Aseptic Transfer System Market Overview:
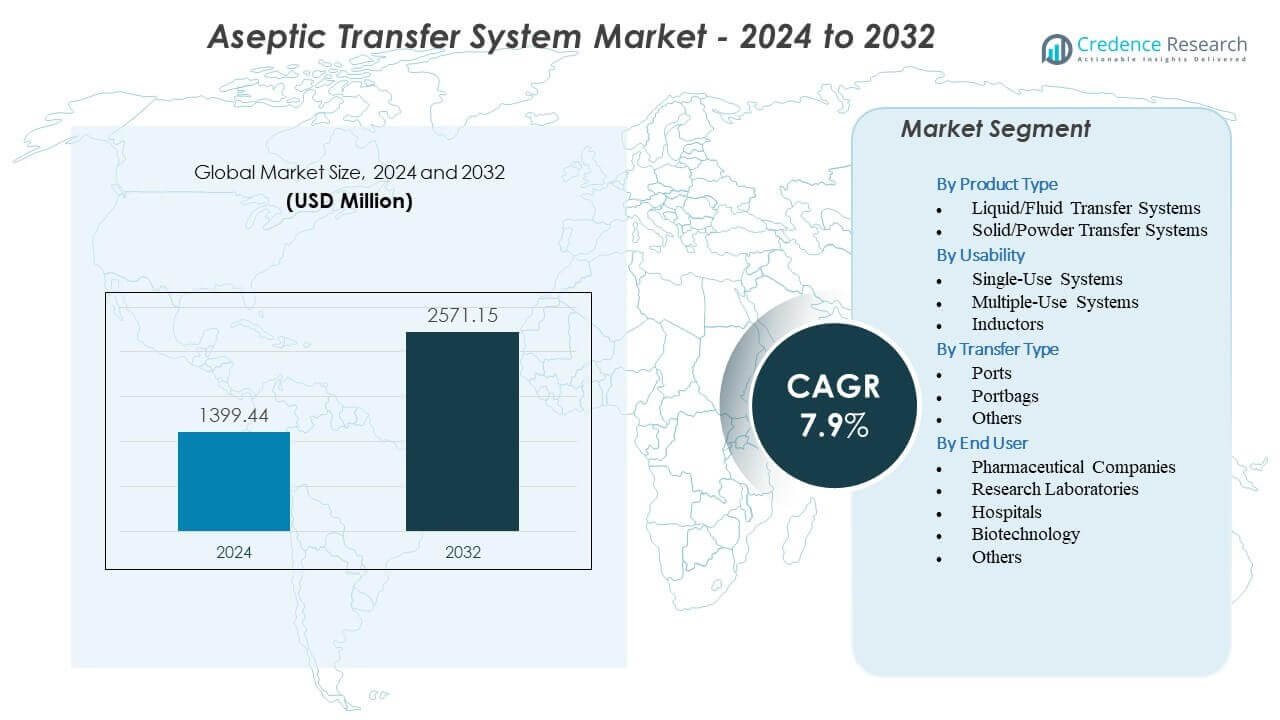
The Aseptic Transfer System Market is projected to grow from USD 1399.44 million in 2024 to an estimated USD 2571.15 million by 2032, with a compound annual growth rate (CAGR) of 7.9% from 2024 to 2032.
| REPORT ATTRIBUTE |
DETAILS |
| Historical Period |
2020-2023 |
| Base Year |
2024 |
| Forecast Period |
2025-2032 |
| Aseptic Transfer System Market Size 2024 |
USD 1399.44 million |
| Aseptic Transfer System Market, CAGR |
7.9% |
| Aseptic Transfer System Market Size 2032 |
USD 12571.15 9515 million |
The market grows as biopharmaceutical companies intensify focus on contamination-free material transfer in sterile operations. Rising output of biologics, vaccines, and high-potency medicines increases dependence on controlled transfer pathways that limit human contact. Facilities adopt single-use ports and transfer assemblies to accelerate batch turnover and reduce cleaning needs. The shift toward isolators, RABS, and modular manufacturing boosts adoption of engineered transfer solutions that enhance sterility and reduce variability. Strong regulatory pressure, especially through global GMP frameworks, encourages investment in validated and automated transfer designs.
North America leads the market due to its advanced sterile manufacturing infrastructure, strong regulatory enforcement, and expanding biologics production capacity. Europe follows with high adoption driven by Annex 1 revisions and extensive aseptic filling and vaccine operations across key countries. Asia Pacific emerges as the fastest-growing region supported by expanding biomanufacturing clusters in China, India, South Korea, and Singapore. Latin America and the Middle East show rising adoption as facilities modernize sterile workflows. Growing investment in high-purity manufacturing strengthens global demand across all regions.
Aseptic Transfer System Market Insights:
- The Aseptic Transfer System Market is projected to grow from USD 1399.44 million in 2024 to USD 2571.15 million by 2032, supported by a 7.9% CAGR driven by rising sterile manufacturing needs.
- Market growth is fueled by stronger demand for contamination-free transfer solutions, wider adoption of closed and single-use systems, and increased biologics and vaccine production.
- Restraints emerge from high integration costs, complex validation requirements, and limited component standardization that slows seamless interoperability across facilities.
- North America leads the market due to mature sterile manufacturing infrastructure and strong regulatory compliance culture, while Europe maintains steady demand driven by Annex 1 alignment.
- Asia Pacific shows the fastest expansion supported by growing biomanufacturing clusters, rising investment in modular plants, and increased adoption of scalable transfer systems.
Access crucial information at unmatched prices!
Request your sample report today & start making informed decisions powered by Credence Research Inc.!
Download Sample
Aseptic Transfer System Market Drivers
Growing Need for High-Purity Transfer Solutions Across Biopharma and Sterile Manufacturing
The Aseptic Transfer System Market expands due to rising production of sterile injectables and biologics. Drug makers focus on contamination control across every step, which pushes adoption of enclosed transfer solutions. Global manufacturers invest in contamination-free workflows to meet strict regulatory expectations. It strengthens the shift toward controlled material movement between clean zones. Single-use systems gain traction because they reduce validation time. Many plants integrate these systems to support higher batch flexibility. It improves product safety while supporting faster changeovers. Production units use these systems to maintain sterility in complex operations.
Rising Compliance Demands and Enforcement of Global GMP and Annex Standards
Regulatory agencies enforce stronger requirements for contamination prevention in sterile facilities, which supports the Aseptic Transfer System Market. Companies face higher pressure to validate every transfer step. It drives new investments in transfer ports, rapid transfer bags, and barrier systems. GMP and Annex 1 guidelines push firms to adopt closed and semi-closed transfer models. Validation teams rely on automated designs to reduce human error. Many sites replace outdated manual processes to strengthen compliance. It encourages wider uptake of engineered transfer mechanisms. Facilities adopt structured systems to maintain predictable sterility performance.
- For instance, Getinge’s DPTE® BetaBags undergo validation cycles proving sterility assurance levels (SAL) of 10⁻⁶ for aseptic loading and unloading.
Expansion of Biologic and Vaccine Manufacturing Pipelines Worldwide
Growing pipelines in biologics, vaccines, cell therapies, and sterile injectables create rising demand for advanced transfer solutions, which supports the Aseptic Transfer System Market. New modalities need strict handling of components and consumables. It encourages developers to shift toward high-containment systems that protect sensitive materials. Vaccine facilities deploy improved transfer ports for faster turnaround. Many sites require scalable transfer systems that match rising batch volumes. Drug makers invest in modular plants that depend on flexible transfer pathways. It improves overall workflow control during complex production cycles. The growth of sterile therapies elevates demand for precision transfer equipment.
- For instance, ABC Transfer’s closed-system ports are certified for over 5,000 sterile docking cycles while maintaining full containment performance.
Adoption of Automation and Robotics for Enhanced Sterility Assurance
Automation reshapes sterile manufacturing workflows, which supports the Aseptic Transfer System Market. Robotic handling reduces personnel involvement, which lowers contamination risk. It enables predictable and consistent material movement. Many companies deploy robotics to meet new production capacity targets. Automated docking units improve environmental control during transfers. Facilities integrate digital monitoring to verify transfer integrity. It helps limit human error in sterile zones. Automation supports repeatable operations for high-throughput facilities.
Aseptic Transfer System Market Trends
Increasing Shift Toward Single-Use Transfer Technologies Across Sterile Production
Demand for single-use transfer bags, ports, and connectors grows due to rising flexibility needs in the Aseptic Transfer System Market. Many facilities adopt disposable transfer routes to reduce cleaning steps. It supports faster batch changeovers in multi-product plants. Single-use systems reduce cross-contamination risk. They also align with rapid facility scale-up needs. Many biotech plants expand disposable integration across upstream and downstream workflows. It strengthens sterility assurance without heavy infrastructure changes. Growing demand for agile manufacturing boosts this trend.
- For instance, Sartorius’ Flexsafe® bags are validated for more than 10,000-fold flex cracking cycles, ensuring mechanical durability in high-frequency transfer workflows.
Growing Integration of Closed Transfer Systems With Isolators and RABS Platforms
A strong trend emerges toward closed and semi-closed models that reduce operator contact, which shapes the Aseptic Transfer System Market. Facilities link transfer systems to isolators for controlled drug handling. It improves sterility protection and reduces surface exposure. Many companies redesign layouts to embed transfer systems into barrier platforms. Automated docking creates stronger seal integrity. It enables safer transfer of high-risk materials. Adoption rises in vaccine and biologics units. Many expansion projects include barrier-linked transfer solutions as a standard practice.
Advancement of Sensor-Enabled and Digitally Monitored Transfer Modules
Digital technologies reshape quality assurance across sterile workflows, which influences the Aseptic Transfer System Market. Sensor-enabled units help operators track integrity in real time. It supports early detection of seal faults. Facilities adopt data-driven dashboards to monitor sterility indicators. Smart transfer ports improve operational visibility. Many manufacturers integrate digital proof logs for audits. It improves documentation accuracy. This trend advances predictive quality management across sterile plants.
Strong Movement Toward Modular and Rapid-Deployment Sterile Manufacturing Units
Modular cleanrooms and POD-based manufacturing units gain traction worldwide, which impacts the Aseptic Transfer System Market. These modular plants need compact and flexible transfer systems. It supports plug-and-produce configurations in expansion projects. Many companies choose systems that enable quick installation. Modular setups help reduce facility build time. Transfer solutions with integrated docking accelerate process readiness. It gives manufacturers a fast start for new product pipelines. Modular adoption increases due to global vaccine and biologics scale-up needs.
- For instance, Cytiva’s KUBio modular units allow operational readiness in less than 18 months, compared to multi-year traditional builds
Aseptic Transfer System Market Challenges Analysis
High Integration Costs and Complex Validation Requirements Across Regulated Facilities
High capital costs and complex qualification processes remain persistent hurdles for sterile plants using the Aseptic Transfer System Market. Facilities often manage lengthy validation cycles for each transfer pathway. It raises operational budgets for many manufacturers. Integration with isolators and RABS demands strong engineering support. Many plants require layout redesigns that delay deployment. Training requirements increase due to system complexity. It challenges smaller companies with limited technical teams. High compliance burdens slow broader market penetration.
Limited Standardization and Compatibility Constraints Across Transfer Components
Lack of standard dimensions and designs across transfer suppliers creates compatibility issues for sterile plants in the Aseptic Transfer System Market. Many operators face challenges aligning ports, bags, and connectors from different vendors. It disrupts workflow continuity. Facilities handle increased procurement complexity due to supplier variation. Engineering teams spend more time customizing interfaces. It restricts rapid scaling during peak demand. Many firms seek harmonized designs to stabilize their operations. Compatibility gaps slow cross-facility standardization.
Aseptic Transfer System Market Opportunities
Accelerated Growth of High-Potency Drug Manufacturing and Advanced Therapy Platforms
High-potency APIs, biologics, and next-generation therapies expand global sterile production needs, which supports new opportunities in the Aseptic Transfer System Market. Many of these products require strong containment solutions. It increases demand for closed transfer systems with enhanced sealing. Companies adopt engineered transfer routes to handle sensitive ingredients. Growth of ADCs and cell therapies strengthens interest in contamination-free transfer models. Many plants explore high-containment expansion projects. It opens doors for innovative transfer designs that improve protection. Global expansion in specialty therapeutics drives new investments.
Rising Adoption of Single-Use and Flexible Manufacturing Facilities Across Global Markets
Global drug makers expand flexible plants that depend on rapid deployment of single-use transfer systems, which supports major opportunities in the Aseptic Transfer System Market. Many facilities increase their reliance on single-use docking options. It helps reduce cleaning and turnaround cycles. Production units adopt lightweight components for faster installation. Many firms value modular integration across multiple process steps. It enhances operational agility in multi-product sites. Growing investments in disposable manufacturing strengthen supplier demand. It sets the stage for scalable growth in emerging regions.
Aseptic Transfer System Market Segmentation Analysis:
By Product Type
Liquid and fluid transfer systems hold strong adoption due to their role in sterile drug production, vaccine handling, and biologics manufacturing. Many facilities prefer these systems because they maintain controlled transfers under high-purity conditions. Solid and powder transfer systems support tablet, API, and lyophilized product workflows that need protected movement between clean zones. It strengthens workflow integrity in high-potency environments where particulate control is critical. Many companies invest in both formats to support multi-product operations. Growing biologics and oncology pipelines increase demand for controlled transfer designs. The Aseptic Transfer System Market benefits from broader adoption of integrated product-type architectures that support sterility and operational reliability.
- For instance, Dec Group’s PTS Powder Transfer System handles powders under contained conditions with an extraction capacity above 4 tons per hour while keeping operator exposure below 1 µg/m³.
By Usability
Single-use systems gain strong traction because they limit cleaning time, reduce cross-contamination risk, and support agile production cycles. Many facilities shift toward disposables to match rapid batch turnover in biologics and personalized therapies. It supports faster validation cycles and greater layout flexibility. Multiple-use systems remain relevant for high-volume environments that value durability and long-term cost control. Inductors support specialized transfer needs in barrier and isolator setups that require stability during movement. Many companies balance disposable and reusable systems to match varied workflows. Strong demand for flexible operations increases interest in hybrid usability designs. The Aseptic Transfer System Market grows through wider adoption across both disposable and durable system categories.
By Transfer Type
Ports remain central due to their role in connecting chambered systems, isolators, and cleanroom zones with predictable sterility. Many drug makers invest in advanced port designs to maintain tighter seals and limit operator exposure. Portbags support efficient movement of components, stoppers, tools, and consumables during sterile processing. It improves workflow speed and reduces contamination risk during fast-paced operations. Other transfer mechanisms support niche or high-containment functions tied to specialized therapies. Many sites adopt multi-mode transfer options to handle diverse batch requirements. Strong reliance on engineered pathways strengthens quality assurance. The Aseptic Transfer System Market benefits from advancement across all transfer types.
- For instance, ChargePoint’s PharmaSafe® Pro port system achieves operator exposure limits below 1 µg/m³ for high-potency compounds, ensuring controlled transfer in OEB5 environments.
By End User
Pharmaceutical companies lead adoption due to their expanding sterile drug portfolios and global facility upgrades. Many plants deploy engineered transfer routes to support biologics, injectables, and high-potency medicines. Research laboratories adopt smaller-scale systems to manage sensitive materials during development. It helps maintain sterility during iterative testing cycles. Hospitals use protected transfer systems for compounding, oncology drug preparation, and cleanroom pharmacy operations. Biotechnology firms integrate flexible transfer designs that support rapid scale-up. Other users include contract manufacturers and diagnostic centers that depend on reliable transfer control. The Aseptic Transfer System Market expands across these end-user groups due to broader investment in controlled sterile workflows.
Segmentation:
By Product Type
- Liquid/Fluid Transfer Systems
- Solid/Powder Transfer Systems
By Usability
- Single-Use Systems
- Multiple-Use Systems
- Inductors
By Transfer Type
By End User
- Pharmaceutical Companies
- Research Laboratories
- Hospitals
- Biotechnology
- Others
By Region
- North America
- Europe
- Germany
- France
- U.K.
- Italy
- Spain
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- South-east Asia
- Rest of Asia Pacific
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Middle East & Africa
- GCC Countries
- South Africa
- Rest of the Middle East and Africa
Regional Analysis:
North America holds the largest share at nearly 38% due to strong biopharmaceutical manufacturing capacity and strict GMP-driven adoption of enclosed transfer solutions. Companies upgrade sterile workflows to support biologics, vaccines, and high-potency drug pipelines. It strengthens demand for ports, portbags, and closed transfer systems across advanced facilities. Investment in automation and single-use technologies drives higher adoption across both the U.S. and Canada. Many plants modernize contamination-control operations to support new product launches. The Aseptic Transfer System Market grows in this region due to strong regulatory expectations and continuous facility expansion.
Europe accounts for nearly 32% of the global share and maintains steady growth supported by Annex 1 updates and the expansion of sterile drug manufacturing. Regional cleanroom upgrades increase demand for engineered transfer pathways. It supports stronger dependence on isolators, RABS, and barrier-integrated transfer systems. Countries including Germany, Switzerland, and the UK lead adoption through large biologics and vaccine production networks. Many plants invest in automated ports and disposable transfer systems to reduce human contact. The Aseptic Transfer System Market benefits from structured regulatory frameworks that drive consistent investment in sterility technology.
Asia Pacific holds close to 22% of the market share and records the fastest growth due to rising pharmaceutical production and expanding biologics clusters. China, India, South Korea, and Singapore upgrade sterile facilities to meet global supply needs. It drives significant demand for scalable and cost-efficient transfer solutions. Growing contract manufacturing activity strengthens uptake across medium and large facilities. Many manufacturers deploy hybrid disposable and reusable systems to meet rapid scale-up targets. The Aseptic Transfer System Market accelerates in this region due to strong investment in vaccine and biosimilar manufacturing.
Shape Your Report to Specific Countries or Regions & Enjoy 30% Off!
Key Player Analysis:
- Getinge AB
- Syntegon Technology GmbH
- Aseptic Technologies
- STERIS plc
- Central Research Laboratories (CRL)
- Flexifill Ltd
- Chargepoint Technology
- Atec Pharmatechnik GmbH
- DESTACO
- QualiTru
- AptarGroup
- Advanta Pass
- Veltek Associates, Inc.
- Tema Sinergie S.p.A. / EMA SINERGIE S.p.A.
- Corning Incorporated
- ABC Transfer SAS
Competitive Analysis:
Competition in the Aseptic Transfer System Market centers on technological advancement, regulatory compliance, and integration strength across isolators, RABS, and cleanroom environments. Leading companies expand portfolios with closed-system architectures that reduce operator exposure and ensure tighter seal integrity. It drives competitive focus toward automation, disposable compatibility, and contamination-control performance. Firms invest in robotics and digital monitoring to enhance reliability during transfer operations. Many competitors strengthen global presence through partnerships with sterile equipment manufacturers. Smaller companies focus on niche segments such as rapid transfer ports, portbag systems, and high-containment solutions. The Aseptic Transfer System Market also shows rising competition from emerging regional players that target cost-efficient and modular system designs.
Recent Developments:
- In December 2025, Marchesini Group acquired a 44.39% equity stake in AST, a U.S. leader in drug product manufacturing, forming a strategic partnership to accelerate growth in aseptic manufacturing and packaging solutions.
- In September 2025, Aseptic Technologies launched the AT-Cellyx, an integrated device for controlled mixing and cooling in cell therapy manufacturing, with an optional in-line cell counter for concentration monitoring prior to filling.
- In May 2025, Syntegon Technology GmbH unveiled its SynTiso aseptic filling line at Pharmatag 2025, featuring 100% in-process control, robotic setup for minimal contamination risk, and capacity for up to 600 containers per minute including syringes, vials, and cartridges.
Report Coverage:
The research report offers an in-depth analysis based on Product Type, Usability, Transfer Type, End User, and Region. It details leading market players, providing an overview of their business, product offerings, investments, revenue streams, and key applications. Additionally, the report includes insights into the competitive environment, SWOT analysis, current market trends, as well as the primary drivers and constraints. Furthermore, it discusses various factors that have driven market expansion in recent years. The report also explores market dynamics, regulatory scenarios, and technological advancements that are shaping the industry. It assesses the impact of external factors and global economic changes on market growth. Lastly, it provides strategic recommendations for new entrants and established companies to navigate the complexities of the market.
Future Outlook:
- Demand rises for advanced sterile transfer designs due to expanding biologics and vaccine production pipelines in the Aseptic Transfer System Market.
- Adoption of single-use ports and transfer bags grows as facilities target flexible layouts and shorter changeover cycles.
- Automation strengthens sterility control through robotic handling and digital integrity monitoring across production lines.
- Barrier-integrated systems gain momentum as more facilities transition to isolators and RABS-driven operations.
- Growing investment in high-potency and specialty drug manufacturing increases demand for secure, closed transfer mechanisms.
- Modular plants and POD-type facilities accelerate uptake of compact and plug-in transfer platforms.
- Digital monitoring expands through sensor-enabled ports that support real-time quality checks and improved audit readiness.
- Hybrid transfer systems combining disposable and reusable components gain interest across multi-product environments.
- Rapid expansion of contract manufacturing encourages adoption of scalable transfer pathways that fit varied production loads.
- Emerging markets invest heavily in sterile manufacturing upgrades, driving broader global penetration for next-generation transfer systems.




