Biobank Market Market Overview:
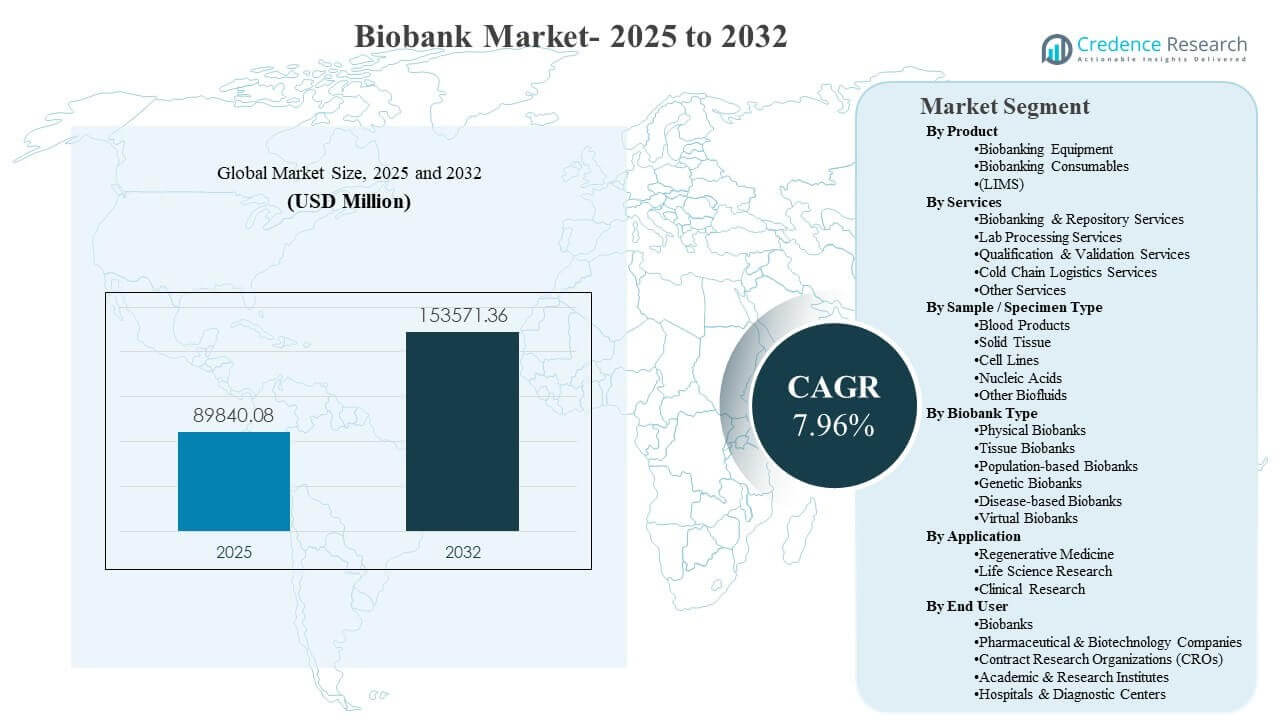
The global Biobank Market size was estimated at USD 89,840.08 million in 2025 and is expected to reach USD 153,571.36 million by 2032, growing at a CAGR of 7.96% from 2025 to 2032. Growth is primarily driven by the expanding volume of biospecimens required for precision medicine, biomarker-led drug development, and large-scale population health studies that depend on standardized collection and long-term preservation. Biobank Market demand is further supported by increased adoption of digitized inventory, chain-of-custody controls, and temperature-stable cold storage infrastructure across research and clinical ecosystems.
| REPORT ATTRIBUTE |
DETAILS |
| Historical Period |
2020-2024 |
| Base Year |
2025 |
| Forecast Period |
2026-2032 |
| Biobank Market Size 2025 |
USD 89,840.08 million |
| Biobank Market, CAGR |
7.96% |
| Biobank Market Size 2032 |
USD 153,571.36 million |
Key Market Trends & Insights
- The global Biobank Market size was estimated at USD 89,840.08 million in 2025 and is projected to reach USD 153,571.36 million by 2032, expanding at a 7.96% CAGR (2025–2032).
- Europe accounted for 35.5% of Biobank Market revenue in 2025, supported by mature academic networks and structured biorepository programs.
- North America represented 31.0% share in 2025, reflecting strong biopharma R&D activity and sample-intensive clinical research.
- Asia Pacific held 25.8% share in 2025, indicating rapid scale-up of infrastructure aligned with genomics and translational research priorities.
- Solid Tissue accounted for the largest share of 36.4% in 2025 within sample/specimen demand, highlighting sustained reliance on tissue-based translational programs.
Segment Analysis
Biobank Market demand is shaped by a combined need for high-integrity sample preservation and high-throughput operational efficiency across research and clinical workflows. Temperature-controlled storage systems, standardized consumables, and robust information management platforms are increasingly deployed together to protect sample quality, reduce handling variability, and ensure audit readiness across distributed repositories. Biobank Market buyers place growing emphasis on end-to-end traceability, consistent metadata capture, and scalable storage capacity as collections expand in size and diversity.
Biobank Market segmentation also reflects the rising influence of advanced therapies and biomarker-driven development. Programs linked to regenerative medicine, multi-omics research, and clinical research require strict control of pre-analytical variables, validated processing steps, and reliable cold-chain logistics from collection sites to repositories. As a result, Biobank Market service models that combine repository management, lab processing, qualification/validation, and controlled logistics are increasingly used to meet performance and compliance expectations.
By Product Insights
Biobanking Equipment remains the most central product group in Biobank Market purchasing decisions due to its direct role in maintaining sample stability across long storage durations. Temperature control systems, alarms/monitoring, and accessories are prioritized to reduce temperature excursions and strengthen compliance documentation. Biobank Market adoption of laboratory information management systems also rises as repositories scale, since digital inventory and chain-of-custody capabilities improve sample discoverability and governance. Biobanking consumables continue to benefit from recurring usage tied to collection volumes and standardized processing workflows.
By Services Insights
Biobanking & Repository services typically lead Biobank Market service demand because biorepository operations require continuous governance, sample integrity controls, and standardized processes across multi-site networks. Lab processing support is increasingly critical as studies demand consistent pre-analytical handling and faster turnaround times. Qualification/Validation services strengthen operational reliability by ensuring storage systems and workflows remain aligned with quality expectations. Cold chain logistics is gaining strategic importance as studies expand geographically and require controlled transport for sensitive specimens.
By Sample / Specimen Type Insights
Solid Tissue accounted for the largest share of 36.4% in 2025. Solid tissue specimens remain highly valued for translational research because tissue-linked clinical context supports biomarker validation and disease characterization. Biobank Market demand for tissue storage also increases with growth in oncology research, immunology programs, and advanced molecular profiling. Standardized collection protocols and improved preservation methods further strengthen the role of solid tissue in long-term research and clinical evidence generation.
By Biobank Type Insights
Physical Biobanks form the operational backbone of Biobank Market activity because centralized infrastructure supports standardized storage, controlled access, and long-term governance. Tissue biobanks and disease-based biobanks remain important due to deep specialization and tighter alignment with clinical pathways. Population-based and genetic biobanks continue to scale as longitudinal research expands, increasing demand for consistent data capture and sample annotation. Virtual biobanks are gaining relevance by improving discoverability and enabling streamlined sample request workflows across distributed repositories.
By Application Insights
Regenerative Medicine accounted for the largest share of 35.9% in 2025. Regenerative medicine demand supports Biobank Market growth by requiring high-integrity biospecimens and controlled storage conditions aligned with advanced therapy development. Biobank Market workflows in regenerative medicine also emphasize traceability, standardized processing, and validated logistics to protect sensitive biological materials. Life science research and clinical research continue to expand biobanking utilization by increasing sample volumes and driving broader adoption of standardized repository practices.
By End User Insights
Biobanks remain a primary end user group in Biobank Market because long-term storage, governance, and standardized access systems are core requirements for repository operations. Pharmaceutical & biotechnology companies drive demand through biomarker-led development and sample-intensive clinical programs that require reliable sourcing, storage, and documentation. Contract research organizations (CROs) strengthen adoption through outsourced trial execution and centralized sample workflows. Academic & research institutes and hospitals & diagnostic centers support steady utilization via translational studies, cohort research, and integrated clinical research initiatives.
Biobank Market Drivers
Expansion of precision medicine and biomarker-led development
Biobank Market growth is driven by rising use of biomarkers to stratify patients, validate targets, and monitor treatment response across drug development programs. Precision medicine approaches increase demand for high-quality biospecimens with consistent pre-analytical handling and reliable long-term storage. Biobank Market workflows increasingly require traceable sample provenance, standardized metadata capture, and robust quality controls to support reproducibility. As sample volumes scale, repositories invest in storage capacity, monitoring systems, and interoperable data tools to improve utilization.
- For instance, the UK Biobank is archiving samples from 500,000 participants with approximately 15 million aliquots split between a −80°C automated archive holding about 9.5 million samples and a −180°C manual liquid nitrogen archive holding about 5.5 million samples, all managed through an integrated laboratory information management system to ensure standardized biomarker-ready material for large-scale precision medicine studies.
Growth in clinical research, longitudinal cohorts, and population health programs
Biobank Market demand increases as longitudinal cohort studies expand and require structured biospecimen collection across large populations. Clinical research relies on consistent sample processing and storage to reduce variability across sites and time periods. Biobank Market infrastructure supports these programs through validated repository operations, standardized lab processing, and controlled logistics that preserve integrity. Larger study scale and longer follow-up periods also increase the need for stable storage and governance practices that can be sustained over many years.
- For instance, the NIH All of Us Research Program Biobank, operated with Mayo Clinic, has processed more than 2.3 million primary tubes into over 9.0 million secondary aliquots from more than 334,000 biospecimen-contributing participants, with long-term plans to store around 35 million biospecimens (roughly 35 samples per participant) to support decades-long longitudinal precision health research.
Rising adoption of automation, standardization, and quality management
Biobank Market buyers increasingly prioritize automation to improve throughput and reduce manual handling errors across collection, processing, and storage workflows. Standardized processes strengthen repeatability and improve confidence in downstream analytics, supporting broader adoption across research networks. Biobank Market investment in alarms, monitoring systems, and audit-ready documentation helps reduce temperature-related risks and supports compliance expectations. As repositories scale, integrated information management tools become more important for inventory control, chain-of-custody, and controlled access governance.
Increasing relevance of advanced therapies and regenerative medicine pipelines
Biobank Market expansion is supported by growth in cell therapy, gene therapy, and tissue engineering programs that require sensitive materials to be preserved under tightly controlled conditions. Regenerative medicine workflows place high emphasis on traceability, validated storage protocols, and controlled transportation to protect material integrity. Biobank Market service demand increases as sponsors seek reliable repository partners to manage complex sample lifecycles. Increased clinical translation further raises requirements for documentation, consistency, and long-term sample availability for follow-up analyses.
Biobank Market Challenges
Biobank Market operations face persistent challenges linked to variability in collection protocols, pre-analytical handling, and metadata completeness across multi-site studies. Inconsistent procedures can reduce comparability and limit downstream analytical value, especially for multi-omics workflows that are sensitive to sample quality. Biobank Market stakeholders also manage governance complexity related to consent frameworks, data access rules, and cross-border sample transfer limitations. These constraints can slow study execution and reduce the usable pool of samples.
- For instance, an applied quality-control study at the Biobank of the Medical University of Bialystok demonstrated that introducing molecular QC thresholds for DNA integrity number (DIN ≥ 7) and RNA integrity number (RIN ≥ 7) enabled them to classify and qualify thousands of stored samples specifically for omics-grade use, reducing the proportion of unusable biospecimens in multi-omics projects.
Biobank Market scalability is challenged by high infrastructure and operating costs for long-term cold storage, system redundancy, and continuous monitoring. Temperature excursions, equipment downtime, and logistics disruptions can create integrity risks that require strict preventive controls. Biobank Market repositories also face pressure to maintain audit readiness and standardized documentation without increasing processing delays. Talent constraints in specialized biorepository operations and informatics can further complicate expansion plans and technology upgrades.
Biobank Market Trends and Opportunities
Biobank Market trends increasingly emphasize digitalization and interoperability to improve sample discoverability and governance across distributed networks. Adoption of information management tools supports standardized inventory, chain-of-custody tracking, and controlled access workflows that improve utilization rates. Biobank Market participants are also investing in tighter monitoring and analytics for temperature performance and compliance reporting. These capabilities improve reliability and support broader participation of hospitals and research centers in multi-site programs.
- For instance, quality control work at Synlab SDN Biobank shows that plasma microRNA profiles remain stable when samples are stored at −80 °C for five years compared with freshly processed controls, illustrating how rigorously monitored biobanks can preserve molecular integrity over long-term, multi-year studies.
Biobank Market opportunities are expanding in service-led models that combine repository management, lab processing, qualification/validation, and controlled logistics under unified quality frameworks. Centralized and hybrid networks can help study sponsors standardize procedures across geographies and reduce operational friction. Biobank Market growth also benefits from increasing use of advanced molecular profiling that raises the value of well-annotated samples. Partnerships that link biobanks with clinical systems and research consortia can further increase demand for scalable infrastructure and data-rich specimens.
Regional Insights
North America
North America represented 31.0% of Biobank Market revenue in 2025, supported by strong biopharma research activity and high adoption of sample-intensive clinical research workflows. Biobank Market demand in North America is reinforced by broad use of biomarker-driven development and translational research programs that require high-quality specimens. Storage modernization, automation adoption, and greater emphasis on compliance-driven monitoring systems continue to shape procurement priorities. Service outsourcing also increases as sponsors seek standardized processing and repository governance across multi-site trials.
Europe
Europe accounted for 35.5% of Biobank Market revenue in 2025, reflecting a strong concentration of academic biobanking networks and structured repository initiatives. Biobank Market strength in Europe is linked to long-running population studies and collaborative research infrastructure that prioritizes standardized consent and sample governance. Repositories in Europe continue to expand digital inventory capabilities to improve sample access and utilization. Increased focus on interoperability and quality systems also supports demand for informatics platforms and validated operational services.
Asia Pacific
Asia Pacific held 25.8% of Biobank Market revenue in 2025, driven by expanding healthcare research capacity and scaling genomics and translational programs. Biobank Market growth in Asia Pacific benefits from rising investment in research infrastructure and increasing participation in multi-site clinical studies. Demand for temperature-stable storage, modern monitoring systems, and standardized processing continues to increase as repositories scale. Regional collaboration and expanded biopharma pipelines support ongoing investment in both equipment and service-led repository models.
Latin America
Latin America represented 5.2% of Biobank Market revenue in 2025, supported by growing clinical research participation and the gradual expansion of formal biorepository infrastructure. Biobank Market development in Latin America is shaped by demand for standardized sample handling and improved cold-chain logistics across distributed collection sites. Repositories increasingly prioritize reliability, documentation, and consistent processing to improve suitability for multi-center studies. Partnerships with research institutions and service providers can accelerate modernization of storage systems and inventory management.
Middle East & Africa
Middle East & Africa accounted for 2.5% of Biobank Market revenue in 2025, reflecting early-stage scaling of structured biobanking initiatives and research infrastructure. Biobank Market progress in Middle East & Africa is supported by targeted investments in healthcare systems, academic programs, and national research initiatives. Demand growth is linked to expanding focus on disease registries, population health studies, and translational research capabilities. Improved governance frameworks and controlled logistics capabilities can further strengthen repository reliability and increase participation in global studies.
Competitive Landscape
Biobank Market competition is shaped by breadth of workflow coverage across storage systems, monitoring, consumables, automation, logistics support, and informatics integration. Vendors differentiate through reliability, temperature stability performance, monitoring and audit-readiness capabilities, and compatibility with standardized biorepository protocols. Service providers compete on repository governance, validated processing, and end-to-end cold-chain execution aligned with research and clinical requirements. Partnerships and acquisitions remain important routes to expand geographic footprint, add specialized capabilities, and strengthen integrated offerings.
Thermo Fisher Scientific Inc. is positioned around end-to-end laboratory and biorepository infrastructure that supports collection, processing, and storage needs across research and clinical environments. Thermo Fisher Scientific Inc. differentiation is typically built on broad portfolio coverage, integration across workflows, and alignment with quality and compliance expectations required by large repositories. Thermo Fisher Scientific Inc. also benefits from scale in lab infrastructure categories that support biobank standardization and long-term operating consistency. Thermo Fisher Scientific Inc. focus on scalable cold storage and supporting lab systems aligns with repositories that need repeatable performance as sample volumes expand.
The industry research and growth report includes detailed analyses of the competitive landscape of the market and information about key companies, including:
Qualitative and quantitative analysis of companies has been conducted to help clients understand the wider business environment as well as the strengths and weaknesses of key industry players. Data is qualitatively analyzed to categorize companies as pure play, category-focused, industry-focused, and diversified; it is quantitatively analyzed to categorize companies as dominant, leading, strong, tentative, and weak.
Shape Your Report to Specific Countries or Regions & Enjoy 30% Off!
Recent Developments
- In December 2024, Merck KGaA (Merck Group) signed a definitive agreement to acquire HUB Organoids Holding B.V., a move aimed at advancing its next‑generation biology portfolio and organoid-based services, thereby enriching upstream sample and model systems that complement Merck’s solutions for biobanking and translational research workflows.
- In February 2025, Thermo Fisher Scientific Inc. announced an expanded strategic partnership with Sanofi that includes the acquisition of Sanofi’s sterile drug product manufacturing site in Ridgefield, New Jersey, strengthening its U.S. capacity to support pharma and biotech customers and indirectly bolstering its position in sample-based and biologics supply chains relevant to the biobank market.
- In December 2025, QIAGEN N.V. completed the acquisition of Parse Biosciences and, in its January 2026 strategic update, highlighted this deal as expanding its sample technologies portfolio into single‑cell analysis, reinforcing QIAGEN’s role in high‑throughput sample processing and data‑rich applications that are increasingly integrated with modern biobank infrastructures.
- In February 2026, Becton, Dickinson and Company (BD) completed the previously announced sale of its biosciences and diagnostics division to Waters Corporation for about USD 17.5 billion, a major portfolio reshaping that refocuses BD on its core medical device and infusion technologies while altering its participation in some analytics and lab segments that interface with biobanking operations.
Report Scope
| Report Attribute |
Details |
| Market size value in 2025 |
USD 89840.08 million |
| Revenue forecast in 2032 |
USD 153571.36 million |
| Growth rate (CAGR) |
7.96% (2025–2032) |
| Base year |
2025 |
| Forecast period |
2025-2032 |
| Quantitative units |
USD million |
| Segments covered |
By Product Outlook: Biobanking Equipment (Temperature Control Systems [Freezers & Refrigerators, Cryogenic Storage Systems, Thawing Equipment], Incubators & Centrifuges, Alarms & Monitoring Systems, Accessories & Other Equipment), Biobanking Consumables, Laboratory Information Management Systems; By Services Outlook: Biobanking & Repository, Lab processing, Qualification/ Validation, Cold Chain Logistics, Other Services; By Sample / Specimen Type Outlook: Blood Products, Solid Tissue, Cell Lines, Nucleic Acids, Other Biofluids; By Biobank Type Outlook: Physical Biobanks, Tissue Biobanks, Population-based Biobanks, Genetic Biobanks, Disease-based Biobanks, Virtual Biobanks; By Application Outlook: Regenerative Medicine, Life Science Research, Clinical Research; By End User Outlook: Biobanks, Pharmaceutical & Biotechnology Companies, Contract Research Organizations (CROs), Academic & Research Institutes, Hospitals & Diagnostic Centers |
| Regional scope |
North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
| Key companies profiled |
Thermo Fisher Scientific Inc.; Merck KGaA; QIAGEN N.V.; Becton, Dickinson and Company; Hamilton Company; Tecan Trading AG; Avantor; Azenta; Cryoport; STEMCELL Technologies; Charles River Laboratories; Lonza; Taylor-Wharton; PHC Holdings Corporation |
| No.of Pages |
340 |
Segmentation
By Product
- Biobanking Equipment
- Temperature Control Systems
- Freezers & Refrigerators
- Cryogenic Storage Systems
- Thawing Equipment
- Incubators & Centrifuges
- Alarms & Monitoring Systems
- Accessories & Other Equipment
- Biobanking Consumables
- Laboratory Information Management Systems
By Services
- Biobanking & Repository
- Lab processing
- Qualification/ Validation
- Cold Chain Logistics
- Other Services
By Sample / Specimen Type
- Blood Products
- Solid Tissue
- Cell Lines
- Nucleic Acids
- Other Biofluids
By Biobank Type
- Physical Biobanks
- Tissue Biobanks
- Population-based Biobanks
- Genetic Biobanks
- Disease-based Biobanks
- Virtual Biobanks
By Application
- Regenerative Medicine
- Life Science Research
- Clinical Research
By End User
- Biobanks
- Pharmaceutical & Biotechnology Companies
- Contract Research Organizations (CROs)
- Academic & Research Institutes
- Hospitals & Diagnostic Centers
By Region
- North America
- Europe
- Germany
- France
- U.K.
- Italy
- Spain
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- South-east Asia
- Rest of Asia Pacific
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Middle East & Africa
- GCC Countries
- South Africa
- Rest of the Middle East and Africa




