Anthrax Vaccine Market Overview:
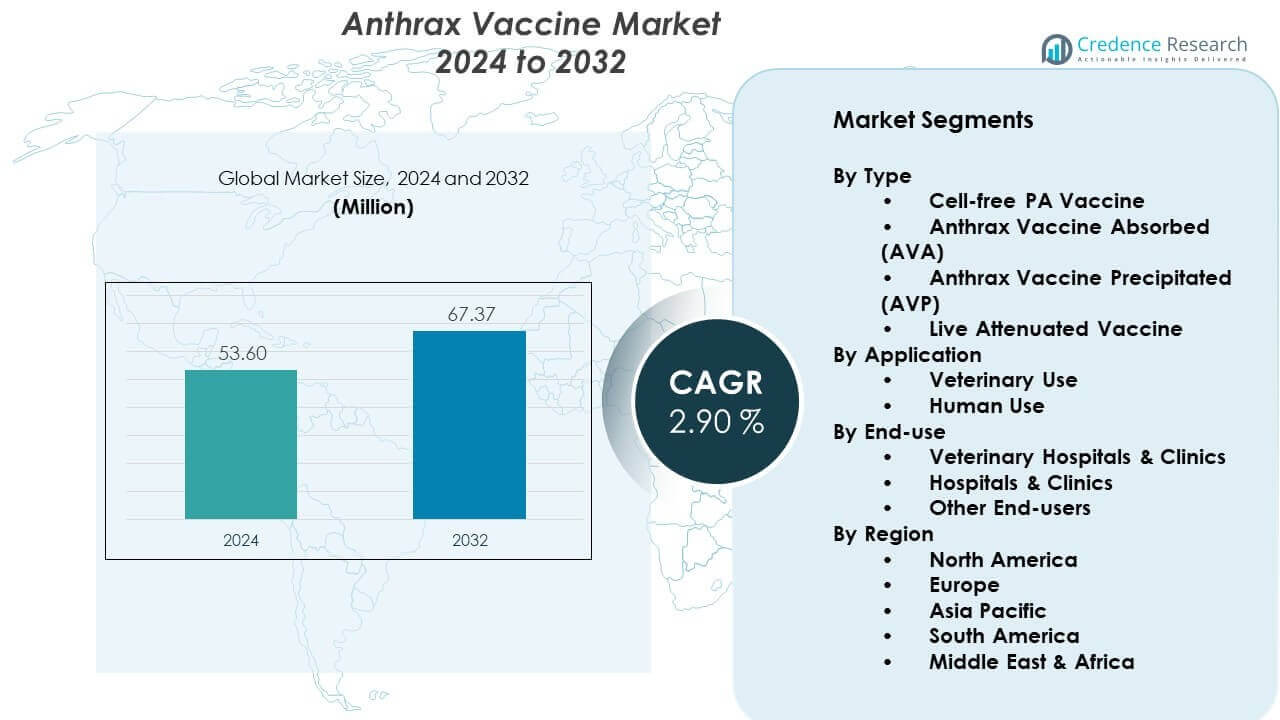
The Anthrax Vaccine Market is projected to grow from USD 53.6 million in 2024 to an estimated USD 67.37 million by 2032, with a CAGR of 2.90% from 2024 to 2032.
| REPORT ATTRIBUTE |
DETAILS |
| Historical Period |
2020-2023 |
| Base Year |
2024 |
| Forecast Period |
2025-2032 |
| Anthrax Vaccine Market Size 2024 |
USD 53.6 million |
| Anthrax Vaccine Market, CAGR |
2.90% |
| Anthrax Vaccine Market Size 2032 |
USD 67.37 million |
Growing concerns around biological threats and livestock-borne infections drive broad adoption of anthrax vaccines. Defense agencies are increasing procurement as global biodefense strategies evolve, while veterinary vaccination programs expand in high-risk regions. Advancements in recombinant vaccine technologies support stronger immune responses, longer protection, and improved safety. Partnerships between vaccine makers and government agencies also push innovation. Rising investments in disease surveillance and rapid-response capabilities continue to reinforce market expansion across human and veterinary health settings.
North America leads due to strong federal biodefense funding, advanced R&D capacity, and structured vaccination programs. Europe follows with solid regulatory support and consistent investments in biological threat preparedness. Asia Pacific continues to emerge as livestock-dense countries strengthen disease prevention frameworks and expand veterinary immunization coverage. Regions in Africa and the Middle East show growing demand due to recurring anthrax outbreaks and rising awareness. Global efforts to improve public health readiness help broaden adoption across both developed and developing countries.
Anthrax Vaccine Market Insights:
- The Anthra Vaccine Market reached USD 53.6 million in 2024 and is projected to hit USD 67.37 million by 2032, registering a CAGR of 2.90% driven by biodefense needs and veterinary immunization programs.
- North America (38%), Europe (30%), and Asia Pacific (22%) dominate due to strong public health systems, established vaccination protocols, and high livestock exposure risk, which support consistent procurement.
- Asia Pacific, holding 22%, is the fastest-growing region due to expanding veterinary vaccination campaigns, large livestock populations, and rising zoonotic awareness in rural areas.
- The veterinary application segment accounts for nearly 60% of total demand, supported by widespread vaccination in livestock-heavy regions facing recurring exposure threats.
- The human use segment holds about 40%, driven by structured immunization of military personnel, laboratory workers, and populations covered under biodefense preparedness programs.
Access crucial information at unmatched prices!
Request your sample report today & start making informed decisions powered by Credence Research Inc.!
Download Sample
Anthrax Vaccine Market Drivers:
Growing Focus on Biodefense Preparedness and Public Health Protection
Global agencies strengthen biodefense programs to reduce vulnerability to anthrax exposure. Nations invest in stronger surveillance systems that support timely containment. The Anthrax Vaccine Market benefits from policy support that encourages wider vaccine deployment. Research institutions reinforce development pipelines that target improved protection profiles. Governments expand strategic reserves to protect high-risk groups. Military forces receive structured vaccination schedules that drive steady demand. Veterinary programs scale immunization in livestock-heavy regions to curb outbreaks. Cross-border cooperation boosts knowledge sharing and builds confidence in advanced prevention tools.
Rising Threat of Zoonotic Transmission Across Endemic Regions
Zoonotic anthrax cases in rural and agricultural zones encourage broader immunization. Livestock producers adopt structured vaccination cycles to protect herds. It creates stable demand where disease prevalence remains high. Public health teams expand awareness programs that support higher uptake. Improved diagnostic capacity enables quick response during suspected events. Regulatory bodies encourage standardized protocols that guide safe vaccine use. International research groups share data on strain behavior that improves formulations. Regional governments prioritize preventive steps during seasonal risk periods.
- For instance, Zoetis Inc. distributes the Sterne Strain-based Anthrax Spore Vaccine, which is widely utilized in endemic regions of sub-Saharan Africa and the Middle East, requiring a 1 ml subcutaneous dose for cattle to provide protective immunity for a duration of 12 months.
Technical Advancements in Recombinant and Next-Generation Vaccine Platforms
Innovation accelerates development of recombinant platforms that offer stronger immune responses. Scientists pursue delivery methods that reduce dosing complexity. The Anthrax Vaccine Market gains traction through improvements that lower adverse event rates. Novel antigen technologies help widen protection coverage in diverse settings. Production methods advance to support higher throughput and controlled purity. It brings consistency that benefits procurement agencies. Research teams collaborate with public institutions to validate new mechanisms. Growing interest in long-acting formulations supports investment.
- For instance, GC Biopharma (formerly Green Cross) developed GC-1109, the world’s first recombinant protein-based anthrax vaccine to receive regulatory approval; clinical data submitted to the South Korean Ministry of Food and Drug Safety confirmed the vaccine achieved a 100% seroconversion rate in Phase II trials using a simplified three-dose schedule.
Strategic Collaborations Supporting Manufacturing Strength and Global Supply
Public-private partnerships reinforce stable supply for emergency programs. Manufacturers invest in capacity expansion that supports long-term contracts. Global procurement bodies secure agreements that reduce supply disruptions. The Anthrax Vaccine Market strengthens through improved distribution networks across remote regions. Logistical systems evolve to maintain cold-chain reliability under harsh conditions. It supports safe transport during crisis events or field deployment. Nations coordinate to share best practices for storage and delivery. Funding agencies stimulate innovation by promoting multi-year development grants.
Anthrax Vaccine Market Trends:
Shift Toward Broader Immunization Coverage in High-Risk Livestock Regions
Veterinary authorities evaluate new strategies that support large-scale herd vaccination. Producers gain stronger disease protection through structured programs. The Anthrax Vaccine Market expands due to consistent uptake in agricultural areas. It supports safer food systems and improved rural health outcomes. Awareness campaigns encourage farmers to follow standardized schedules. Data systems track infection patterns with improved accuracy. Field studies test novel delivery tools suited for remote locations. Regional alliances enhance training programs for animal health workers.
Rapid Strengthening of National Stockpiles and Emergency Deployment Protocols
Governments update preparedness frameworks to ensure uninterrupted supply during crises. Emergency response teams create clear distribution paths for rapid deployment. The Anthrax Vaccine Marketaligns with evolving defense strategies. It helps reduce response time during unexpected biological threats. Strategic reserves expand to cover broader population categories. Policy reforms support streamlined approval during urgent scenarios. Agencies conduct simulation exercises that refine operational protocols. New guidelines aim to unify standards across departments.
- For instance, the U.S. Food and Drug Administration (FDA) utilized the “Animal Rule” to grant approval to CYFENDUS in July 2023, allowing for a two-dose post-exposure prophylaxis regimen that enables the Strategic National Stockpile to protect a larger number of individuals with 33% fewer doses per person compared to the previous regimen.
Growing Adoption of Digital Tools in Monitoring and Post-Vaccination Assessment
Digital systems help track immunization cycles with greater transparency. Health teams utilize data dashboards to review risk zones. The Anthrax Vaccine Marketbenefits from better forecasting models. It supports informed planning for procurement cycles and emergency actions. Mobile tools assist field workers in recording vaccination outcomes. AI-based systems evaluate reporting trends that strengthen surveillance. Cloud platforms help integrate veterinary and human health records. Data-driven decisions improve efficiency in resource allocation.
Increasing Movement Toward Safer and More Stable Formulations for Harsh Environments
Scientists concentrate on thermostable variants that simplify transport in remote areas. Research groups explore adjuvants that enhance immune memory. The Anthrax Vaccine Market sees stronger interest in rugged formulations. It supports wider distribution in geographies with limited infrastructure. Advancements help reduce dependence on cold storage. New packaging designs withstand long field conditions. Pilot programs test compact vials for mobile units. Efforts aim to create solutions suited for diverse climates.
- For instance, researchers at the University of New Mexico, in collaboration with the National Institutes of Health (NIH), developed a protocell-based vaccine delivery system that maintains 100% antigen stability at temperatures up to 37°C for 30 days, specifically designed to bypass the traditional cold chain requirements for anthrax vaccines in tropical climates.
Anthrax Vaccine Market Challenges Analysis:
Regulatory Complexity and Slow Advancement of Novel Vaccine Platforms
Authorities maintain strict requirements that extend timelines for new approvals. Developers navigate complex documentation that demands detailed evidence. The Anthrax Vaccine Market advances under tight scrutiny from global regulators. It often faces long validation cycles that hinder rapid upgrades. Limited clinical trial populations reduce speed of data generation. Companies manage high compliance costs during each stage. Variability in regional standards complicates multinational submissions. Frequent updates in guidelines require constant adaptation.
Supply Constraints, Limited Awareness, and Logistical Gaps in Remote Regions
Manufacturers experience constraints due to specialized production steps. Distribution systems struggle in rural zones with limited cold-chain support. The Anthrax Vaccine Market encounters reduced uptake where awareness remains low. It depends on trained staff who remain scarce in certain countries. Outreach programs face difficulty in sustaining consistent engagement. Veterinary sectors lack unified reporting tools that track coverage. Market expansion slows where funding remains inconsistent. Health agencies continue to address infrastructure gaps.
Anthrax Vaccine Market Opportunities:
Expansion of Vaccination Programs in Emerging Agricultural Economies
Large livestock populations in developing regions create strong prospects for wider immunization. Producers seek structured protection strategies that help maintain herd safety. The Anthrax Vaccine Marketbenefits from rising veterinary modernization efforts. It gains momentum where governments promote disease-control incentives. Training initiatives help rural workers adopt better practices. New partnerships with agro-industry groups support awareness. Field-ready technologies simplify administration during high-risk seasons. Broader coverage strengthens long-term market stability.
Acceleration of R&D Toward Safer, Potent, and Thermostable Vaccine Solutions
Research teams explore advanced platforms that deliver improved immune durability. Innovations target simplified dosing methods for mobile deployment. The Anthrax Vaccine Marketsees value in next-generation solutions. It encourages investment that supports rapid prototype testing. Formulations designed for extreme climates expand global reach. Developers focus on scalable production to meet emergency demand. International agencies support funding to advance breakthroughs. Progress fosters new commercial pathways across human and veterinary domains.
Anthrax Vaccine Market Segmentation Analysis:
By Type
The Anthrax Vaccine Market divides into Cell-free PA Vaccine, Anthrax Vaccine Absorbed (AVA), Anthrax Vaccine Precipitated (AVP), and Live Attenuated Vaccine. Each type serves different protection needs across human and veterinary settings. Cell-free PA vaccines gain strong demand due to cleaner profiles and wider acceptance. AVA maintains steady use in military and high-risk groups. AVP supports targeted immunization where traditional methods remain preferred. Live attenuated vaccines hold importance in livestock-heavy regions with recurring outbreaks. It strengthens disease management where rapid immunity is required. Product diversification supports broader adoption across global programs.
- For instance, Emergent BioSolutions produces BioThrax, the only FDA-licensed Anthrax Vaccine Adsorbed (AVA), which requires a 3-dose primary series at 0, 1, and 6 months for pre-exposure prophylaxis and has been administered to over 3 million military personnel since its licensure.
By Application
This segment includes Veterinary Use and Human Use. Veterinary applications lead demand due to high exposure risk in livestock populations. It helps reduce transmission in agricultural regions and supports national disease-control mandates. Human use remains focused on defense personnel, laboratory workers, and outbreak-specific protection. Rising awareness in rural communities strengthens uptake. Both segments reflect different safety standards and delivery needs that shape procurement strategies.
- For instance, Colorado Serum Company produces the Anthrax Spore Vaccine for veterinary use, which utilizes the non-encapsulated Sterne Strain 34F2 to provide a protective immunity duration of 1 year in cattle, horses, sheep, and goats following a single annual dose.
By End-use
End-use demand covers Veterinary Hospitals & Clinics, Hospitals & Clinics, and Other End-users. Veterinary hospitals support large-scale vaccination drives and routine herd protection. Hospitals and clinics manage human immunization for occupational groups and emergency events. Other end-users include government agencies and research institutions that maintain vaccine reserves. It ensures continuous supply for strategic stockpiles and preparedness programs. Diverse end-use roles reinforce stable growth across global markets.
Segmentation:
By Type
- Cell-free PA Vaccine
- Anthrax Vaccine Absorbed (AVA)
- Anthrax Vaccine Precipitated (AVP)
- Live Attenuated Vaccine
By Application
By End-use
- Veterinary Hospitals & Clinics
- Hospitals & Clinics
- Other End-users
By Region
- North America
- Europe
- Germany
- France
- U.K.
- Italy
- Spain
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- South-east Asia
- Rest of Asia Pacific
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Middle East & Africa
- GCC Countries
- South Africa
- Rest of the Middle East and Africa
Regional Analysis:
North America
North America holds 38% of the Anthrax Vaccine Market due to strong federal biodefense programs and structured stockpiling systems. It benefits from advanced regulatory oversight and established vaccination practices for military and laboratory workers. Public agencies maintain coordinated preparedness frameworks that support stable procurement. Veterinary vaccination programs strengthen disease control in livestock regions with known exposure risks. High awareness and long-term government investment keep the region dominant. It continues to lead global adoption through consistent funding and robust supply capabilities.
Europe
Europe accounts for 30% of the global market, supported by strict public health policies and active disease-surveillance networks. The Anthrax Vaccine Market gains steady demand from standardized vaccination protocols in human and veterinary sectors. It benefits from coordinated initiatives across member states that target rural outbreak risks. Research institutions contribute to ongoing product advancements that strengthen the region’s preparedness. Southern and Eastern Europe report rising uptake driven by growing zoonotic threat awareness. Strong manufacturing presence and funding frameworks sustain market growth across the region.
Asia Pacific, South America, and Middle East & Africa
Asia Pacific holds 22% of the global share due to large livestock populations and expanding government-led vaccination programs. It strengthens prevention strategies in rural zones with limited healthcare access. South America captures 6%, influenced by agricultural reliance and periodic outbreaks in cattle-producing regions. Middle East & Africa hold 4%, where recurring anthrax cases and improving awareness stimulate demand. The Anthrax Vaccine Market expands in these regions through targeted disease-control efforts and improving vaccine accessibility. It supports broader public and veterinary health initiatives across developing economies.
Shape Your Report to Specific Countries or Regions & Enjoy 30% Off!
Key Player Analysis:
Competitive Analysis:
The Anthrax Vaccine Market features a concentrated competitive landscape led by established manufacturers with strong government contracts and defense partnerships. Companies strengthen their positions through regulatory approvals, capacity expansion, and long-term procurement agreements. It relies on trusted suppliers with proven manufacturing quality and reliable cold-chain systems. Players focus on improving vaccine purity, extending durability, and reducing adverse reactions. Firms expand veterinary portfolios to meet rising livestock protection needs. Growth accelerates through collaborations with public health agencies and military research units. Competitive momentum increases as firms invest in next-generation platforms that support higher safety and stability.
Recent Developments:
- In February 2026, Colorado Serum Company was highlighted as a leading player in the veterinary anthrax vaccine market, providing essential live-culture vaccines for animal use. The company’s specialized focus on high-quality, cost-effective livestock vaccines makes it a vital partner for agricultural biosecurity programs aimed at controlling outbreaks in domestic herds.
- In January 2026, Emergent BioSolutions announced it received a delivery order valued at up to $21.5 million from the U.S. Department of War to supply BioThrax® (Anthrax Vaccine Adsorbed) for military personnel at high risk of exposure. This order follows a December 2024 contract option from BARDA for $50 million to procure doses of CYFENDUS®, the company’s two-dose adjuvanted anthrax vaccine approved for post-exposure prophylaxis.
Report Coverage:
The research report offers an in-depth analysis based on By Type, By Application and By End-use. It details leading market players, providing an overview of their business, product offerings, investments, revenue streams, and key applications. Additionally, the report includes insights into the competitive environment, SWOT analysis, current market trends, as well as the primary drivers and constraints. Furthermore, it discusses various factors that have driven market expansion in recent years. The report also explores market dynamics, regulatory scenarios, and technological advancements that are shaping the industry. It assesses the impact of external factors and global economic changes on market growth. Lastly, it provides strategic recommendations for new entrants and established companies to navigate the complexities of the market.
Future Outlook:
- Growing global preparedness initiatives are expected to expand procurement programs and strengthen long-term demand across human and veterinary sectors.
- Advancements in recombinant platforms will support safer, more durable formulations that improve protection for high-risk groups and field operators.
- Expansion of livestock vaccination campaigns will help control outbreaks in agricultural regions and boost market penetration in developing economies.
- Strengthening stockpile strategies by defense and public health agencies will improve supply stability and encourage long-term contracts with manufacturers.
- Rising awareness of zoonotic transmission risks will drive broader adoption of structured immunization in rural communities.
- Development of thermostable and rugged vaccine variants will enable distribution in remote regions with limited cold-chain infrastructure.
- Strategic partnerships between vaccine developers and government agencies will accelerate innovation and streamline regulatory pathways.
- Digital systems will enhance tracking, post-vaccination assessment, and forecasting accuracy, improving response preparedness.
- Increased research funding will accelerate discovery of new antigen designs and immune-enhancing technologies, supporting next-generation solutions.
- Growing emphasis on coordinated global health security will reinforce demand for vaccines that support both preventive and rapid-response frameworks.




