evaluating drug efficacy and monitoring patient response during treatment. It supports pharmaceutical companies in identifying the most suitable candidates for clinical trials, reducing attrition rates, and enhancing regulatory compliance. Growing pipeline activity for NASH therapeutics is increasing the demand for reliable biomarker solutions. This segment will continue to expand as more therapies progress toward commercialization.
- For instance, Madrigal Pharmaceuticals’ Phase II trial of resmetirom integrated NASH biomarker profiling in 125 patients to correlate thyroid receptor activation with liver histology outcomes.
By Distribution Channel:
By distribution channel, the market is categorized into hospital-based laboratories, diagnostic laboratories, and academic research centers. Hospital laboratories dominate due to the availability of advanced testing infrastructure and direct access to patient populations. It strengthens adoption by offering integrated diagnostic services and collaboration opportunities with healthcare providers. Diagnostic laboratories are growing with rising demand for outsourced testing and cost-effective solutions. Academic research centers contribute by advancing biomarker validation and supporting drug development partnerships.
- For instance, Quest Diagnostics operates over 2,200 patient service centers in the U.S. and runs more than 50 state-of-the-art laboratories, with its Clifton, New Jersey lab capable of performing over 200,000 tests per day using advanced robotics-based platforms.
By Application:
The market by application includes diagnostic, prognostic, and therapeutic monitoring. Diagnostic applications hold the largest share due to the growing need for early detection of NASH. It drives clinical adoption by enabling non-invasive assessment of disease progression. Prognostic biomarkers are gaining importance for predicting outcomes and guiding long-term care strategies. Therapeutic monitoring applications are also expanding with increasing integration of biomarkers in drug trials and clinical practice. This diversification strengthens the role of biomarkers in precision healthcare.
Segmentations:
By Drug:
- Obeticholic Acid
- Elafibranor
- Selonsertib
- Cenicriviroc
- Others
By Distribution Channel:
- Hospital-Based Laboratories
- Diagnostic Laboratories
- Academic Research Centers
By Application:
- Diagnostic
- Prognostic
- Therapeutic Monitoring
By Region:
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East & Africa
Regional Analysis:
North America:
North America accounted for 42% market share in 2024, supported by advanced healthcare infrastructure and strong diagnostic adoption. The United States leads the region due to a high prevalence of obesity, diabetes, and metabolic syndrome, which are major risk factors for NASH. The non alcoholic steatohepatitis biomarkers market benefits from robust clinical research activities and active pharmaceutical pipelines in the country. It is also driven by favorable reimbursement policies and high patient awareness of non-invasive testing. Canada contributes through growing investments in liver disease research and supportive government initiatives. Increasing collaborations between diagnostic firms and academic institutions further strengthen regional growth.
Europe:
Europe held 28% market share in 2024, with Germany, the United Kingdom, and France leading adoption. Strong government funding for clinical trials and emphasis on precision medicine drive biomarker integration across healthcare systems. The non alcoholic steatohepatitis biomarkers market in Europe is supported by rising healthcare expenditures and structured screening programs. It is further driven by collaborations between pharmaceutical companies and diagnostic developers to accelerate biomarker validation. Germany remains a hub for clinical research, while the UK focuses on digital health integration in diagnostics. France emphasizes early diagnosis through public health programs, creating opportunities for biomarker-based solutions.
Asia-Pacific:
Asia-Pacific accounted for 21% market share in 2024, led by China, India, and Japan. Rising healthcare investments and growing patient populations with lifestyle-related disorders drive strong demand for advanced diagnostics. The non alcoholic steatohepatitis biomarkers market in this region benefits from increasing clinical adoption and government-led healthcare reforms. It is further fueled by a rising focus on early detection and expanding private healthcare infrastructure. China dominates due to large-scale investments in biotechnology, while India is advancing through improved diagnostic accessibility. Japan continues to lead in technological integration, particularly in imaging and proteomics. Expanding collaborations between local firms and global players are expected to boost regional penetration.
Shape Your Report to Specific Countries or Regions & Enjoy 30% Off!
Key Player Analysis:
- GENFIT
- Siemens Healthineers AG
- Prometheus Laboratories
- BioPredictive
- AstraZeneca
- Quest Diagnostics
- Exalenz Bioscience Ltd (meridian bioscience)
- Pfizer Inc.
- Labcorp
- Bristol-Myers Squibb Company
Competitive Analysis:
The non alcoholic steatohepatitis biomarkers market is highly competitive with a mix of diagnostic firms, pharmaceutical companies, and research-driven organizations shaping growth. Key players include GENFIT, Siemens Healthineers AG, Prometheus Laboratories, BioPredictive, AstraZeneca, and Quest Diagnostics. These companies focus on advancing biomarker platforms that enable early detection, accurate disease staging, and effective therapeutic monitoring. It is supported by strong R&D investments, strategic collaborations, and expanding clinical trial activities aimed at integrating biomarkers into NASH treatment pathways. Market leaders are also emphasizing the development of non-invasive solutions to reduce reliance on liver biopsy and improve patient compliance. Partnerships between pharmaceutical firms and diagnostic developers are central to accelerating validation and commercialization. Competitive intensity is further driven by the race to establish biomarkers as regulatory-approved endpoints in drug development, positioning the market for rapid innovation and long-term growth.
Recent Developments:
- In March 2025, GENFIT completed a non-dilutive royalty financing agreement with HealthCare Royalty (HCRx), securing up to €185 million with €130 million upfront to support the development of its pipeline, including ACLF therapies.
- In April 2025, Siemens Healthineers and Tower Health announced a 10-year Value Partnership to enhance medical equipment and technology modernization, focusing on upgrades in diagnostic imaging and digital intelligence tools across several departments.
- In August 2025, Prometheus Laboratories entered into a collaboration with Celltrion to advance Anser Testing for therapeutic drug monitoring in the treatment of inflammatory diseases.
Report Coverage:
The research report offers an in-depth analysis based on Drug, Distribution Channel, Application and Region. It details leading Market players, providing an overview of their business, product offerings, investments, revenue streams, and key applications. Additionally, the report includes insights into the competitive environment, SWOT analysis, current Market trends, as well as the primary drivers and constraints. Furthermore, it discusses various factors that have driven Market expansion in recent years. The report also explores Market dynamics, regulatory scenarios, and technological advancements that are shaping the industry. It assesses the impact of external factors and global economic changes on Market growth. Lastly, it provides strategic recommendations for new entrants and established companies to navigate the complexities of the Market.
Future Outlook:
- The non alcoholic steatohepatitis biomarkers market will expand with rising prevalence of obesity and diabetes.
- Non-invasive biomarker solutions will gain wider acceptance, reducing dependence on traditional liver biopsies.
- Pharmaceutical companies will increasingly integrate biomarkers into clinical trial design and drug development.
- Advancements in genomics, proteomics, and metabolomics will drive accuracy in disease detection and monitoring.
- Artificial intelligence and digital health platforms will enhance biomarker interpretation and predictive capabilities.
- Collaborations between diagnostic firms and pharmaceutical developers will accelerate innovation and commercialization.
- Emerging economies will present new opportunities as healthcare investments and patient awareness rise.
- Reimbursement approvals and supportive regulatory policies will strengthen biomarker adoption in clinical practice.
- Personalized medicine strategies will boost demand for biomarker-based patient stratification and treatment monitoring.
- Expanding partnerships among academic institutions, research organizations, and technology providers will shape future growth.




